Chapter 6 Section 2 Covalent Bonding and Molecular

Chapter 6 Section 2 Covalent Bonding and Molecular Compounds

Molecule n Neutral group of atoms n Held together by covalent bond n Individual unit n Able to exists on its own n May be same element joined together n Could be two or more elements joined together
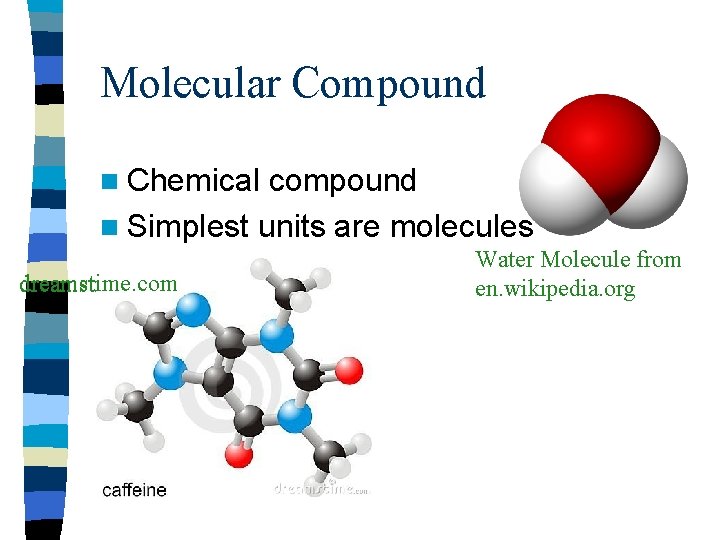
Molecular Compound n Chemical compound n Simplest units are molecules dreamstime. com Water Molecule from en. wikipedia. org

Chemical Formula n Tells the number of atoms of each kind in a chemical compound n Uses the symbols for the atoms from the periodic table n Uses subscripts to tell how many of each element are needed n Diatomic molecules have only two atoms

Molecular Formula n Shows the types of atoms n Shows the number of atoms in a single molecule n C 12 H 22 O 11 n What is this formula for? n Sucrose AKA table sugar

Formation of a Covalent Bond n Two atoms are far apart n Overall potential energy is zero n Atoms approach each other n Charged parts of the atoms begin to interact n Nuclei and electrons attract each other n Total potential energy drops

Formation of a Covalent Bond Continued n At the same time the attraction is happening n The positive part of the nuclei repel each other n The negative electrons repel each other n This increases the potential energy n Strength of attraction depends on repulsion and distance
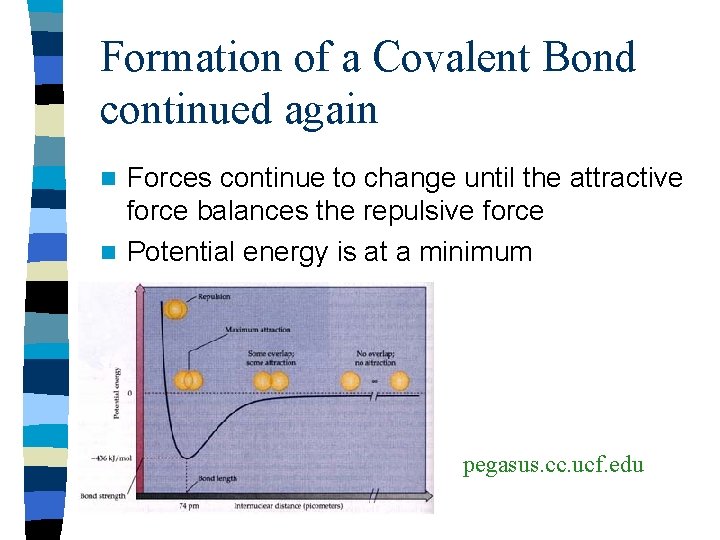
Formation of a Covalent Bond continued again Forces continue to change until the attractive force balances the repulsive force n Potential energy is at a minimum n pegasus. cc. ucf. edu

Characteristics of the Covalent Bond n Bond length-distance between the nuclei of two bonded atoms n Bond energy-energy required to break a chemical bond and form neutral isolated atoms

The Octet Rule n Noble-gas atoms exist unbonded in nature n Stable because of their completely filled outer shell n S and p are completely filled totaling 8 electrons n Other main group elements share, lose or get electrons to make a full octet

Exceptions to the Octet Rule n n n Boron has just 3 valence electrons Tends to form compounds that give it just 6 total valance electrons Other elements might exceed the 8 electrons Phosphorous in PF 5 Sulfur in SF 6 Usually when bonded to highly electronegative elements

Electron Dot Notation n Only need to use the outermost energy level electrons n Need a way to keep track of these electrons n Electron-dot notation is that way n It is yet another electron notation n It is okay to scream, I understand

Electron Dot Notation n n n n Only valence electrons of an atom are shown Dots are used to represent electrons Placed around elements symbol Number of valence electrons can be found from the periodic table The column is the same as the valence electrons Start on the left first column has 1, second column has 2 Skip the transitions Continue to count across

Example 3 n Write the electron dot notation for hydrogen. middleschoolchemistry. com

Example 4 n Write the electron dot notation for nitrogen.

Lewis Structures n Formulas in which atomic symbols represent nuclei and inner shell electrons n Dot pairs of dashes between atoms are electrons n Dots singles are unshared electrons

Lewis Structures thenakedscientists. com
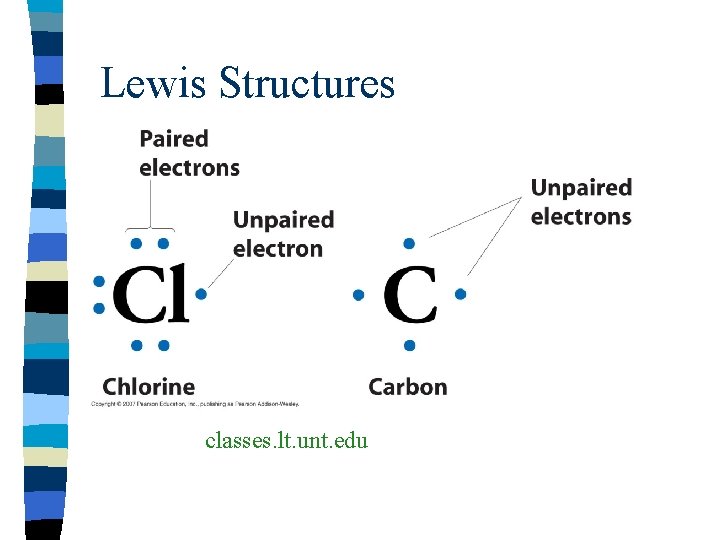
Lewis Structures classes. lt. unt. edu

Structural Formula n Show number, kind, arrangements, and bonds n Does NOT show unshared pairs of atoms n Single bond is one shared pair of electrons n Represented by one line

Example 6 n Draw the Lewis structure for iodomenthane.
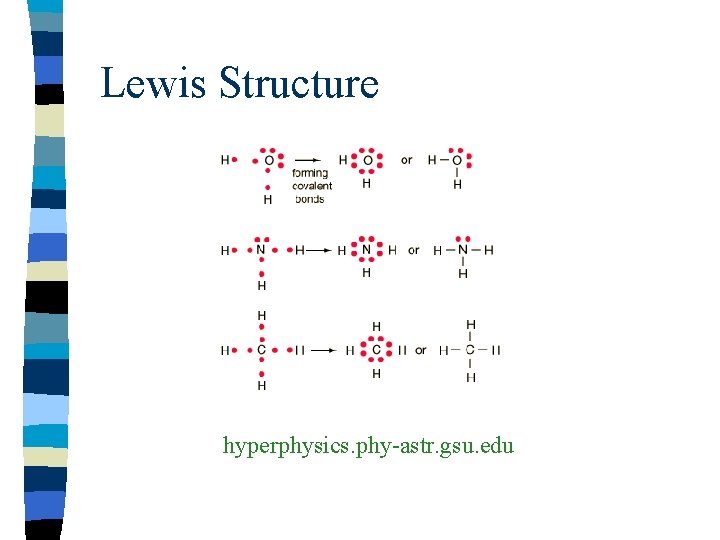
Lewis Structure hyperphysics. phy-astr. gsu. edu
Example 7 n Draw the Lewis structure for each of the following n H 2 O n CH 4 O n HCl

Multiple Covalent Bonds n n n n Some a toms can share more than one pair of electrons Carbon, Nitrogen, and Oxygen Double bond-two pairs of electrons shared Represented by two parallel lines Triple bond-three pairs of electrons shared Represented by three parallel lines These are called multiple bonds
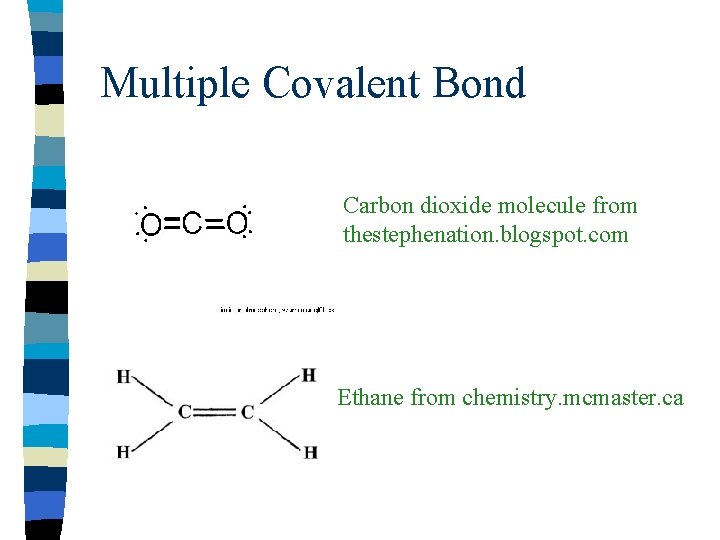
Multiple Covalent Bond Carbon dioxide molecule from thestephenation. blogspot. com Ethane from chemistry. mcmaster. ca

Example 8 n Draw the Lewis structure for methanol, CH 2 O, which is also known as formaldehyde.
Example 9 n Determine the Lewis structure for each of the following molecules n O 2 n C 2 H 4 n C 2 H 2

Resonance Structures Some molecules may take more than one structure n That is there is more than one way the molecule might be put together n Chemist think of compounds as bouncing between the possibilities aka resonating n Resonance-bonding that cannot be correctly represented by a single Lewis structure n

Covalent Network Bonding n These are not the individual units we have been dealing with n We will work with them more in chapter 7
- Slides: 28