Chapter 6 Principles of Stereochemistry Bring Your Model

Chapter 6 Principles of Stereochemistry ***Bring Your Model Kits to Class!***
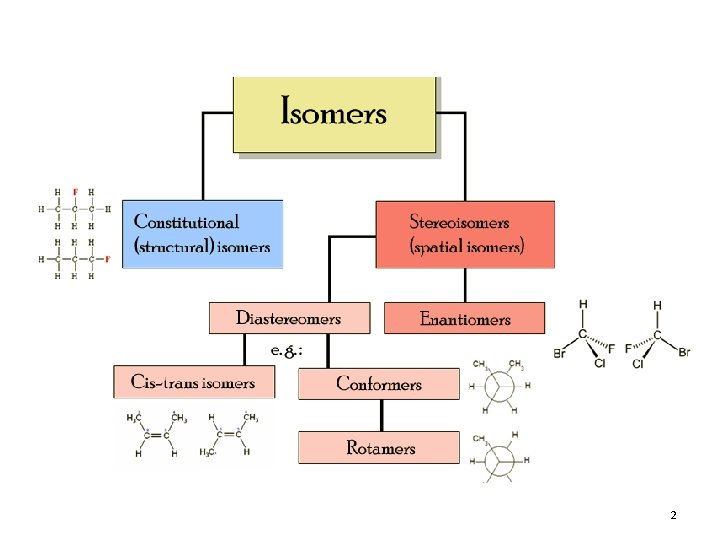
2
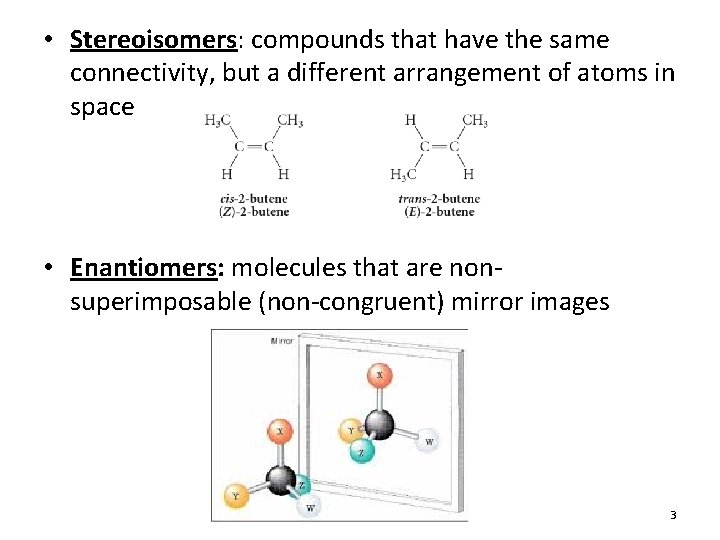
• Stereoisomers: compounds that have the same connectivity, but a different arrangement of atoms in space • Enantiomers: molecules that are nonsuperimposable (non-congruent) mirror images 3
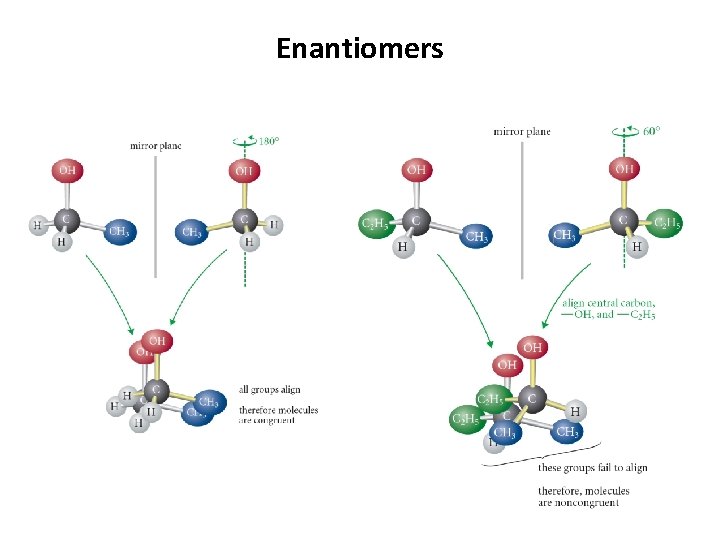
Enantiomers
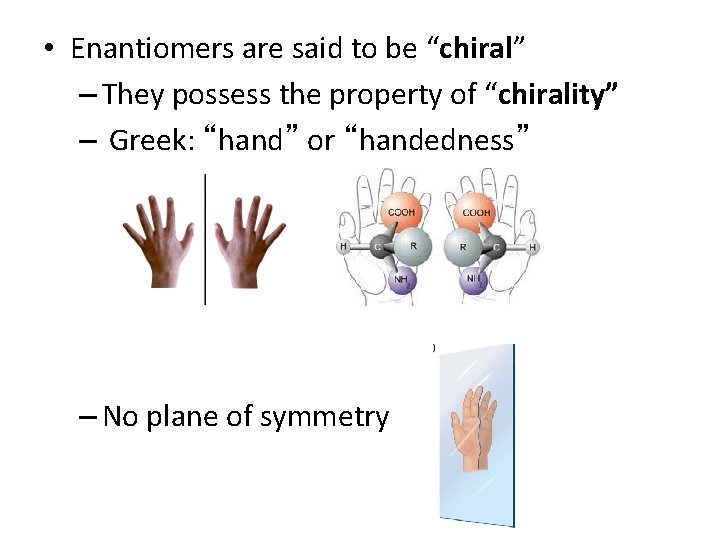
• Enantiomers are said to be “chiral” – They possess the property of “chirality” – Greek: “hand” or “handedness” – No plane of symmetry
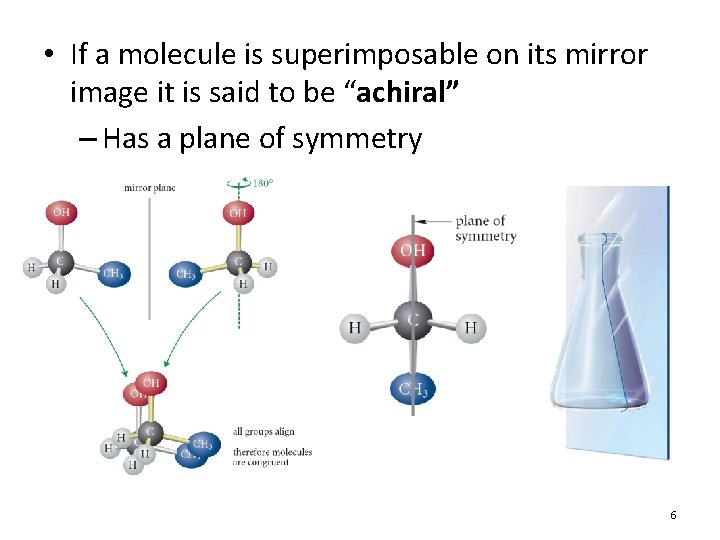
• If a molecule is superimposable on its mirror image it is said to be “achiral” – Has a plane of symmetry 6

Chiral Carbons • Many chiral molecules contain one or more asymmetric carbons – Called “Chiral Carbons” – Attached to four different groups – Denoted by an asterisk – Source of stereoisomerism • Interchange of groups = other stereoisomer • Stereocenters 7
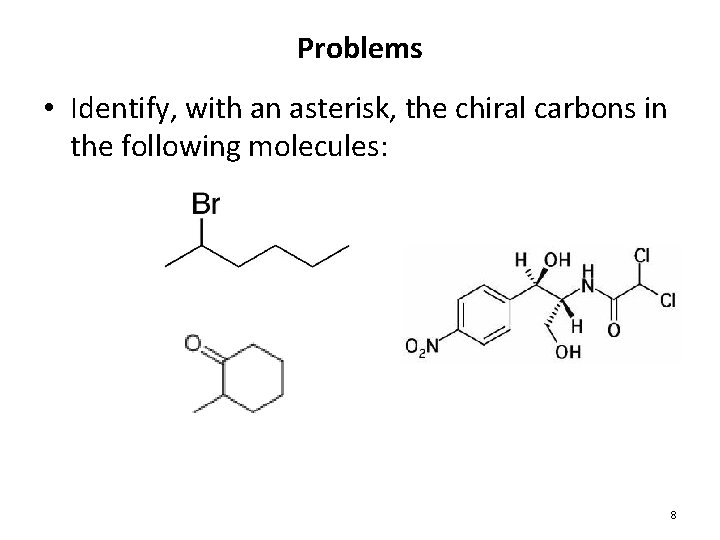
Problems • Identify, with an asterisk, the chiral carbons in the following molecules: 8
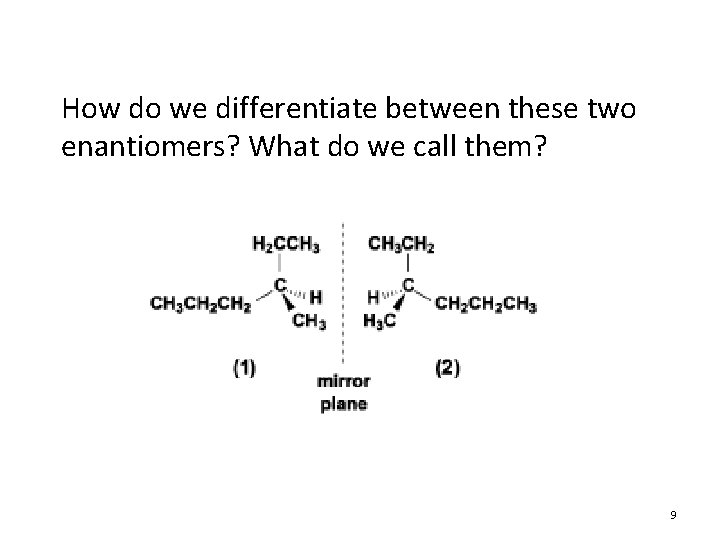
How do we differentiate between these two enantiomers? What do we call them? 9

Nomenclature of Enantiomers 1) Identify your chiral carbon 2) Prioritize your substituents on the chiral carbon according to the Cahn-Ingold-Prelog priority rules used to determine E and Z for alkenes – Highest priority = 1, Lowest Priority = 4 10

Priority Assignment Rules a) Identify the atoms directly attached to the C and rank them according to atomic #. – Higher atomic # = higher priority – Higher isotopic mass = higher priority 11
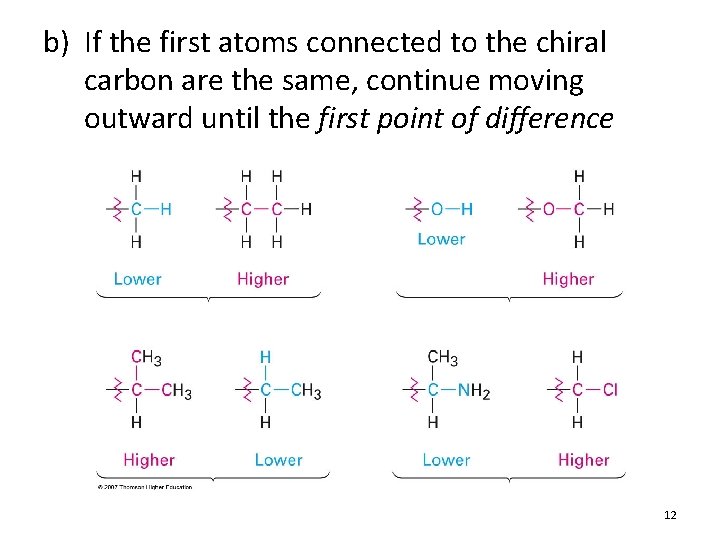
b) If the first atoms connected to the chiral carbon are the same, continue moving outward until the first point of difference 12
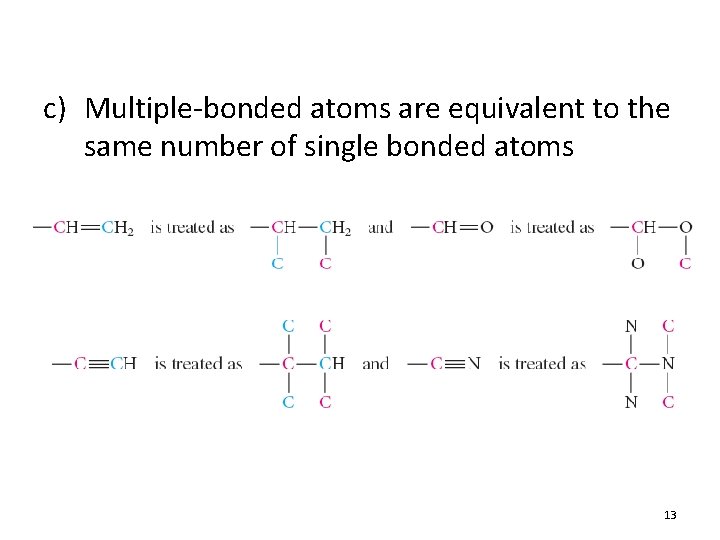
c) Multiple-bonded atoms are equivalent to the same number of single bonded atoms 13

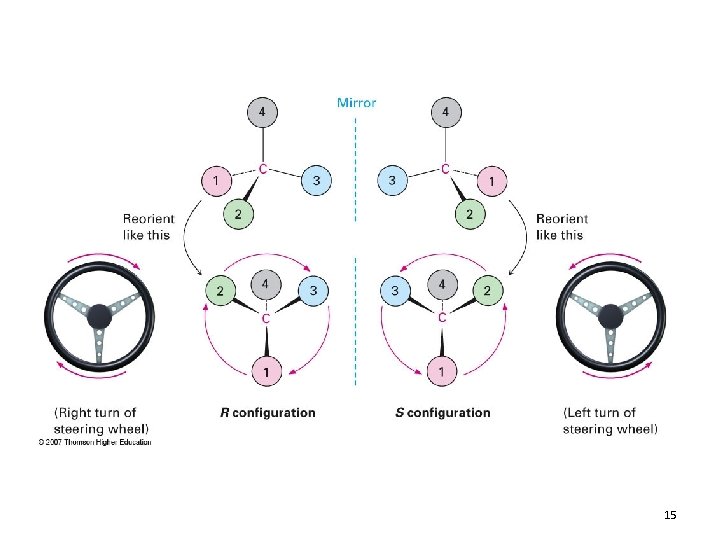
3) Point the lowest priority substituent away from you and look at the 3 remaining groups in a plane 4) Count 1, 2, 3 • If you go CW = R • (rectus, Latin “proper”) • If you go CCW = S • (sinister, Latin “left”) 14
15
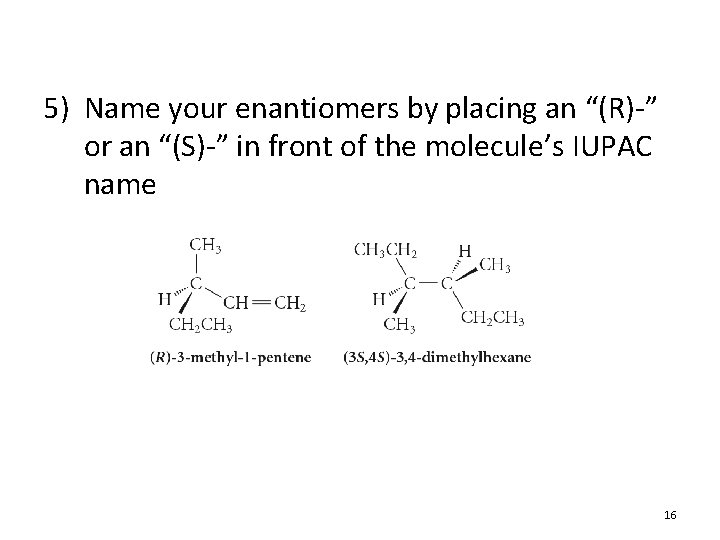
5) Name your enantiomers by placing an “(R)-” or an “(S)-” in front of the molecule’s IUPAC name 16
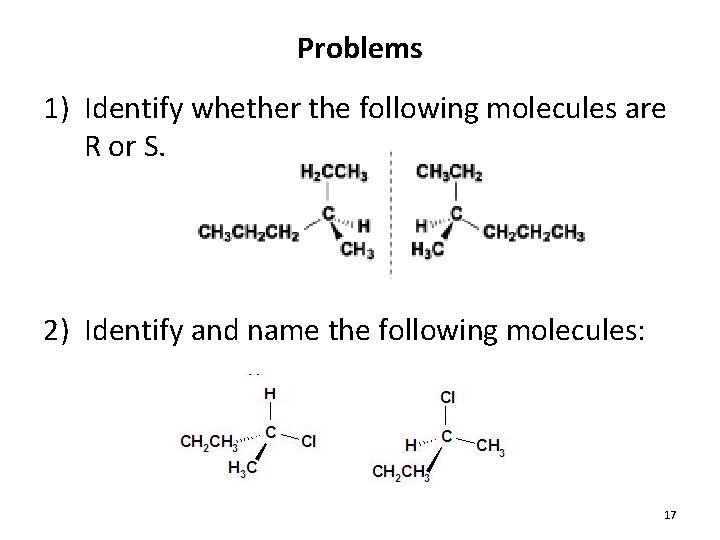
Problems 1) Identify whether the following molecules are R or S. 2) Identify and name the following molecules: 17
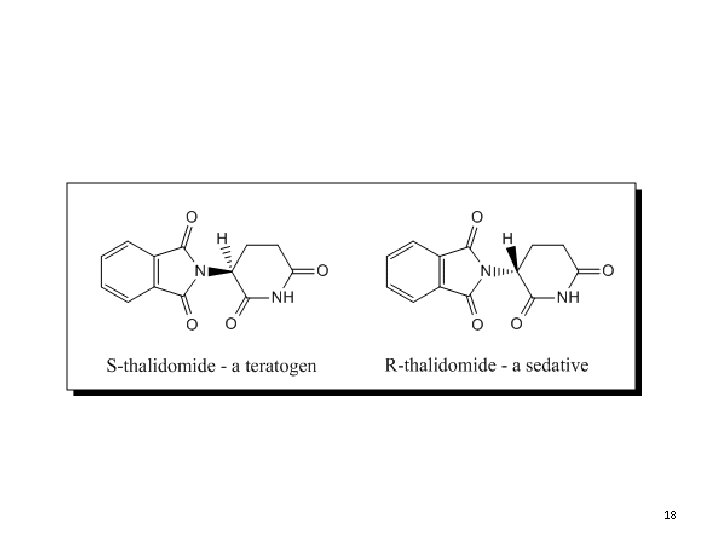
18
- Slides: 18