Chapter 6 Section 3 Periodic Trends Atomic Radius








- Slides: 11

Chapter 6 Section 3 Periodic Trends

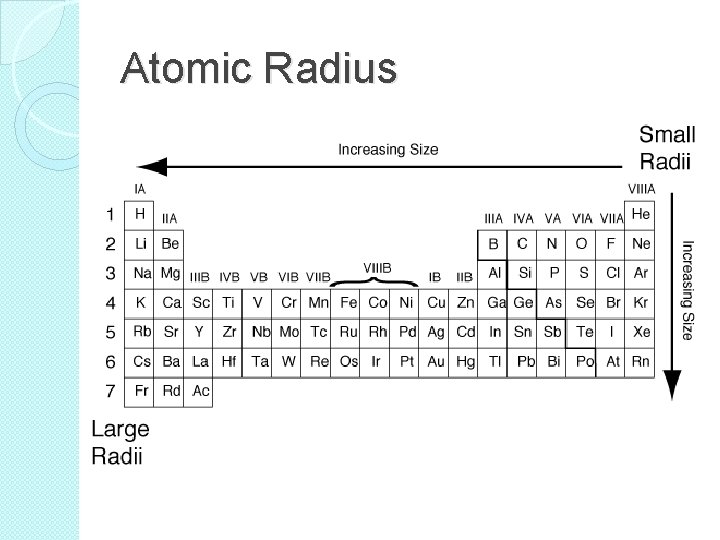
Atomic Radius �Many properties of elements tend to change in a predictable way. �Atomic Size is one of those properties. �In general, there is an increases in atomic radius as you move from right to left across a period. �Also, there is an increase in atomic radius as you move down a group.

Atomic Radius

Ionic Radius �A quick note about ions: Atoms can gain or lose electrons to form ions. �An ion is an atom or a bonded group of atoms that has a positive or negative charge. �When atoms lose electrons and form positive ions, they become smaller. �When atoms gain electrons and form negative ions, they become larger.

Ionization Energy �To form a positive ion, an electron must be removed from a neutral atom. This requires energy. �The energy is needed to overcome the attraction between the positive charge of the nucleus and the negative charge of the electron. �Ionization energy is defined as the energy required to remove an electron from a gaseous atom.

Ionization Energy �Think of ionization energy as how strongly an atom’s nucleus holds onto its electrons. �High ionization energy indicates the atom has a strong hold on its electrons. �These atoms will be less likely to form positive ions.

Ionization Energy �Low ionization energy indicates the atom loses its electrons easily. �These atoms are likely to form positive ions. �After removing the first electron, it is possible to remove another. This is known as the second ionization energy.

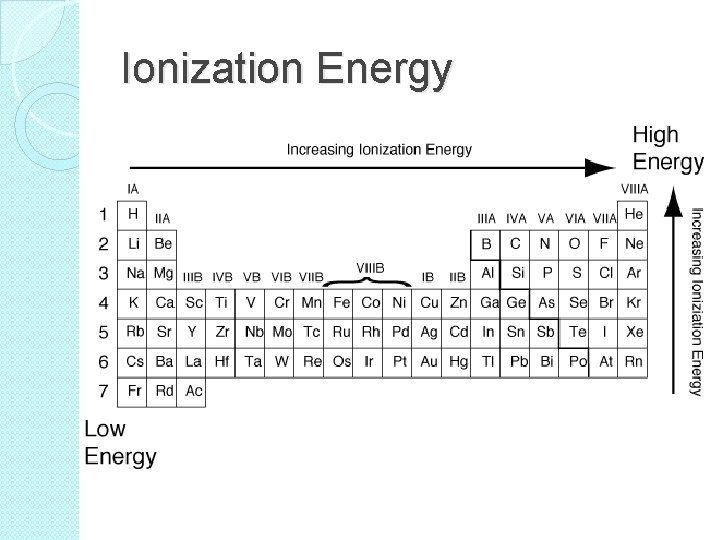
Ionization Energy �Ionization energy generally increases as you move from left to right across a period. �Ionization energy generally increase as you move up a group.

Ionization Energy

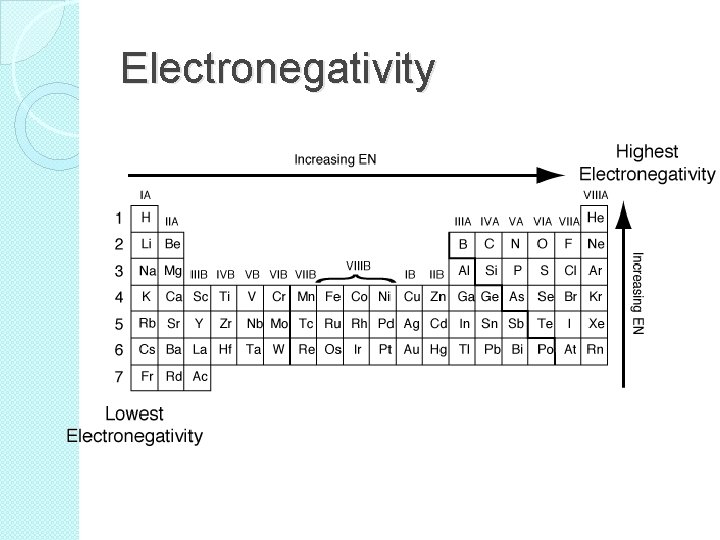
Electronegativity �The electronegativity of an element indicates the relative ability of its atoms to attract electrons in a chemical bond. �Electronegativity generally increases as you move up a group. �Also, electronegativity generally increases as you move left to right

Electronegativity