CHAPTER 6 METABOLISM Metabolism refers to all of

CHAPTER 6 METABOLISM

• Metabolism refers to all of the chemical reactions performed by an organism • Thousands of chemical reactions occur in a cell • These reactions are arranged into pathways that intersect • Metabolic pathway- begins with a specific molecule which is altered in a series of steps, resulting in a certain product

• Each step of a metabolic pathway is catalyzed by a specific enzyme • Mechanisms are in place to regulate enzymes based on the needs of a cell

• Catabolic pathways- degradative processes that break down complex molecules into simpler compounds • These release energy • Ex. Cellular respiration- glucose is broken down in the presence of oxygen, to form CO 2 and water • Pathways can have more than one starting molecule or product • Energy that was stored in the organic molecule is now available to do work for the cell

• Anabolic pathways – consume energy to build complex molecules from simpler ones • Sometimes called biosynthetic pathways • Ex. Protein synthesis from amino acids

• Catabolism- (downhill) • Anabolism (uphill) • Energy released from “downhill” catabolism can be stored and used to drive “uphill” anabolic pathways • Bioenergetics- the study of how organisms manage their energy resources

• Energy- the capacity to cause change • Kinetic energy- energy of motion • Heat (thermal) energy- kinetic energy associated with the random mov’t of atoms or molecules • Potential energy- energy of location or structure (stored) • Chemical energy- biologically it is the potential energy available for release in a chemical reaction

• Organisms are energy transformers!! • Some energy will always be lost during an energy transformation

• Thermodynamics – the study of energy transformations that occur in a collection of matter • Open system- energy (and sometimes matter) can be transferred between the system and its surroundings • Closed system- isolated from its surroundings

• Organisms are open systems – • They absorb energy (ex. Light/chemical) • Release energy (ex. Heat and metabolic waste products like carbon dioxide)

1 st law of thermodynamics • Energy of the universe is constant • Energy can be transferred and transformed but it can not be created or destroyed • This is known as the principle of conservation of energy • Ex. Green plants do not create energy, they convert it from light to chemical

2 nd law of thermodynamics • Only a small amount of the chemical energy from food is transferred into actual useful energy for an organism • In most energy transformations some energy is converted to heat and is lost to the surroundings • What is lost is an unstable form of energy , and this makes the universe more disordered.

2 nd law continued… • Entropy- measure of disorder or randomness • 2 nd law states- every energy transfer or transformation increases the entropy of the universe • For a process to occur spontaneously, it must increase the entropy of the universe

Order vs disorder • Living systems increase the entropy of their surroundings • They can also create order within themselves • Energy flows into an ecosystem as light and leaves as heat • The entropy of a single organism can decrease as long as the entropy of the universe is still increasing

• Exergonic reaction – proceeds with a net release of free energy • Can occur spontaneously

• Endergonic reaction- absorbs free energy from its surroundings • Are not spontaneous

• Chemical reactions are exergonic in one direction and endergonic in the opposite direction • All reactions are reversible

Equilibrium • Reactions in a closed system eventually reach equilibrium • Once in equilibrium, no work can be done • Metabolism on a whole can never be at equilibrium or else a cell would be dead!

• Like most systems, our cells are not in equilibrium • Constant flow of energy and materials in and out keep metabolic pathways of cells from reaching equilibrium • Organisms are open systems

Cells do 3 main kinds of work 1) Mechanical work- ex. mov’t of cilia, mov’t of chromosomes, muscle contractions 2) Transport work – ex. Pumping substances across the membrane agains the gradient (active transport) 3) Chemical Work – ex. Pushing of endergonic reactions like synthesis of polymers

Energy coupling • The use of an exergonic process to drive an endergonic one • ATP mediates most energy couplings • ATP is usually the immediate source of energy to power cellular work

ATP • Adensine triphosphate – • Consists of: • Ribose sugar • Adenine (nitrogenous base) • Chain of three phosphate groups

ATP continued • Bonds between the phosphate groups can be broken by hydrolysis • When the terminal phosphate bond is broken, an inorganic phosphate molecule leaves the ATP • The ATP then becomes ADP (adenosine diphosphate) • This is exergonic and releases 7. 3 kcal of energy per mole of ATP hydrolyzed

• Phosphate bonds are called “high energy” because they release energy when hydrolyzed • But- they are not unusually difficult to break

• When ATP is hydrolyzed the energy released heats the surroundings • Ex. - when someone shivers the muscle contraction hydrolyzes ATP and warms the body In most cases in the cell however, generating heat alone would be inefficient and dangerous

• Instead, in most cases specific enzymes help the cell to couple the energy of ATP hydrolysis directly to endergonic processes • This is done by transferring a phosphate group from ATP to some other molecule (like a reactant) • The molecule that receives the phosphate is said to be phosphorylated

• The newly phosphorylated molecule is less stable (more reactive) than the original form of the molecule • The phosphorylated molecule then undergoes a change that performs work

Regeneration of ATP • ATP is needed continuously by a cell doing work • Luckily ATP is a renewable resource that can be regenerated by adding a phosphate to ADP • The free energy to phosphorylate ADP comes from exergonic breakdown reactions (catabolism) in the cell
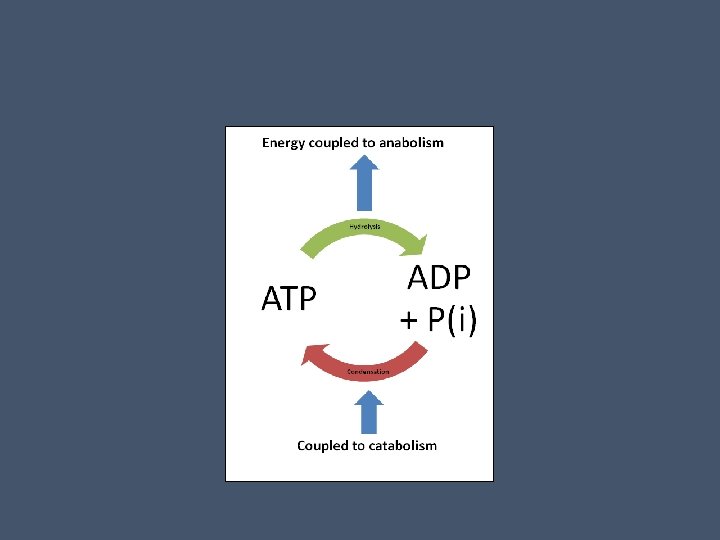

• This is the ATP cycle and it couples exergonic and endergonic reactions within the cell • A muscle cell can recycle 10 million molecules of ATP per second! • Regeneration of ATP from ADP and P is endergonic!

• Because this is endergonic, it is not spontaneous • Catabolic (exergonic) pathways like cellular respiration and photosynthesis (in plants) provide the energy to make ATP • The chemical potential energy stored in ATP drives most cellular work

• Spontaneous reactions do not necessarily occur quickly • Catalysts are needed to speed up most chemical reactions • Catalyst – a chemical agent that speeds up a reaction without being consumed by the reaction • Enzyme – protein catalyst

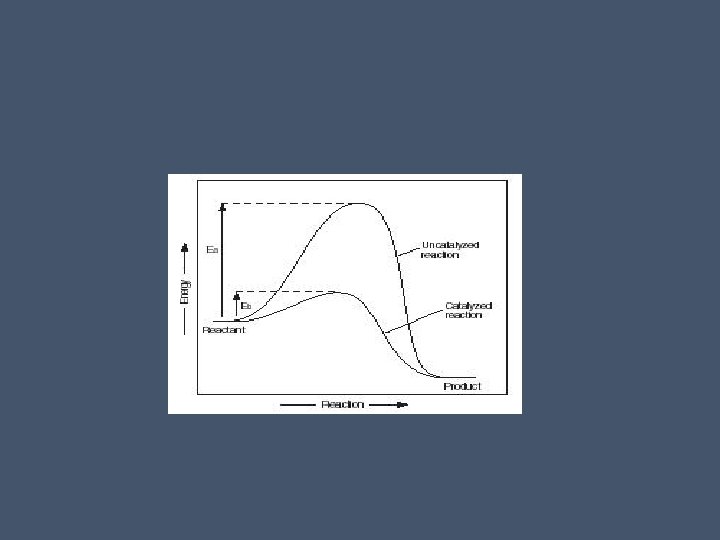
Activation Energy • Every chemical reaction involves breaking bonds and forming new ones • Activation energy – the initial investment of energy for starting a reaction (EA) • As reactants are being “pushed” uphill the free energy increases and the molecules become more unstable • as activation energy is reached and bonds begin to break, the reactants are said to be in a transition state which is very unstable

• The “downhill” portion of the reaction is the forming of new bonds (loss of free energy) • Activation energy can come in the form of heat that reactants absorb • The bonds in reactants break only when they have absorbed enough energy to become unstable and are more reactive

Why? • Thermal energy increases the movement of reactant molecules so they collide more often and more forcefully, breaking them apart • Thermal energy also makes molecular bonds more likely to break

Speeding up reactions • Heat will speed up reactions but this is not appropriate for biological systems because heat will denature molecules • Also, heat would speed up all reactions in a cell, which is not good! • Enzyme catalysts are the perfect solution • They are specific and work by lowering the activation energy • Enzymes do NOT change the free energy changes of a reaction • They only speed up the rate

Substrate Specificity • Substrate- reactant an enzyme acts on • Enzymes binds to substrate(s) forming an enzyme-substrate complex • While they are joined, the enzyme converts the reactants into the products
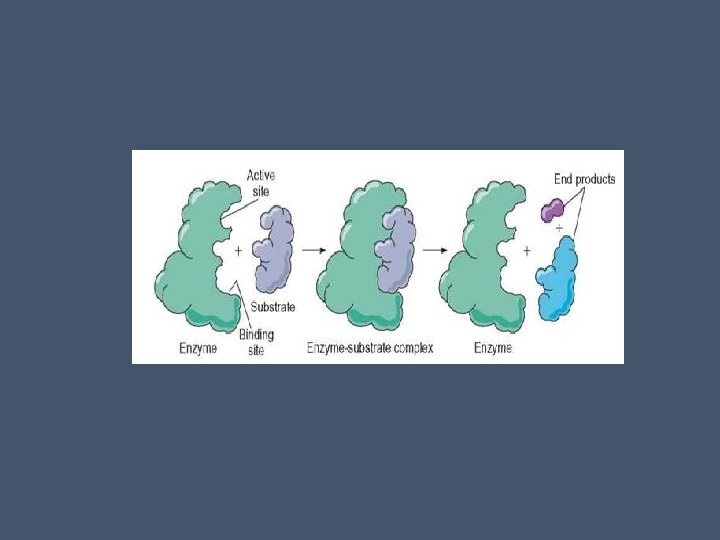

• Enzymes are extremely specific, catalyzing only one type of reaction each • This is due to the enzyme’s (protein’s) 3 -D shape which is a result of its AA sequence • Only one restricted area binds to a substrate- this is the active site • Active site is usually a groove on the surface of the enzyme

• Shape of the active site matches the shape of the substrate • The active site is not rigid- as the substrate enters, interactions between chemical groups cause the active site to fit even more closely to the shape of the substrate • This is called the induced fit (like a clasping handshake) • Induced fit brings the chemical groups of the active site into position to enhance their ability to catalyze the reaction
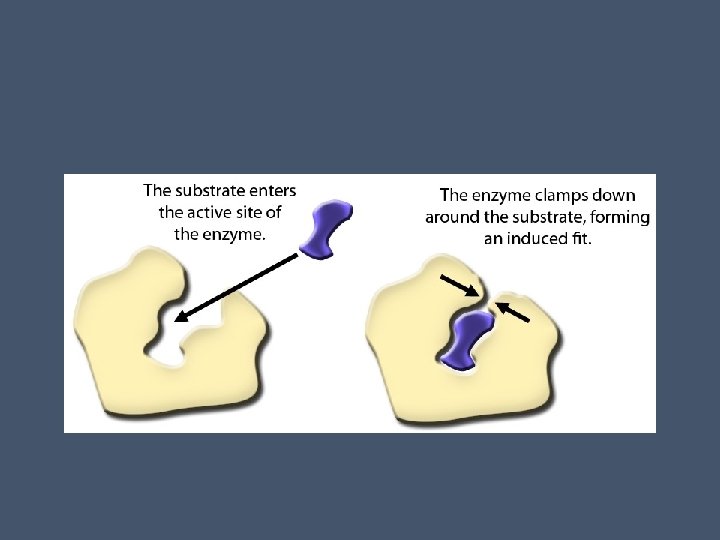

• Substrate is usually held in the active site by weak interactions (hydrogen or ionic bonds) • Side chains (R groups) of a few amino acids that make up the active site catalyze the conversion of substrate to product • Once formed, the product leaves the active site

• The Enzyme is now free to do the same with another molecule of the same reactant • Certain enzymes are able to catalyze 1000 molecules of a substrate in one second • Because enzymes are reusable and work so quickly, very small amounts of enzyme can have a very large metabolic impact

• Most metabolic reactions are reversible • An enzyme can catalyze both the forward and reverse reactions • Which reaction will occur more often depends on the relative concentrations of the reactants and products • An enzyme always catalyzes a reaction in the direction of equilibrium

• The rate that an enzyme converts a particular reactant to product depends on the initial concentration of the reactant • But, it gets to a point where all of the enzymes are “engaged” so the rate can not increase any longer • This is when an enzyme is said to be saturated • When this occurs, the only way to speed up the reaction further is to add more enzyme • Cells can do this naturally by making more enzyme molecules

What effects enzymes… Enzyme activity can be affected by environmental conditions like temp and p. H Can also be affected by chemicals specifically targeting a particular enzyme

Temp and p. H • Optimal conditions for any enzyme will allow the active site to be in the proper 3 -D conformation • UP TO A POINT, enzyme action increases with an increase in temp • After this threshold is met, the enzyme will begin to denature and the rate will drastically decrease • Each enzyme has an optimal temp

• Most human body enzymes have optimal temps between 35 -40 C • The same goes for p. H and optimal p. H for most human body enzymes is 6 -8 • Exception- pepsin (p. H 2)

Cofactors • Nonproteins that help enzyme activity • May be bound to the enzyme permanently or may bind and release like a substrate • Coenzymes are cofactors that are organic • Ex. Most vitamins
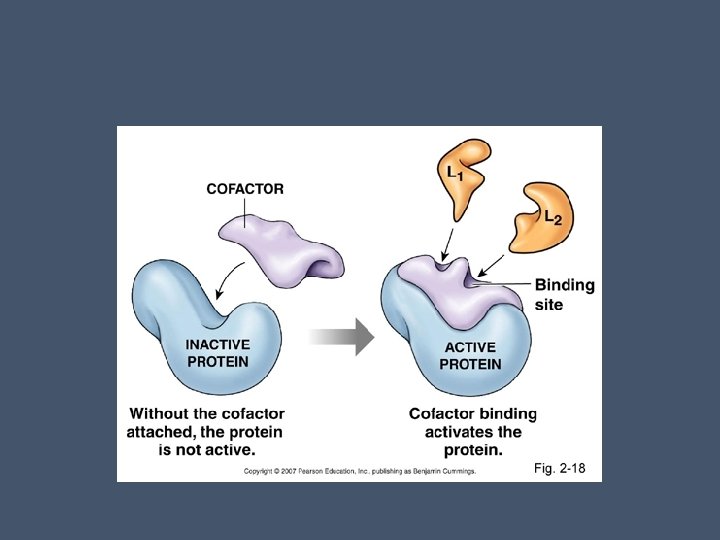

Enzyme Inhibitors • Certain chemicals selectively inhibit the action of specific enzymes • Inhibitors can be classified as either reversible or irreversible depending on how they bond to the enzyme

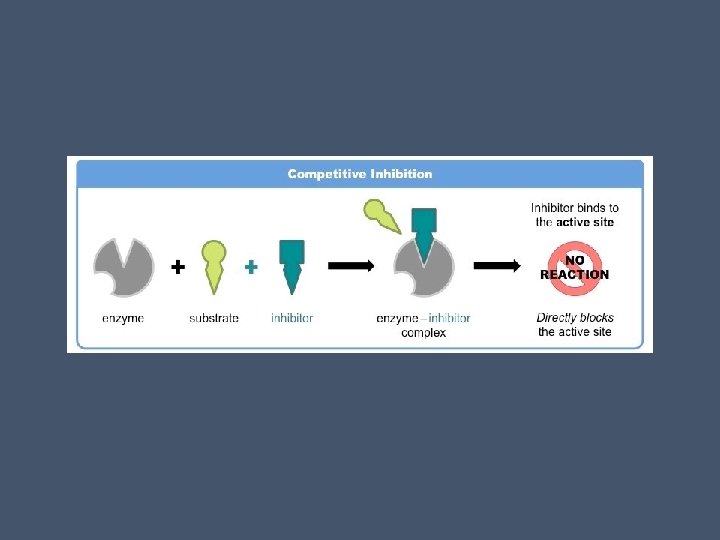
Competitive inhibitors • Occurs when inhibitors resemble the normal substrate molecule in structure and compete for the active site • These reduce the productivity of enzymes by blocking substrates from entering the active sites • This can be overcome by increasing the concentration of substrate

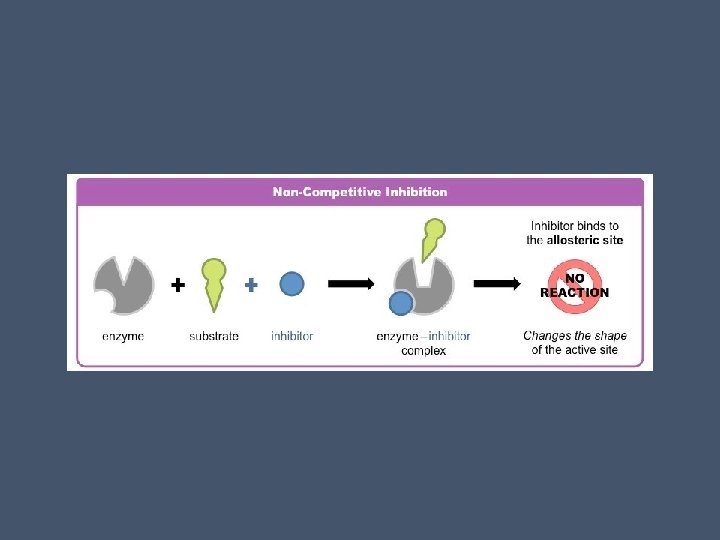
Noncompetitive Inhibitors • Do not directly compete with the substrate • They impede enzymatic reactions by binding to another part of the enzyme • This causes the enzyme to change shape • This makes the active site less effective • Increasing substrate concentration will have no effect here

• Toxins and poisons are often irreversible enzyme inhibitors • Antibiotics can be inhibitors of specific enzymes in bacteria • Penicillin – blocks the active site of an enzymes that many bacteria need to make their cell walls

• Some molecules in the cell naturally regulate enzyme activity by acting as inhibitors • This selective inhibition helps control cellular metabolism

Regulating enzyme activity • Cells need to be able to regulate which enzymes work when and where in a cell • Cells do this by : • Switching on or off the genes that code for specific enzymes • Regulating activity of enzymes once they are made

Feedback Inhibition • Common method of metabolic control • When a metabolic pathway is switched off by the inhibitory binding of the end product to an enzyme that acts early in the pathway • This prevents the cell from wasting chemical resources by synthesizing more than is necessary
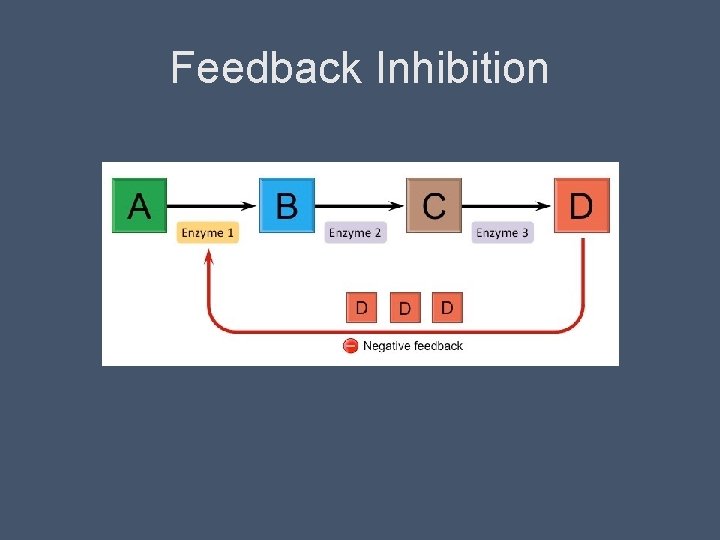
Feedback Inhibition

Enzyme Localization • Structures within the cell can help bring order to metabolism • Some enzymes used for several steps in a pathway will be found together in a multi-enzyme complex • This serves to control and speed up a sequence of reactions • The product of the first enzyme becomes the substrate of the next enzyme in line • This continues until the product is released

• These complexes can be in fixed places in the cell and act as structural components of some membranes • Some are enclosed within a specific organelle- ex. Cell resp in mitochondria
- Slides: 63