Chapter 6 Section 1 4 Chemical Bonding Chemical
Chapter 6 Section 1 - 4 Chemical Bonding

Chemical Bond… • A mutual _____ attraction between the _____ and _________ of different atoms that binds the atoms together. • Most atoms are at a _____ energy state when bonded to other atoms rather than as independent particles

Types of chemical bonds: 1. 2. 3. 4. Covalent-network Ionic Metallic
• Ionic bonding • Results from the ______ attraction between large numbers of _______ (+ ions) and ______ (- ions) • Covalent bonding Results from the ______ of electron pairs between 2 atoms

Ionic Bonding • _______ differences in electronegativity values between atoms: – One atom attracts an e- so strongly that it actually PULLS IT AWAY from the other atom…electron is ________ • Δ electronegativity > ____

Nonpolar Covalent Bond • Bonding electrons are shared ______ by the bonded atoms, resulting in a balanced distribution of electrical charge • Δ electronegativity: ______ (roughly)

Polar-covalent bond • Bonding electrons are shared ______ because one atom pulls, although not very strongly, more on the electron pair than the other atom • Δ electronegativity: _______ • Still relatively small differences, so no atom really “wins” & e- is shared
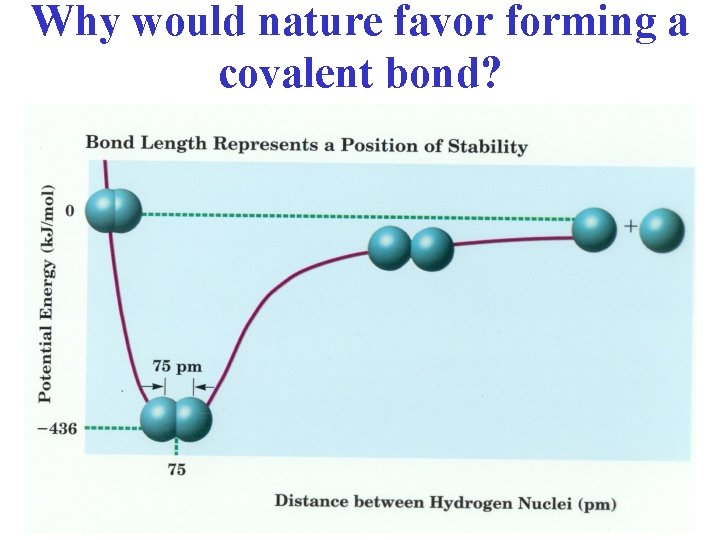
Why would nature favor forming a covalent bond?
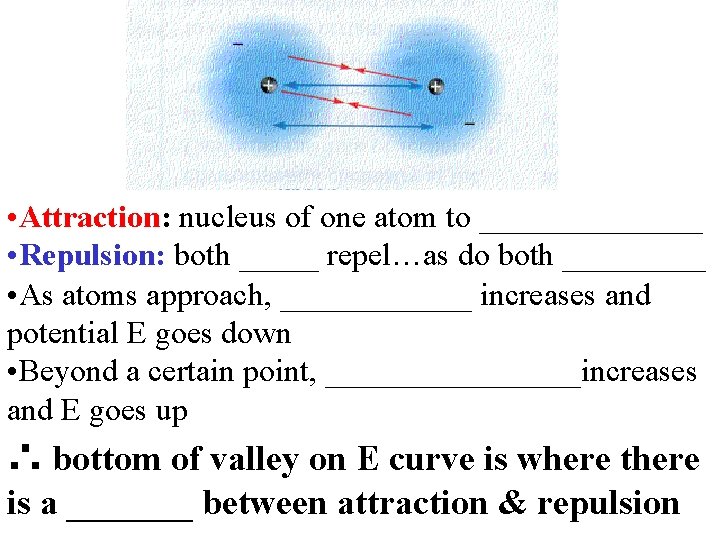
• Attraction: nucleus of one atom to _______ • Repulsion: both _____ repel…as do both _____ • As atoms approach, ______ increases and potential E goes down • Beyond a certain point, ________increases and E goes up ∴ bottom of valley on E curve is where there is a _______ between attraction & repulsion

• MOLECULE: neutral group of atoms held together by _________ bonds • MOLECULAR COMPOUND: a cmpd that is made of molecules • MOLECULAR FORMULA: shows the _______ & _______ of atoms making up a molecule i. e. H 2 O • DIATOMIC MOLECULE: molecule containing only ____ atoms i. e. H 2
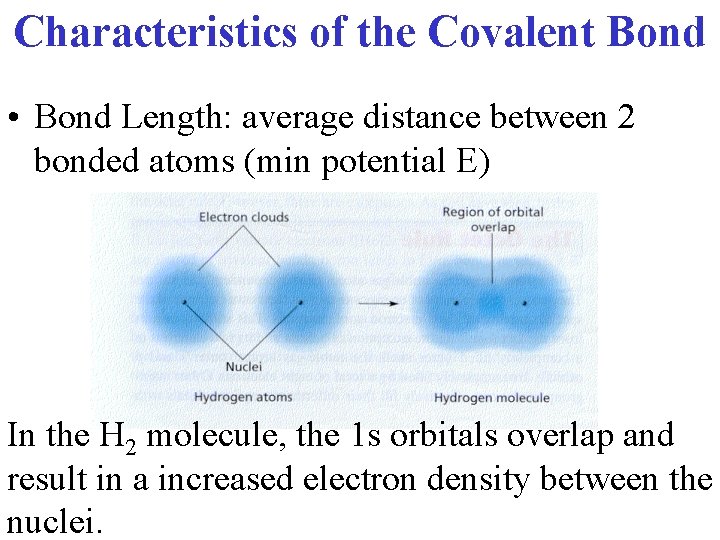
Characteristics of the Covalent Bond • Bond Length: average distance between 2 bonded atoms (min potential E) In the H 2 molecule, the 1 s orbitals overlap and result in a increased electron density between the nuclei.
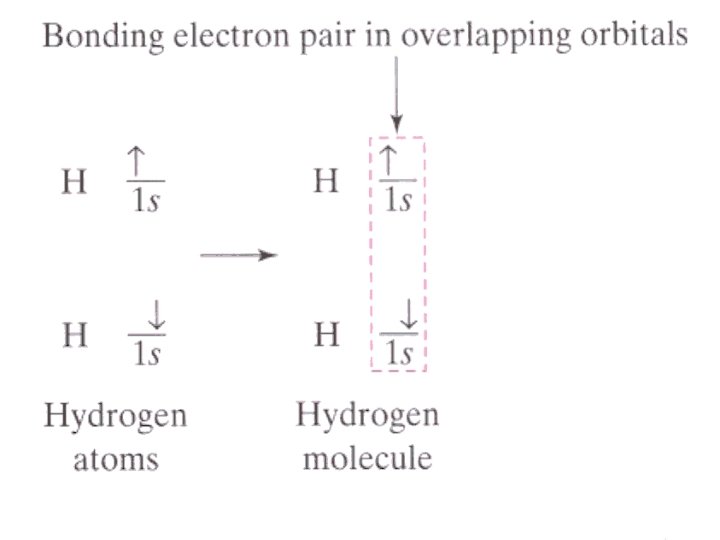

Bond energy: • Energy required to ______ a chemical bond and form neutral isolated atoms • Measured in _________ • ____ value • Bond energy and bond length values will vary depending on what atoms an element is bonded to…ave values found on tables P. 168)

Octet Rule • Chemical cmpds tend to form so that each atom, by gaining, losing or sharing electrons, has an ________ of electrons in its highest (outermost) energy level • Example of exceptions: – H: only has 2 valence e-s required to reach stability – B: only has 6 valence e-s “ “ – Some elements have > 8 (occurs when atom bonds with highly electroneg elements & some “d” e-s are involved in bonding

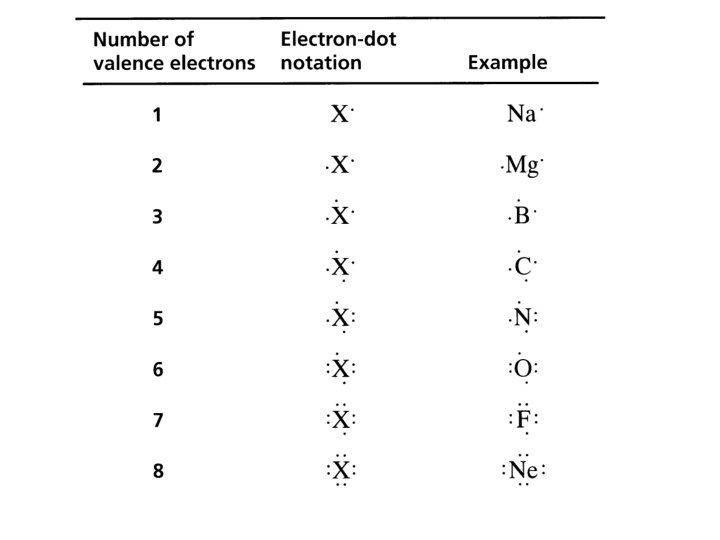
Electron Dot Diagrams • Electron configuration notation in which only the valence electrons of an atom of a particular element are shown, indicated by dots placed around the element’s symbol • F:
![“other” dot diagram format • Mg: [Ne]3 s 2 • “paired” electrons are shown “other” dot diagram format • Mg: [Ne]3 s 2 • “paired” electrons are shown](http://slidetodoc.com/presentation_image/95ada6f9310de4e51e226af2b149a911/image-17.jpg)
“other” dot diagram format • Mg: [Ne]3 s 2 • “paired” electrons are shown as a pair

Lewis Structures • Formulas in which…. – atomic symbols are represent __________________ – Dot-pairs or dashed between 2 atoms represent _______________ – Dots adjacent to only one atomic symbol represent _______________
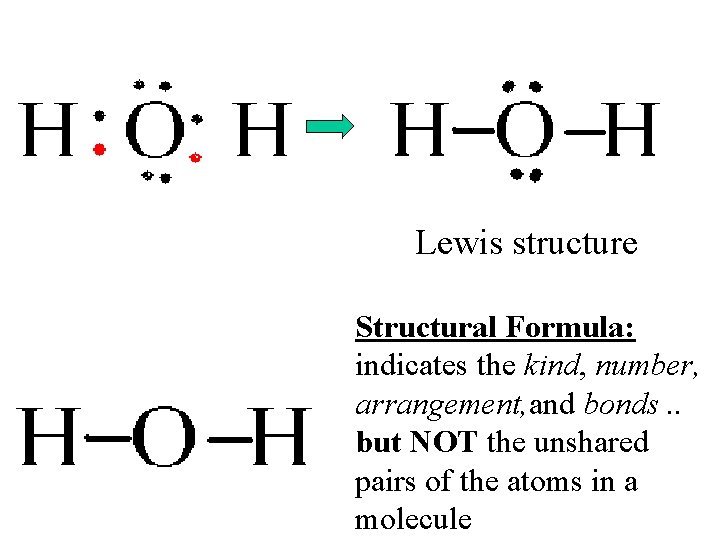
Lewis structure Structural Formula: indicates the kind, number, arrangement, and bonds. . but NOT the unshared pairs of the atoms in a molecule
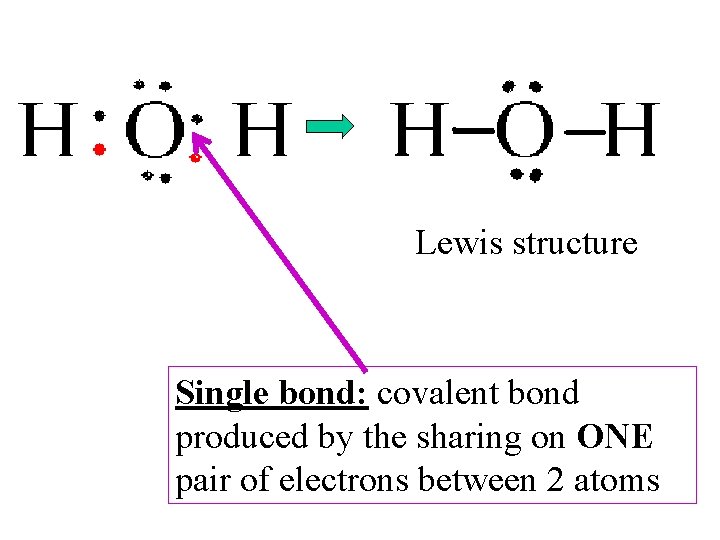
Lewis structure Single bond: covalent bond produced by the sharing on ONE pair of electrons between 2 atoms

Draw the Lewis structure for the following molecules: • • • IBr CH 3 Br F 2 O Si. Cl 4 C 2 HCl (? ? )

Multiple Covalent Bonds • DOUBLE BOND – sharing of ___ pairs of electrons between 2 atoms • TRIPLE BOND – sharing of _____ pairs of electrons between 2 atoms • Bond Energies • triple double single • …. . triple bonds are _____ and _______

Resonance Structures • bonding in molecules or ions that cannot be correctly represented by a single Lewis structure

Covalent-network Bonding Covalent bonding exists throughout a large network of atoms…will be discussed in chapter 12 i. e. carbon atoms in a diamond

Ionic Compound • composed of positive and negative ions that are combined so that the number of positive charges are equal to (cancel out) the number of negative charges • Na+1 Cl-1 • Mg+2 Cl 2 -1 (Mg+2 Cl-1) • Not called “molecules”…but called _______________

Formation of Ionic Cmpds • Atoms form ions when they _______ ein order to attain _____________ • Ions reach a ______ potential E through the electrical forces of attraction between oppositely charged particles when they combine in an orderly arrangement known as a _______ • Forces of repulsion (like-charged ions, adjacent e- clouds) _______ the forces of attraction (opp-charged ions, nuclei-electron of adj. ions)

Sodium chloride, Na. Cl • The chemical formula of an ionic cmpd is the simplest formula of the cmpd…it does NOT represent a discrete GROUP of separate atoms like a molecule does • Covalent bonding: simplest unit is a ________ • Ionic bonding: simples unit is a _____________
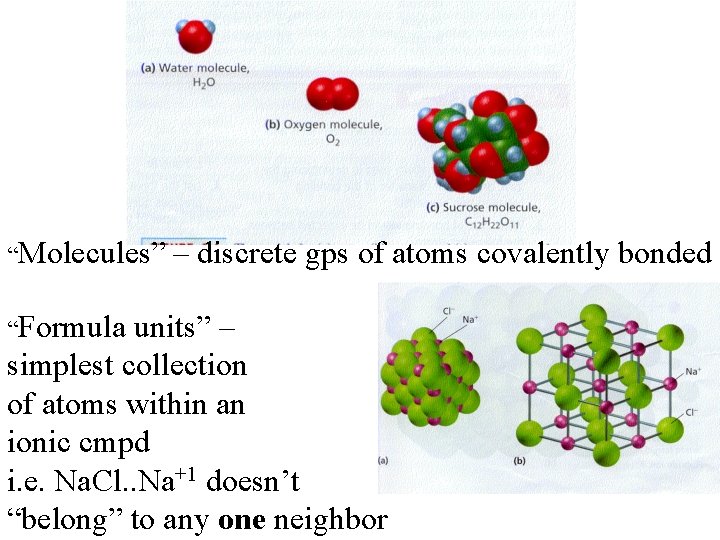
“Molecules” “Formula – discrete gps of atoms covalently bonded units” – simplest collection of atoms within an ionic cmpd i. e. Na. Cl. . Na+1 doesn’t “belong” to any one neighbor

Lattice Energy • Term used to describe ______ in ionic cmpds • Energy __________ when one of an ionic crystalline cmpd is formed from gaseous ions • _____ value indicates that E is ____ • The _____ the value, the more E released, the more stable the bonding will be (true also for bond E) • i. e. Na. Cl – 787. 5 k. J/mol

Which ionic cmpd has a higher melting point…. Mg. O or Ca. O ?

Ionic vs. Covalent Properties IONIC • strong force that holds _________ • stronger forces of attraction _______ ions MOLECULAR • strong covalent bond __________ • _______ F of attraction ______ molecules • ∴_____ melting • ∴melt at _____ temps & many vaporize & boiling pts; don’t at room temp vaporize at room temp
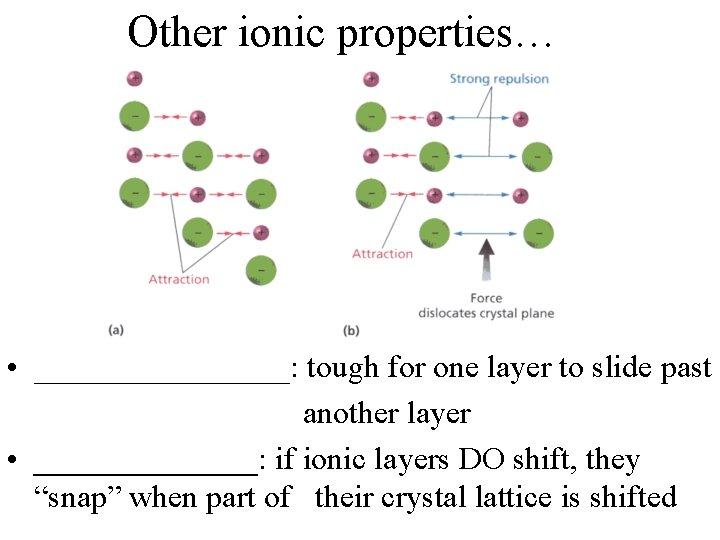
Other ionic properties… • ________: tough for one layer to slide past another layer • _______: if ionic layers DO shift, they “snap” when part of their crystal lattice is shifted

Ionic vs. Covalent Properties IONIC • non-conductors in solid state because _________ • conductors in _______ state • conductors when _________ MOLECULAR • non-conductors in ________

Polyatomic Ions • A _______ group of ______ bonded atoms • Combine with ions of opposite charge to form _________ cmpds

Draw the Lewis structure for the phosphate ion

Metallic Bond Model • Metals have ________ electrons in their highest E level • Metals frequently have many _________________ • Vacant orbitals of adjacent atoms overlap which allows _________________

Metallic Bond Model (con’t) • “_________ electrons” - e-s don’t stay in one locality like… – covalent bonding: stay in the ________________ – Ionic bonding: e-s are bound to ________________ • mobile electrons form a “____ of electrons”

Metallic Bonding • the chemical bonding that results from the attraction between __________________ • _______ sharing of many e-s where each atom contributes its valence e-s which are then free to move about the mostly vacant outer orbitals of all the metal atoms

Metallic Properties • _____ electrical & thermal conductivity – due to high mobility and delocalization of e-s • _______ (shine) – metals absorb E and become “excited” very easily because many of their orbitals are separated by extremely small ∆E…shine occurs when photons are emitted when excited e-s return to ground state

Metallic Prop. (con’t) • ____________ (ability to be hammered/beaten into thin sheets) and __________ (ability to be drawn, pulled, or extruded to produce wire) because metallic bonding is the same in all directions and a shift in layers of atoms is inconsequential

Metallic Bond Strength • Expressed in the _________ value where the bonded atoms in the metallic solid state are converted into __________ metal atoms in the gaseous state (usually↑heat of vap, the ____ the bond strength) • Determined by – __________________
- Slides: 41