Chapter 6 Matter Mixtures Pure Substances Elements Compounds

Chapter 6
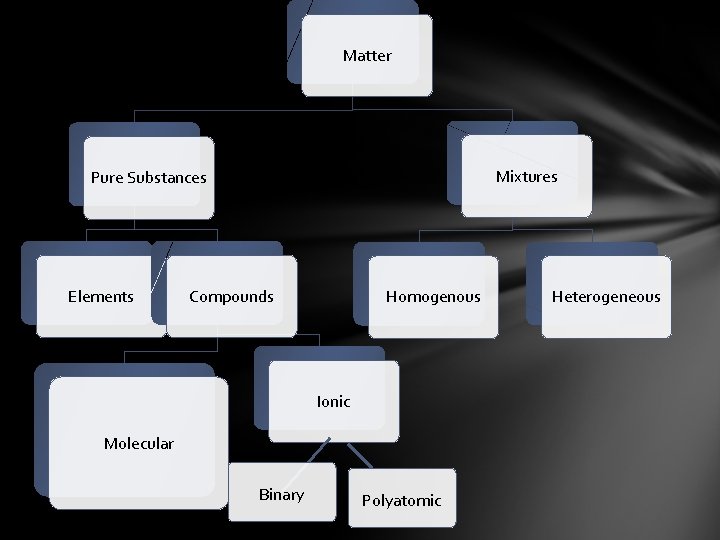
Matter Mixtures Pure Substances Elements Compounds Homogenous Ionic Molecular Binary Polyatomic Heterogeneous

Bond Types Two types bonding: Ionic – between a metal and a nonmetal - polyatomic- group of nonmetals that carry a charge Covalent – between two or more nonmetals

Properties of Ionic Compounds Most ionic compounds are crystals at room temperature Ions are arranged by repeating 3 dimensional patterns High melting points due to large attractive force between ionselectrostatic attraction

Properties continued s. Can conduct electric current when melted or dissolved in waters. Why?

Electrolytes- substances that conduct electricity when dissolved how gatorade works http: //www. gatorade. com/frequently_asked_questions/default. aspx

Franklinite-Zinc Iron Manganese Oxide

Fluorite-Ca. F 2

Aragonite-Ca. CO 3
Fluorescent


Molecules and Molecular compounds Covalent bond- atoms held together by the sharing of electrons What is the difference between an ionic compound a molecular compound?

http: //www. youtube. com/watch? v=Qqjc. Cvz. Wwww&feature=relatedhttp: //www. y outube. com/watch? v=Qqjc. Cvz. Wwww&feature=related
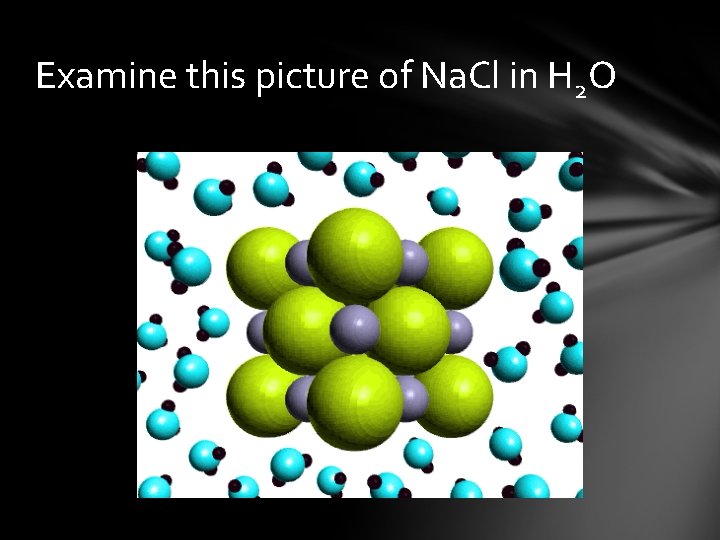
Examine this picture of Na. Cl in H 2 O

Ionic compounds are made of + and – charged ions arranged in repeating 3 -D patterns ex: Na. Cl Electrons transferred Molecular compound is made of elements chemically combined sharing electrons to complete octet ex: H 2 O

Molecular compounds have lower melting and boiling points than ionic compounds


Naming Ionic Compounds Metals go first, nonmetals go second Metal keeps its name Names on anions typically end in -ide



Draw the Lewis Structure for the formula of Na. Cl

What about Magnesium chloride?

Criss Cross Method

Transition metals In forming ions, transition metals lose the valence shell s electrons first, then as many d electrons as are required to reach the charge of the ion

Naming transition metals Stock name and classical name • Stock – preferred naming system, uses Roman numerals in parentheses to placed after the name of the element to indicated the numerical value of the charge ex: Fe 2+ would be iron(II) • Classical- older method uses root word with different suffix -ic and –ous

Polyatomic ions Two or more atoms that are bound together by covalent bonds, but have gained or lost an electron in order to become more stable Act as a whole when forming an ionic compound

Ex: Iron exists as two different ions Fe 2+ and Fe 3+ Fe 2+ is known as iron II (ferrous) Fe 3+ is known as iron III (ferric)


Common Molecules www. reciprocalnet. org/edumodules/commonmolecules/element/list. html#simple

Molecules and Molecular compounds Covalent bond- atoms held together by the sharing of electrons What is the difference between an ionic compound a molecular compound?

http: //www. youtube. com/watch? v=Qqjc. Cvz. Wwww&feature=relatedhttp: //www. y outube. com/watch? v=Qqjc. Cvz. Wwww&feature=related
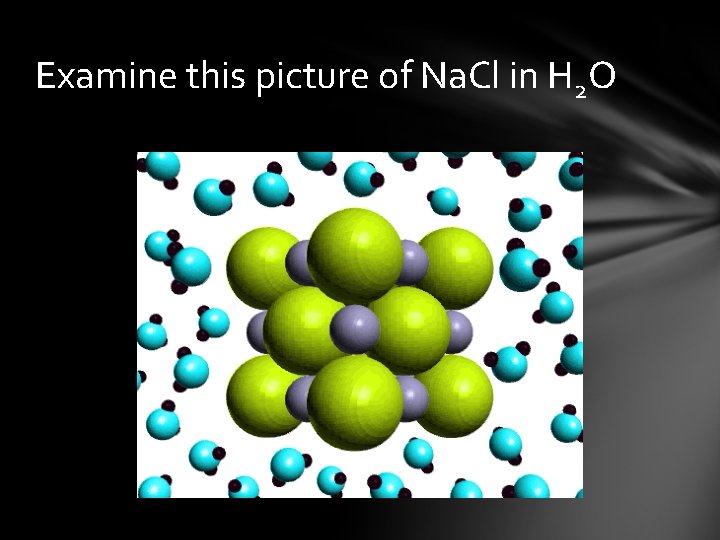
Examine this picture of Na. Cl in H 2 O

Ionic compounds are made of + and – charged ions arranged in repeating 3 -D patterns ex: Na. Cl Molecular compound is made of elements chemically combined sharing electrons to complete octet ex: H 2 O

Molecular compounds have lower melting and boiling points than ionic compounds

Molecular formulas Molecular formula-chemical formula of a molecular compound that shows how many atoms of each element are in a molecule

Ex: H 2 O • Subscript (2) after the H indicates there are 2 hydrogen atoms and one oxygen atom How many carbon and oxygen atoms are in a molecule of CO 2?

Naming Molecular Compounds 1 – mono (only used on the second element) 2 - di 3 - tri 4 - tetra 5 - pent 6 - hexa 7 - hepta 8 – octo 9 – nona 10 – deca Use prefixes indicating the number of atoms of each element

Examples: CO Carbon monoxide CO 2 Carbon dioxide
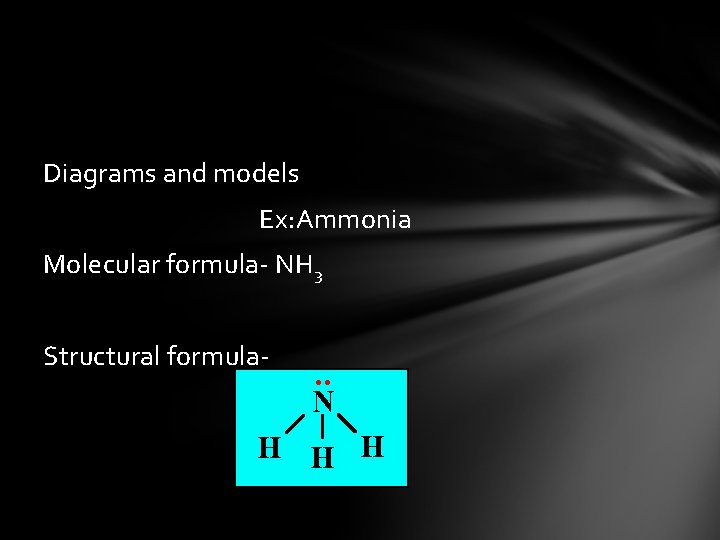
Diagrams and models Ex: Ammonia Molecular formula- NH 3 Structural formula-
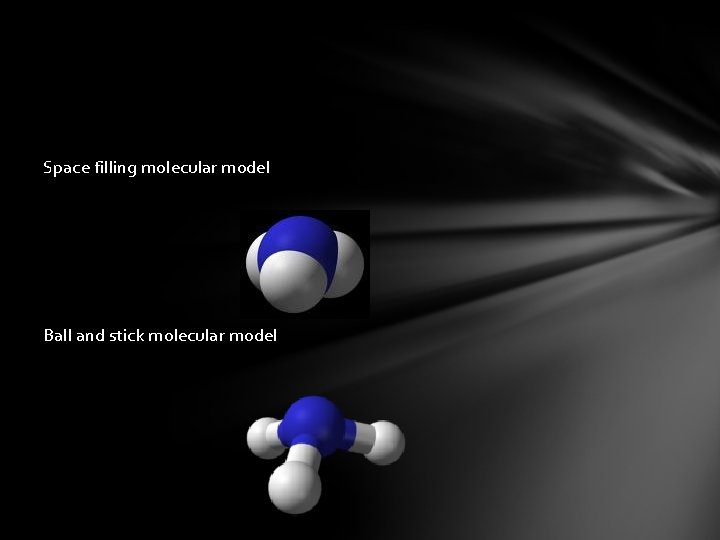
Space filling molecular model Ball and stick molecular model

Bonds Single Bondsbetween two atoms, share 1 pair of electrons Double bondsbetween two atoms, share 2 pair of electrons Triple Bonds- between two atoms, share 3 pair of electrons

Metallic Bonds Metallic bonding theory- metal cations are held together by a sea of free floating electrons

Physical properties of metals Explained by the sea of electrons • Good conductors – because electrons flow freely through sea of electrons • Ductile and malleable- sea of electrons insulates the metal cations from each otherunder pressure metal cations slide past each other

Alloys Mixtures of two or more elements, at least one being a metal Enhances properties of metals

Ex: Sterling Silver • 92. 5% silver • 7. 5% copper Better durability but soft enough to make jewelry

Atomium- Brussels Belgium Designed to resemble a crystal of iron magnified 165 billion times –made of aluminum alloy panels and connected by steel tubes. Top sphere is 92 meters above ground


Review Questions True or False Molecular compounds tend to have higher melting and boiling points than ionic compounds

Review Questions True or False: 1. Most molecular compounds are composed of two or more nonmetallic elements.

TRUE

Review Questions True or False Atoms in molecular compounds share electrons.

TRUE

FALSE

- Slides: 54