Chapter 7 7 2 Ionic Bonds Ionic Compounds

Chapter 7 7. 2: Ionic Bonds & Ionic Compounds

The Formation of Ionic Bonds n Objectives n Describe the formation of ionic bonds and the structure of ionic compounds. n Generalize about the strength of ionic bonds based on the physical properties of ionic compounds. n Categorize the formation of an ionic bond as exothermic or endothermic.

The Formation of Ionic Bonds n A positively charged cation is attracted to a negatively charged anion. n The electrostatic force of attraction holding oppositely charged particles together is called an IONIC BOND.

Ionic Compound n Defined as a compound having an ionic bond n It is a compound that is composed entirely of ions yet has a “ 0” overall charge.

Ionic Compounds n. A binary ionic compound contains only 2 different elements – a metal cation and a nonmetal anion (ex. Na. Cl)

Forming Ionic bonds n Even though they contain charged particles, ionic compounds are electrically neutral. the number of electrons lost by a cation must equal the number of electrons gained by the anion. n This means, n The electrical charges of the cations and anions must balance (the total of all the charges has to add up to zero).
![Example: Na. Cl See Table 4, p. 211 Na Cl + [Na] [ Cl Example: Na. Cl See Table 4, p. 211 Na Cl + [Na] [ Cl](http://slidetodoc.com/presentation_image_h/9c5566e6d5bacc1cec74a68440581665/image-7.jpg)
Example: Na. Cl See Table 4, p. 211 Na Cl + [Na] [ Cl ] Na. Cl Showing ionic compound formation using electron dot structures

The Formation of Ionic Bonds n Consider the formation of the ionic compound, calcium fluoride, using electron configurations. n Calcium is a group 2 A metal: [Ar] 4 s 2 How many electrons will calcium lose to gain a noble gas configuration? n What is its charge? n n Fluorine is a group 7 A nonmetal: [He] 2 s 22 p 5 n How many electrons will fluorine gain to attain a noble gas configuration? n What is its charge?

The Formation of Ionic Bonds n The number of electrons lost must equal the number of electrons gained; the total of the charges must add to zero. The # of electrons lost does not equal the # gained. n The total of the charges does not equal 0 (+2 + -1 = +1). n n Therefore, another F ion is needed – each F will gain 1 electron, for a total of two. That means, the # of electrons lost will equal the # gained; the total of the charges will be 0.

The Formation of Ionic Bonds n One Ca+2 will form an ionic bond with two F-. n The formula for the compound will be Ca. F 2 – the subscript of 2 below F means there are 2 F- in the compound. n The name of the compound will be a combination of the names of the 2 ions that make it up - calcium fluoride. n Practice worksheet

Properties of Ionic Compounds n The cation & anion have a regular, repeating pattern; forming an ionic crystal n There is no such thing as 1 unit of Na. Cl – Large numbers of ions are found together in a crystal, in a ratio that balances the charges. n For example, in Na. Cl, cations and anions are in a 1: 1 ratio
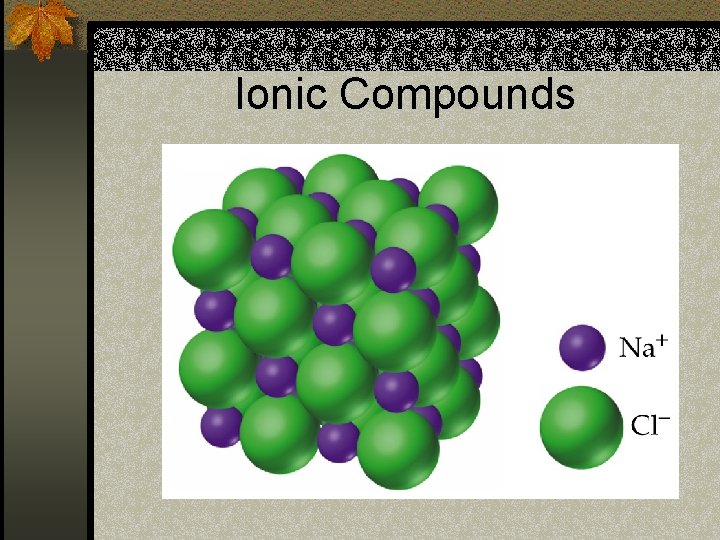
Ionic Compounds

Ionic Compounds n The formula, Na. Cl, is an empirical formula – through the subscripts, it gives the smallest possible ratio of ions – 1 Na+ to 1 Cln This ratio is determined by the number of electrons transferred in ionic bonding

Ionic Crystals n Each type of crystal has a characteristic shape. Na. Cl Calcium carbonate

Ionic crystal n The 3 -d geometric arrangement of particles is called a crystal lattice. n Ionic compounds have different crystal lattice structures because they have differently charged ions making up that structure.
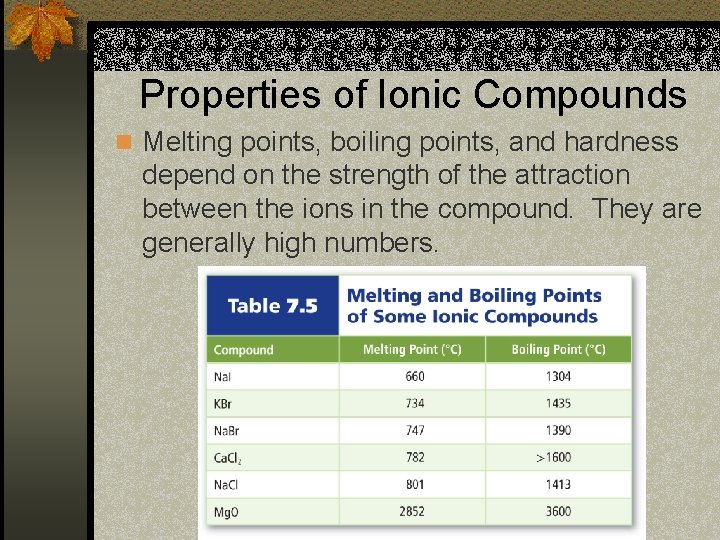
Properties of Ionic Compounds n Melting points, boiling points, and hardness depend on the strength of the attraction between the ions in the compound. They are generally high numbers.

Properties of Ionic Compounds n Dissolve readily in water because they dissociate: Na. Cl --> Na+(aq)+ Cl-(aq) n In a solid, ions are locked into position and electrons cannot flow freely—solid ions are poor conductors of electricity. n Ionic compounds, in a liquid state or in aqueous solution, are good conductors of electricity since electrons can move freely. n Ions in aqueous solution that conduct electricity are called electrolytes.

Properties of Ionic Compounds n Ionic crystals are hard, yet they are brittle. n This figure demonstrates how and why crystals break when an external force is applied.

The Formation of Ionic Bonds n During any chemical reaction, energy is either absorbed or released. n If energy is released, the reaction is exothermic.

The Formation of Ionic Bonds n The formation of ionic compounds from positive and negative ions is almost always exothermic. n The attraction of the positive ion for the negative ion forms a more stable system that is lower in energy than the individual ions.

The Formation of Ionic Bonds n The energy required to separate one mole* of ions in an ionic compound is referred to as the lattice energy. n The more negative the lattice energy, the stronger the force of attraction, the harder to separate the ions. n * I mole = 6. 02 x 1023 ions

Trends in Lattice Energy n In smaller ions, the ions are closer in space and require a greater lattice energy to separate. See Na. F vs. Na. I in Table 6, p. 217 n Compounds of ions with larger ionic charges will have greater lattice energy. Compare Na. Cl and Sr. Cl 2
- Slides: 22