Chapter 6 The Periodic Table 1 Mendeleevs Periodic

Chapter 6 The Periodic Table 1

Mendeleev’s Periodic Table (3) properties (physical & chemical) of the elements repeat in an orderly way from row to row of the table. Periodicity is the tendency to recur at regular intervals. 2

Quick-write Why isotopes of an element have the same chemical properties? 3
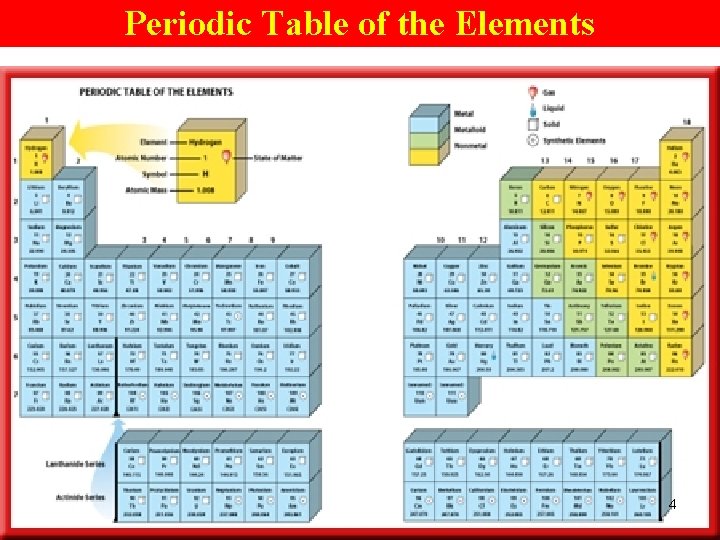
Periodic Table of the Elements 4
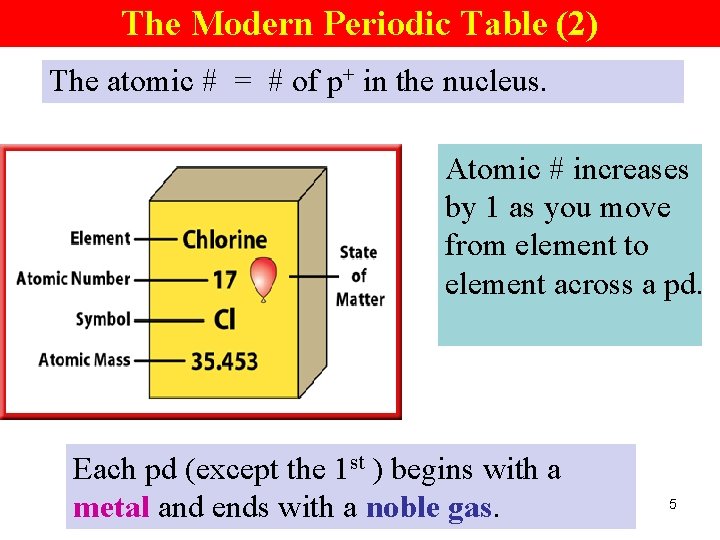
The Modern Periodic Table (2) The atomic # = # of p+ in the nucleus. Atomic # increases by 1 as you move from element to element across a pd. Each pd (except the 1 st ) begins with a metal and ends with a noble gas. 5
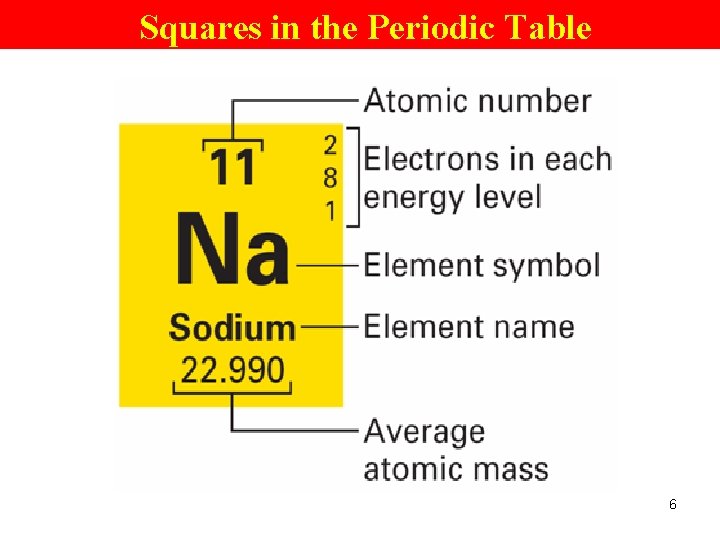
Squares in the Periodic Table 6

The Modern Periodic Table (3) • The properties of the elements change in an orderly progression from left to right. • The pattern in properties repeats after group 18 (VIIIA). This regular cycle illustrates periodicity in the properties of the elements. 7

The Periodic Law • states that the physical and chem properties of the elements repeat in a regular pattern when they are arranged in order of increasing atomic #. 8

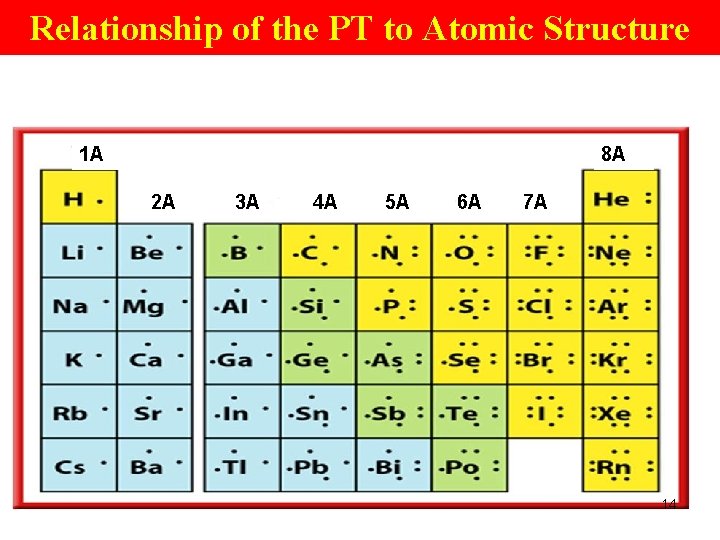
Relationship of the PT to Atomic Structure Periodic Table • elements are arranged according to atomic # • atomic # tells the # of p+ (and e- too) it has. 9

Relationship of the PT to Atomic Structure • The lineup starts with H, which has 1 e-. • He comes next in the 1 st pd because He has 2 e-. Li has 3. 10

Relationship of the PT to Atomic Structure • Notice on the PT that Li starts a new period. • Why does the 1 st pd have only 2 elements? Only 2 e- can occupy the 1 st EL in an atom. The 3 rd e- in Li must be at a higher EL. 11

Relationship of the PT to Atomic Structure • Li starts a new period at the far left in the table and becomes the 1 st element in a gp. A group • elements in a vertical column. • Elements have similar chem properties. • Elements in the ‘A’groups have same # valence e- 12
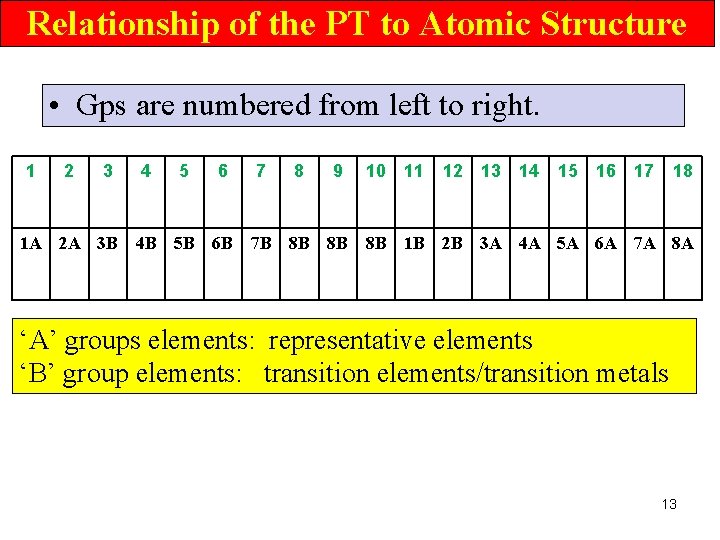
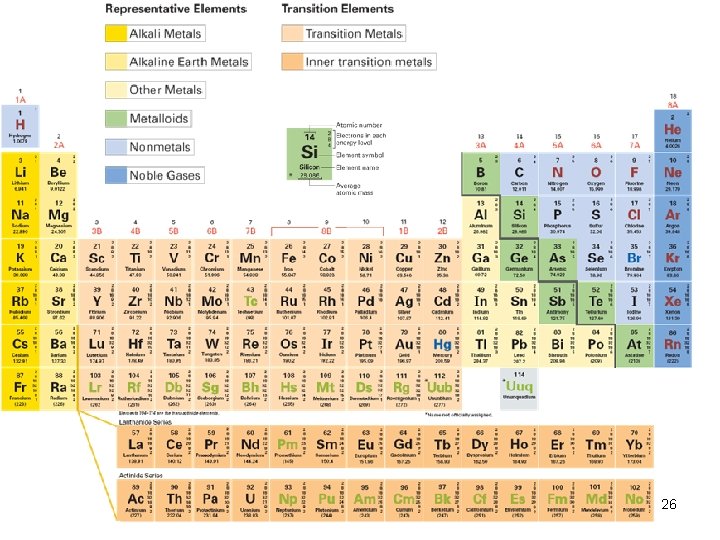
Relationship of the PT to Atomic Structure • Gps are numbered from left to right. 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 1 A 2 A 3 B 4 B 5 B 6 B 7 B 8 B 8 B 8 B 1 B 2 B 3 A 4 A 5 A 6 A 7 A 8 A ‘A’ groups elements: representative elements ‘B’ group elements: transition elements/transition metals 13
Relationship of the PT to Atomic Structure 1 A 8 A 2 A 3 A 4 A 5 A 6 A 7 A 14

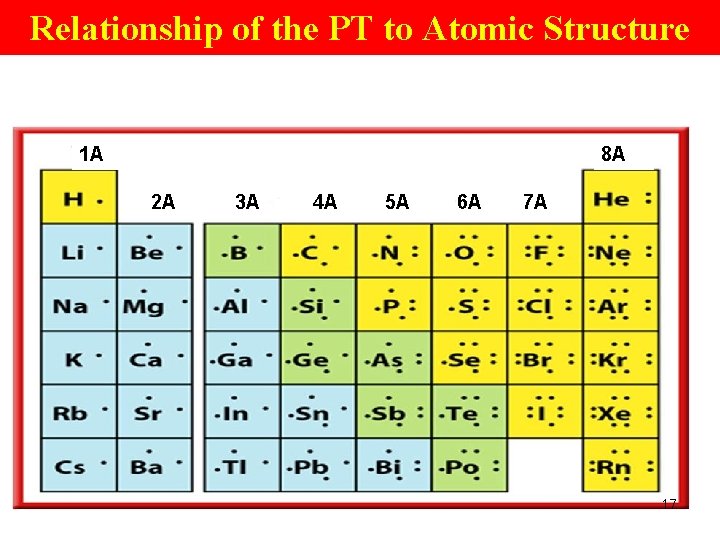
Atomic Structure of Elements Within a Pd (2) • Every period starts with a gp 1 element (H, Li, Na, K, . . . ) • has 1 e- at a higher EL than the noble gas of the preceding pd. • has 1 valence e-. 15

Atomic Structure of Elements Within a Pd • Move across a period to the next…, the # of valence e- increases by 1. • Gp 18 elements have the max # of 8 valence e- in their outermost EL. 16
Relationship of the PT to Atomic Structure 1 A 8 A 2 A 3 A 4 A 5 A 6 A 7 A 17

Atomic Structure of Elements Within a Pd • Gp 18 (VIIIA) elements --- noble gases. • The noble gases, with a full complement of valence e-, are generally unreactive. Octet configuration→most stable econfiguration except He (duplet) 18

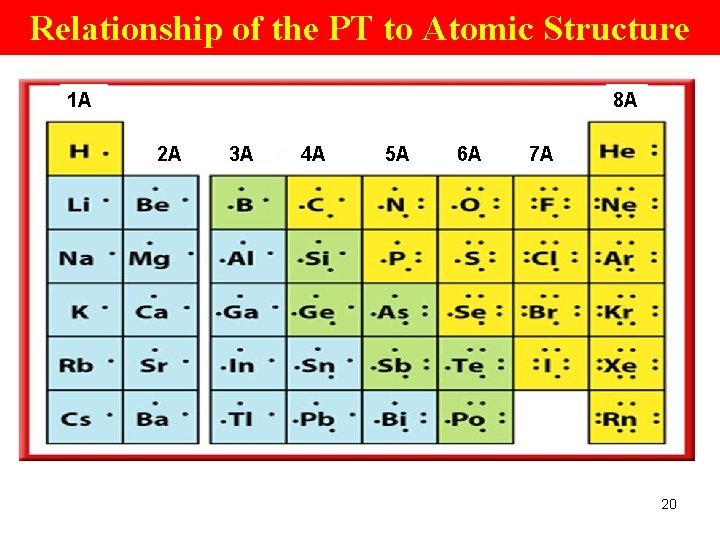
Atomic Structure of Elements Within a Pd The period # (1 -7) of an element is the same as the # of its highest EL, • so the valence e- of an element in the 2 nd period are in the 2 nd EL. • A period 3 element such as Al has its valence e- in the 3 rd EL. 19
Relationship of the PT to Atomic Structure 1 A 8 A 2 A 3 A 4 A 5 A 6 A 7 A 20

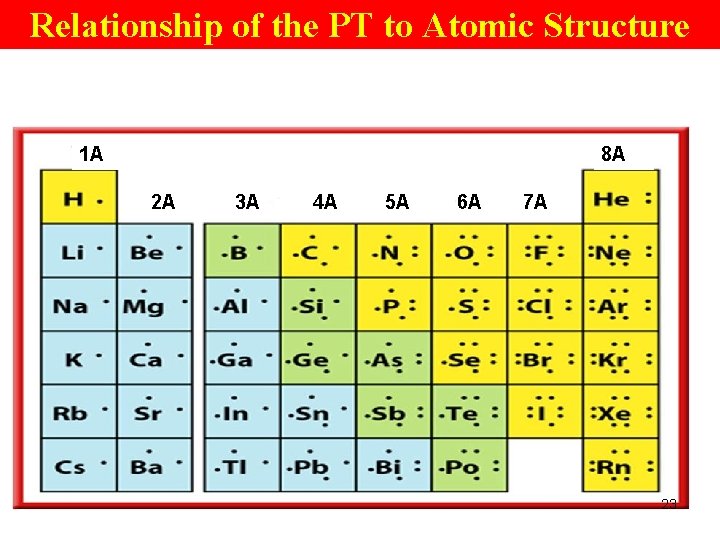
Atomic Structure of Elements Within a Gp • # of valence e- changes from 1 to 8 from left to right across a pd; • After Gp 18, the pattern repeats. • For the main gps (A) elements, the gp # is related to the # of valence e-. • The main gp elements are those in Gps 1(1 A), 2(A), 13(3 A), 14(4 A), 15(5 A), 16(6 A), 17(7 A), and 18(8 A). 21

Atomic Structure of Elements Within a Gp • For elements in gps 1(1 A) and 2(2 A), 4(4 A), the gp # = # valence e-. • For elements in gps 13(3 A), 14(4 A), 15(5 A), 16(6 A), 17(7 A), and 18(8 A), the 2 nd digit in the gp # is = to the # valence e-. 22
Relationship of the PT to Atomic Structure 1 A 8 A 2 A 3 A 4 A 5 A 6 A 7 A 23

Atomic Structure of Elements Within a Gp • Because elements in the same gp have the same # of valence e-, → similar chemical properties. • Na is in Gp 1 → 1 valence e-. • Because other elements in gp 1 also have 1 valence e-, → similar chem properties. 24

Atomic Structure of Elements Within a Gp • Cl is in Gp 17 (7 A) and has 7 valence e-. • All the other elements in gp 17 also have 7 valence e- → similar chem properties. • Throughout the PT, elements in the same gp have similar chem properties because the have the same # of valence e-. 25
6. 2 26

Atomic Structure of Elements Within a Gp 5 categories: Ø alkali metals in Group 1(6; except H), Ø alkaline earth metals in Group 2 (6), Ø halogens in Group 17(VIIA), and Ø noble gases in Group 18(VIIIA) (octet). Ø transition metals (B groups) 27

Atomic Structure of Elements Within a Gp Because the PT relates gp and pd # to valence e-, it’s useful in predicting atomic structure and, chem properties. 28

Atomic Structure of Elements Within a Gp • e. g. O, in Gp 16(6 A) and Pd 2, has 6 valence e - (the same as the 2 nd digit in the gp #), and these e- are in the 2 nd EL (because O is in the 2 nd pd). • O has the same # of valence e- as all the other elements in Gp 16(6 A) → similar chem properties. 29
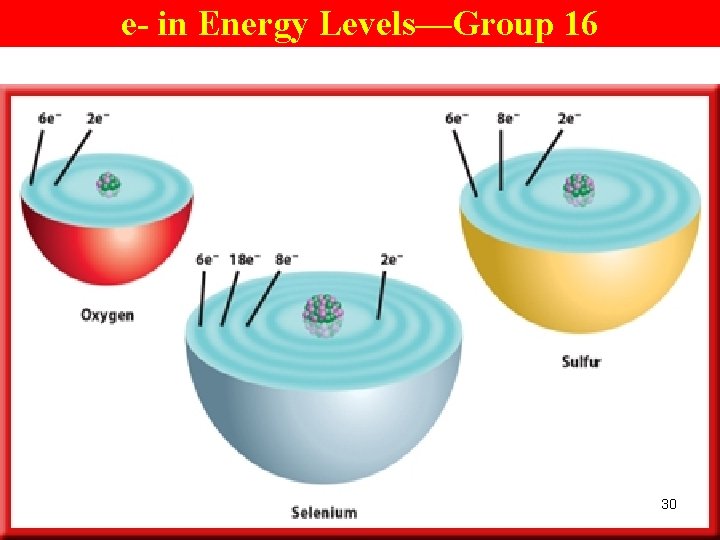
e- in Energy Levels—Group 16 30

Physical States and Classes of the Elements • close to 80% (≈90) of the elements are metals occupying the entire left side (except H) and center. • All metals except Hg exist as solid at ‘usual’ temps • Nonmetals occupy the upper-rt-hand corner. • 18 nonmetals; 11 gases at ‘usual’ temp • Metalloids (7) : located along the boundary between metals and nonmetals. 31

Physical States and Classes of the Elements are classified as basis on physical and chem properties • Metals (alkali, alkaline earth, transition) • metalloids, or • Nonmetals 32
elements metalloids 33

Metals Ø luster, Ø conduct heat and electricity Ø usually bend without breaking Ø malleable Ø ductile With the exception of Sn, Pb, and Bi, metals have 1, 2, or 3 valence e-. 34

Metals (2) • All metals except Hg are solids at ‘usual’ temp; • Hg is a liquid at ‘usual’ temperature • most have extremely high m. p. and b. p. m. p. of Fe : about 1700°C 35
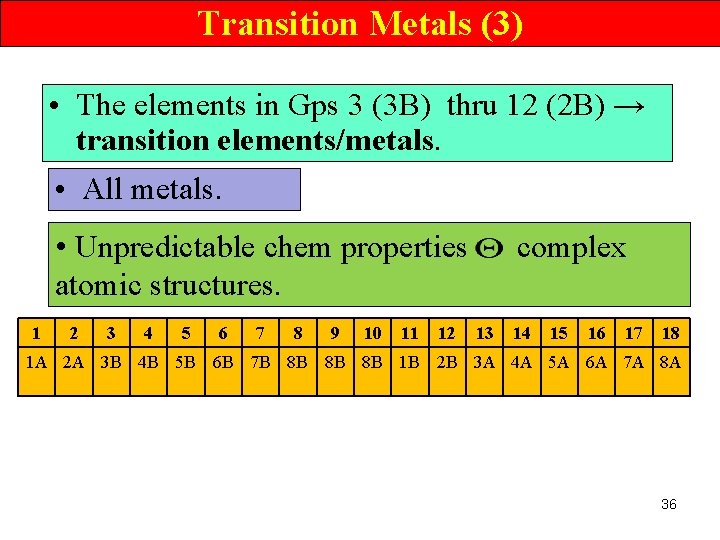
Transition Metals (3) • The elements in Gps 3 (3 B) thru 12 (2 B) → transition elements/metals. • All metals. • Unpredictable chem properties atomic structures. 1 2 3 4 5 6 7 8 9 10 11 12 complex 13 14 15 16 17 18 1 A 2 A 3 B 4 B 5 B 6 B 7 B 8 B 8 B 8 B 1 B 2 B 3 A 4 A 5 A 6 A 7 A 8 A 36
Transition Metals (5) • 2 series of elements (58 -71 and 90 -103) are placed below the main body of the table. • separated from the main table very wide. the table • inner transition elements. 37
Transition Metals (6) • The 1 st series (lanthanides) element # 57, lanthanum. follow • 14 elements • rare earth elements (0. 01 %). • similar properties. 38

Metals (7) • The 2 nd series of inner transition elements, the actinides • radioactive • none beyond U (92) occur in nature. 39

lanthanides and actinides • Unpredictable chem properties • complex atomic structures. 40

Nonmetals (1) Some nonmetals are abundant in nature • The nonmetals O and N make up 99 % of Earth’s atm. 41

Nonmetals (2) C is found in more cpds than all the other elements combined. The many cpds of C, N, and O are important in a wide variety of applications. 42

Nonmetals (3) nonmetals (most) § don’t conduct electricity and heat § are brittle when solid. § Many are gases (11) at ‘usual’ temp 43

Nonmetals (4) § Their m. p. and b. p. are lower than those of metals. § With the exception of C, nonmetals have 5, 6, 7, or 8 valence e-. § lack the luster of metals. 44
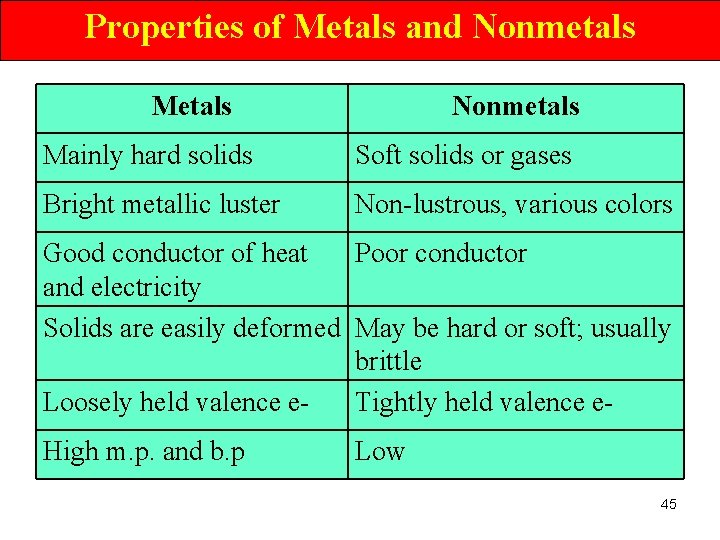
Properties of Metals and Nonmetals Metals Nonmetals Mainly hard solids Soft solids or gases Bright metallic luster Non-lustrous, various colors Good conductor of heat Poor conductor and electricity Solids are easily deformed May be hard or soft; usually brittle Loosely held valence e. Tightly held valence e. High m. p. and b. p Low 45

Metalloids • have some chemical and physical properties of metals and nonmetals. • lie along the border between metals and nonmetals. 46

Metalloids • Si is probably the most well-known metalloid (computer chips) • six others 47

Metalloids (3) A semiconductor • is an element that • does not conduct electricity as good as a metal, • but does conduct slightly better than a nonmetal. 48

Semiconductors and Their Uses • TV, computer, handheld electronic games, MP 3, cell phones, and calculator are electrical devices 49

Semiconductors and Their Uses Ø metals generally are good conductors of electricity, Ø nonmetals are poor conductors, Ø semiconductors fall in between the 2 extremes. 50
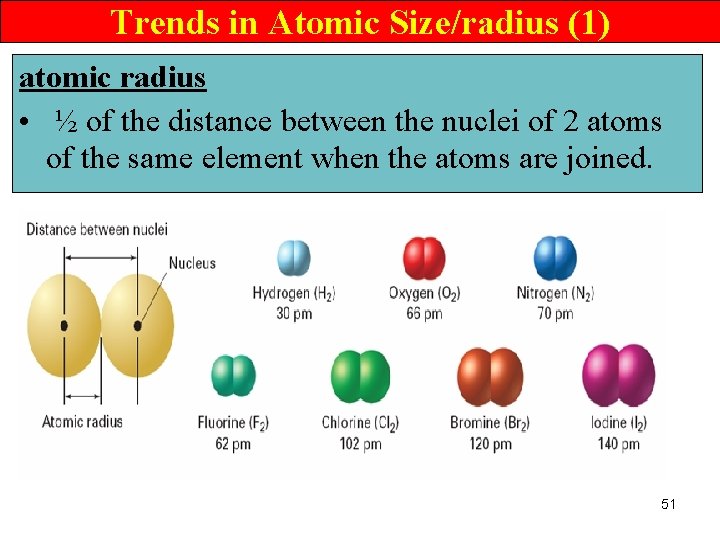
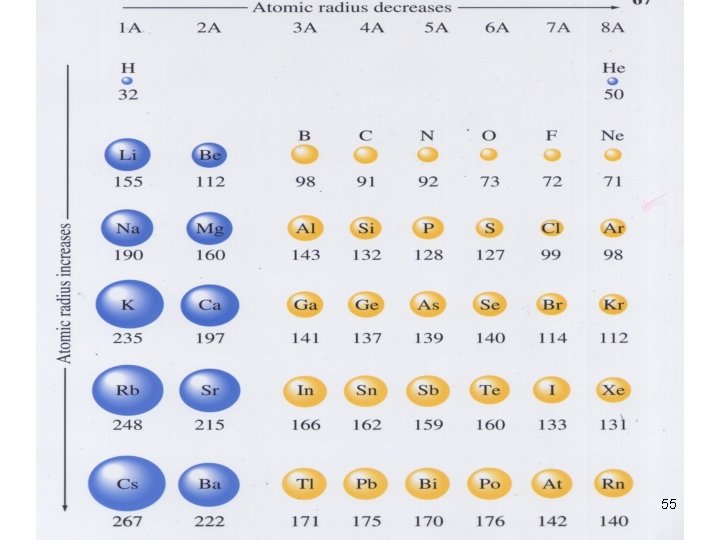
Trends in Atomic Size/radius (1) atomic radius • ½ of the distance between the nuclei of 2 atoms of the same element when the atoms are joined. 51

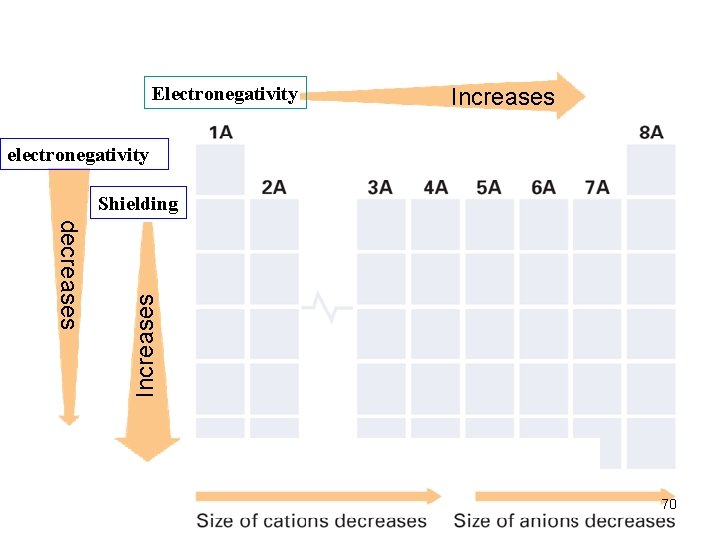
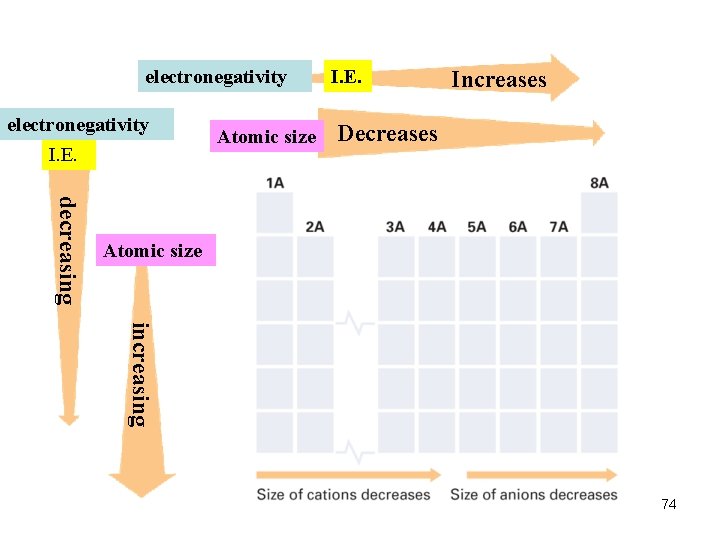
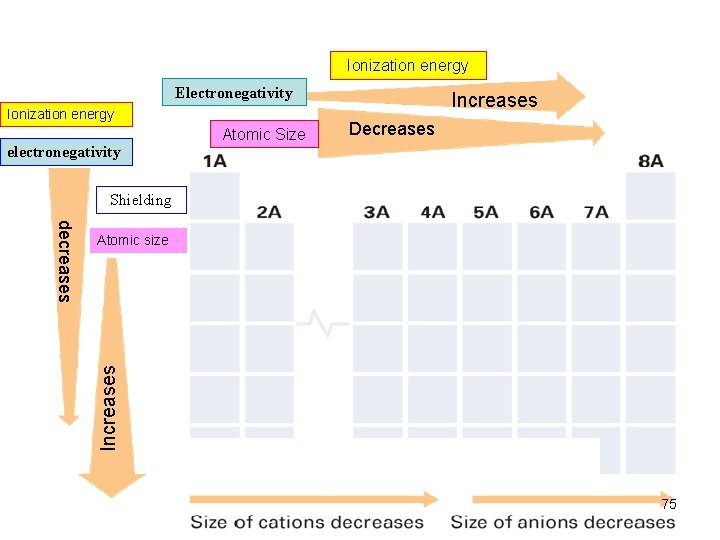
Trends in Atomic Size/radius (2) Group Trend Øincreases from top to bottom within a gp and Periodic Trend Ødecreases from left to right across a pd. 52

Shielding Effect Shielding effect • go down a gp of the PT, elements possess more and more EL/subshells/orbitals • EL/subshells/orbitals are blocking the attractions of the e- in the outer shells by the nucleus. • Also the e- in the inner shells are repelling the ein the outer shells preventing them to move closer to the nucleus. 53
Trends in Atomic Size/radius Atomic Size/radii decreases 54
55
Group and periodic trend Atomic sizes/radii decreasing Atomic sizes/radii increasing 56

Explain Periodic Trend—atomic sizes Periodic Trend Ø e. g. Li and F Øsame EL and # orbitals Ø i. e. same shielding effect Ø # p+ increases Ø nuclear attraction increases Ø pulls e- towards the nucleus Ø smaller (atomic radii…) 57

Explain Group Trend—atomic sizes Group trend • # p+ increases; stronger nuclear attraction • # EL & orbitals increases; higher shielding effect on the nuclear attraction • Shielding effect is more significant than the effect of increase in nuclear attraction • Bigger (atomic radii …. ) 58
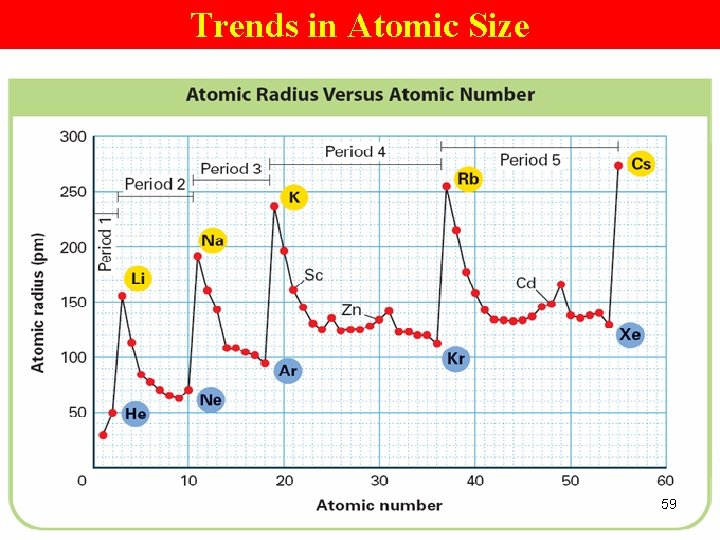
Trends in Atomic Size 59

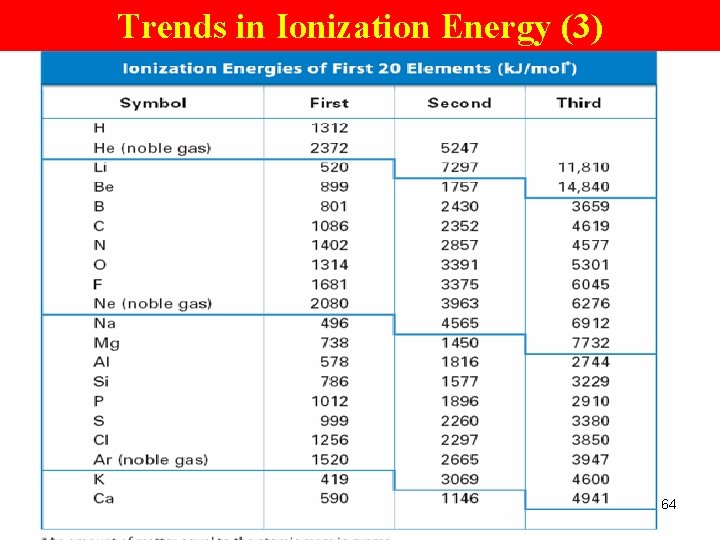
Trends in Ionization Energy (1) Ionization energy Ø amt of energy required to remove an e- from an atom in gaseous state Ø e. g. Na → Na+ + e. Na atom Na ion Valence e- • 1 st I. E. : energy required to remove the 1 st e- from an atom. • 2 nd I. E. : energy required to remove an e- from an ion with a 1+ charge. (needs a lot more energy) 60

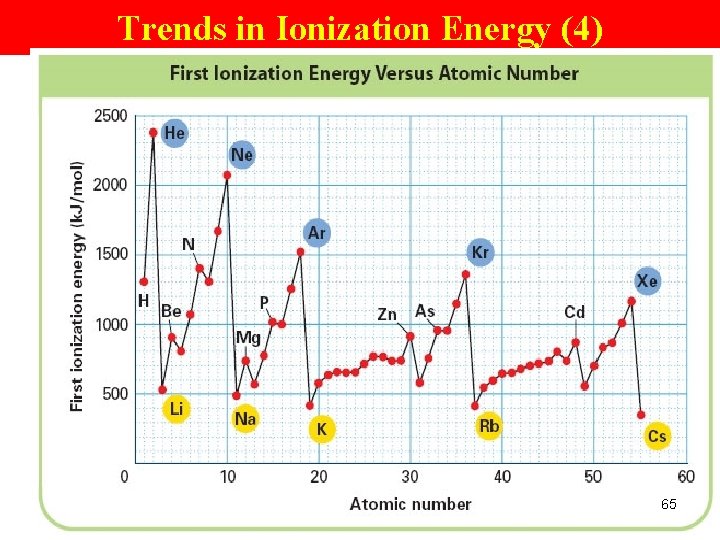
Trends in Ionization Energy (2) Periodic Trend Ø increases across a pd Group Trend Ø decreases down a gp (means less energy is used to remove an e-. ) 61

Group and period trend Ionization energy increasing Ionization energy decreasing 62
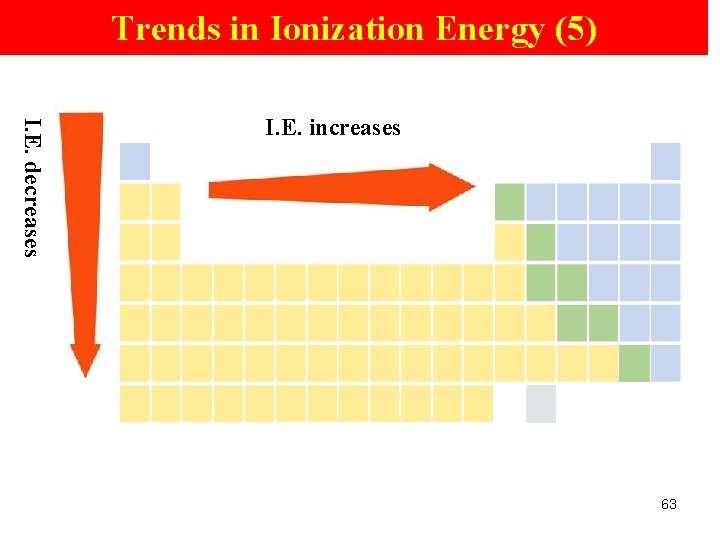
Trends in Ionization Energy (5) I. E. decreases I. E. increases 63
Trends in Ionization Energy (3) 64
Trends in Ionization Energy (4) 65

Explain Period Trend—I. E. (7) Periodic Trend • shielding effect remains the same (why? ) • # p+ increase • stronger nuclear attraction • atom size decreases • valence e- closer to the nucleus • more tightly attracted • not so likely to lose • need more energy to remove an e • Higher I. E. 66

Explain group trend—I. E. (6) Group Trend Ø shielding effect increases (why? ) Ø atomic sizes increases (more EL & orbitals) Ø valence e- are farther from nuclei Ø valence e- not so tightly attracted/held Ø easier to lose Ø lower I. E. 67

Trends in Electronegativity (1) Electronegativity (e- affinity) (of an element) Ø the ability of an atom of an element to attract e - when the atom is in a cpd. e. g. in a H 2 O molecule, the e- of H atoms move closer to the O atom (attracted by the nucleus of O atom; O is more electronegative Group Trend ØDecreases down a gp. Periodic Trend Ø increases across a pd. 68
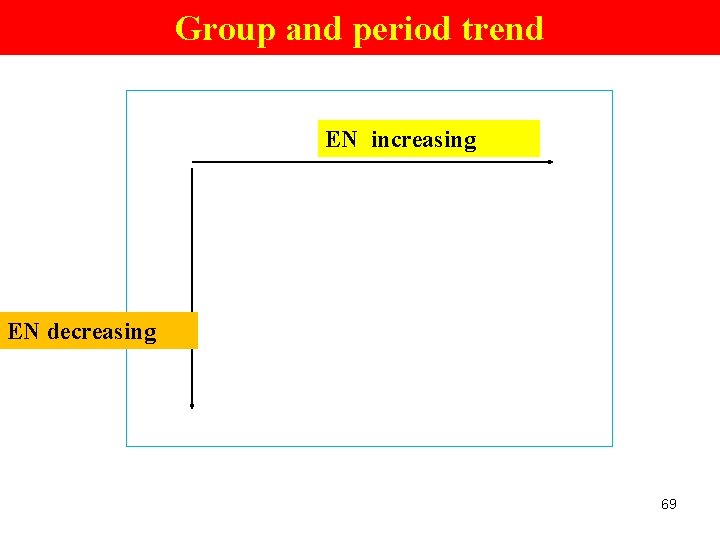
Group and period trend EN increasing EN decreasing 69
Electronegativity Increases electronegativity decreases Increases Shielding 70

Explain group Trend—Electronegativity(2) Group Trend • shielding effect increases (why? ) • nuclear attraction becomes less significant (why? ) • atoms are getting bigger • the nucleus is farther from the surface of the atom. • nuclear attraction not strong enough to attract more outside e- (e- of other atoms in a cpd) 71

Explain Periodic Trend—Electronegativity(3) Periodic Trend Ø same # EL and orbitals→same shielding effect Ø # p+ increase→ increase in nuclear attraction Ø atom smaller; nucleus closer to the surface of atom Ø more likely to attract e- of another atom in the cpd Ø higher EN 72
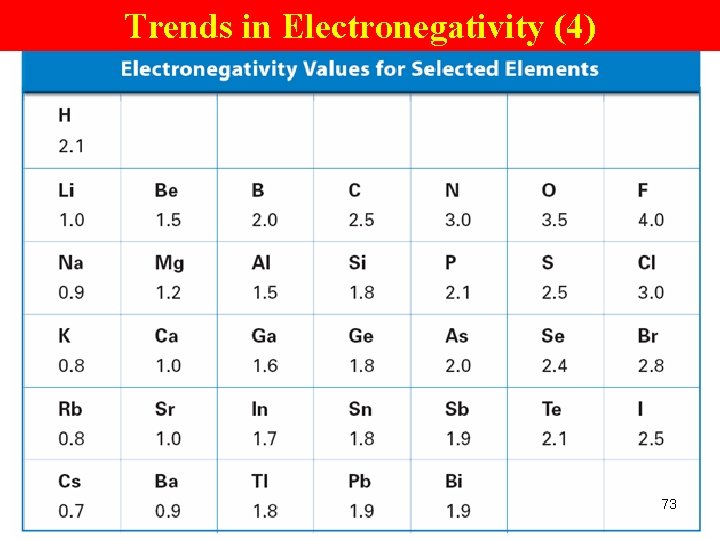
Trends in Electronegativity (4) 73
electronegativity I. E. Atomic size I. E. Increases Decreases decreasing Atomic size increasing 74
Ionization energy Electronegativity Ionization energy electronegativity Atomic Size Increases Decreases Shielding Increases decreases Atomic size 75

Ions An ion • an atom or group of atoms that have a +ve or –ve charge • formed when e- are transferred between atoms • Cation (+ve) and anion (-ve) 76

Cations and Anions Group IA, IIIA elements (metals) lose eto form cations. Na → Na+ + e. Na atom Mg → Na ion Mg 2+ + 2 e- Mg ion Al → Al 3+ + 3 e- Cations: • +vely charged ions • metals form cations by losing valence e- 77

Cations and Anions Group VA, VIA and VIIA (nonmetals) form ions by gaining e-. N + 3 e- → N 3 N atom O + Nitride ion 2 e- → Oxygen atom Cl + Chlorine atom O 2 Oxide ion e- → Cl. Chloride ion Anions • -vely charged ions • nonmetals form anions by gaining e- 78
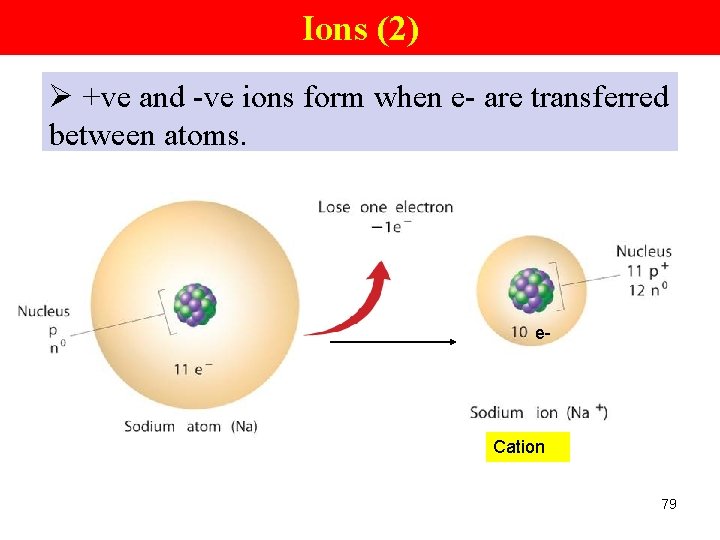
Ions (2) Ø +ve and -ve ions form when e- are transferred between atoms. e- Cation 79
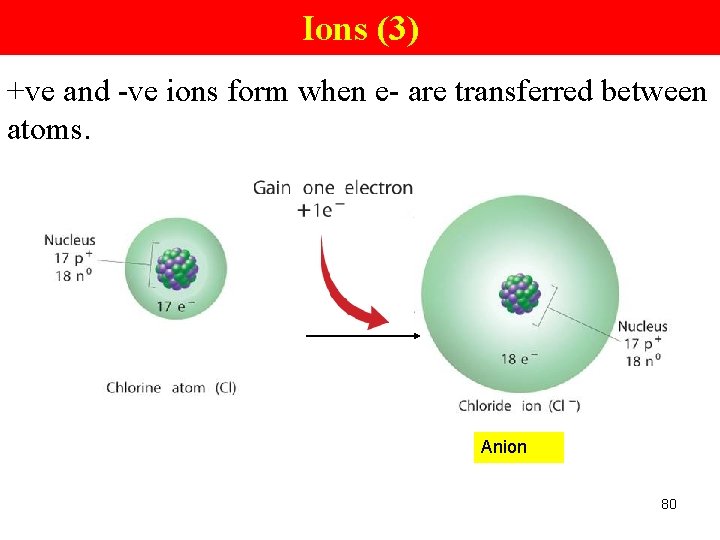
Ions (3) +ve and -ve ions form when e- are transferred between atoms. Anion 80

CST Problem 1 Which of the following atoms has six valence electrons? A magnesium (Mg) B silicon (Si) C sulfur (S) D argon (Ar) 81

CST problem 2 Which of the following is a monatomic gas at STP? A chlorine B fluorine C helium D nitrogen 82

CST problem 3 Which of the following elements has the same Lewis dot structure as silicon? A germanium (Ge) B aluminum (Al) C arsenic (As) D gallium (Ga) 83

CST problem 3 Which of the following ordered pairs of elements shows an increase in atomic number but a decrease in average atomic mass? A Ag to Pd B Co to Ni C Ge to Sn D Cr to Mo 84

CST problem 4 Iodine would have chemical properties most like A B C D manganese (Mn) tellurium (Te) chlorine (Cl) xenon (Xe) 85

CST problem 5 Graph showing ionization energy changes with atomic number 86

CST problem 5 (cont. ) The chart above shows the relationship between the first ionization energy and the increase in atomic number. The letter on the chart for the alkali family of element is A W. B X. C Y. D Z. 87

The End 88

Transition Elements 2 types of transition elements--classified based on their e-confign. Øtransition metals and Øinner transition metals. 89
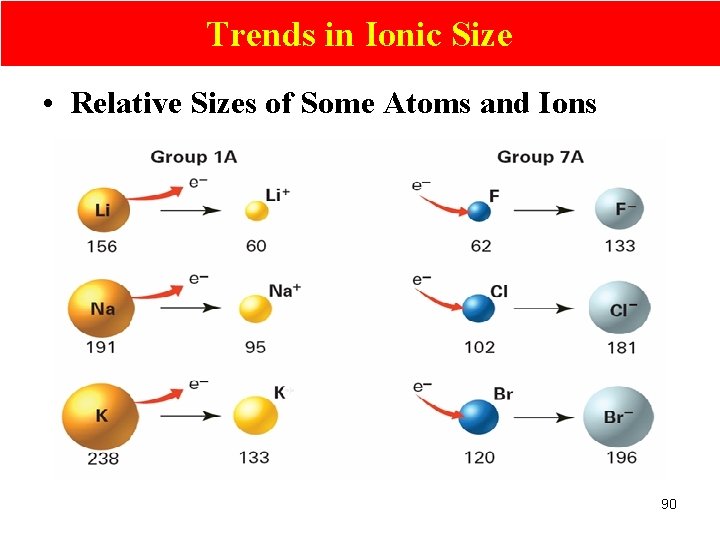
Trends in Ionic Size • Relative Sizes of Some Atoms and Ions 90
- Slides: 90