Chapter 6 Lipids and Membranes 1 Lipid classification

Chapter 6 Lipids and Membranes 1 -Lipid classification 2 -Properties of lipid aggregates 3 -Biological membranes 4 -Membrane assembly and protein targeting 5 -Lipoproteins Lippincott’s Illustrated Reviews: Biochemistry. (2005). P. C. Champe. , R. A. Harvey. , D. R. Ferrier. 3 rd edition. Lippincott Williams & Wilkins.

Biological membranes § Are organized assemblies of lipids and proteins with small amounts of carbohydrate. § They are permeable barriers to the passage of materials § They regulate the composition of the intracellular medium by controlling the flow of nutrients, waste products, ions, etc. , into and out of the cell. § They do this through membrane-embedded “pumps” and “gates” that transport specific substances against an electrochemical gradient or permit their passage with such a gradient.

Lipid § Lipids: (Greek: lipos, fat) § Are substances of biological origin § Soluble in organic solvents such as chloroform and methanol § Sparingly soluble, if at all, in water. § Fats, oils, Certain vitamins and hormones, and most nonprotein membrane components are lipids.

A. Fatty Acids § Are carboxylic acids with long-chain hydrocarbon side groups § They are rarely free in nature § Occur in esterified form as the major components of the various lipids § In higher plant and animals, the predominant fatty acid residues are those of the C 16 and C 18 species palmitic, oleic, linoleic, and steric acids.

§ Fatty acids with <14 or >20 carbon atoms are uncommon. § Most fatty acids have an even number of carbon atoms because they are usually biosynthesized by the condensation of C 2 units. § Over half of the fatty acid residues of plant and animal lipids are § Unsaturated (contain double bonds) and § Are often polyunsaturated (contain two or more double bonds)

Bacterial fatty acids Are rarely polyunsaturated § Commonly branched, hydroxylated § Contain waxes (esters of fatty acids and long-chain alcohols) produced by certain plants. §

Figure 6. 1 Structural formula of some C 18 fatty acids. The double bonds all have the cis conformation.
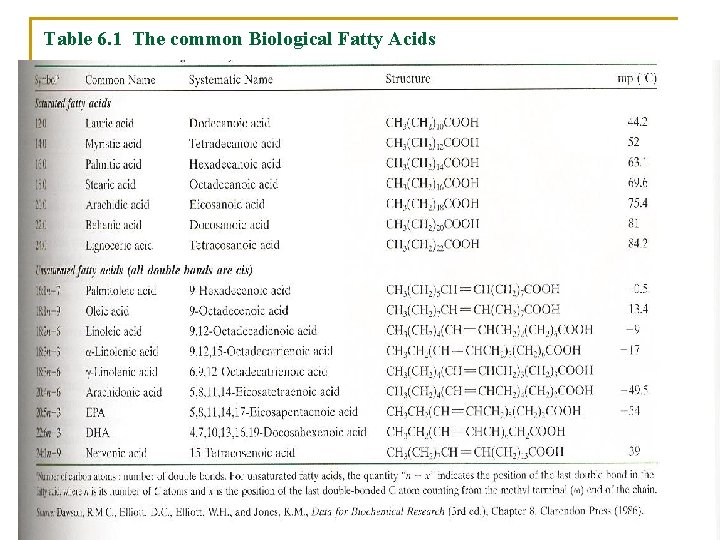
Table 6. 1 The common Biological Fatty Acids

1. The Physical Properties of fatty acids vary with their degree of unsaturation Ø The first double bond of an unsaturated fatty acid commonly occurs between its C 9 and C 10 atoms counting from the carboxyl C atom (a Δ 9 - or 9 -double bond). Ø In polyunsaturated fatty acids, the double bonds tend to occur at every third carbon atom toward the methyl terminus of the molecule (such as –CH=CH–CH 2– CH=CH–). Ø Double bonds in polyunsaturated fatty acids are almost never conjugated (as in –CH=CH–).

Ø Triple bonds rarely occur in fatty acids or any other compound of biological origin. Ø Two important classes of polyunsaturated fatty acids are denoted n– 3 (or ω– 3) and n-6 (or ω– 6) fatty acids. Ø This nomenclature identifies the last doublebonded carbon atom as counted from the methyl terminal (ω) end of the chain.

Ø Fatty acid double bonds almost always have the cis configuration: Ø This puts a rigid 30° bend in the hydrocarbon chain of unsaturated fatty acids that interferes with their efficient packing to fill space. Ø The consequent reduced Van der Waals interactions cause fatty acid melting points to decrease with their degree of unsaturation Ø Lipid fluidity likewise increases with the degree of unsaturation of their component fatty acid residues Ø This phenomenon has important consequences for membrane properties.

Ø Saturated fatty acids are: 1. Highly flexible molecules 2. Can assume a wide range of conformations because there is relatively free rotation about each of their C–C bonds. 3. Their fully extended conformation is that of minimum energy because this conformation has the least amount of steric interference between neighboring methylene groups. 4. Melting points (mp) of saturated fatty acids, increase with molecular mass
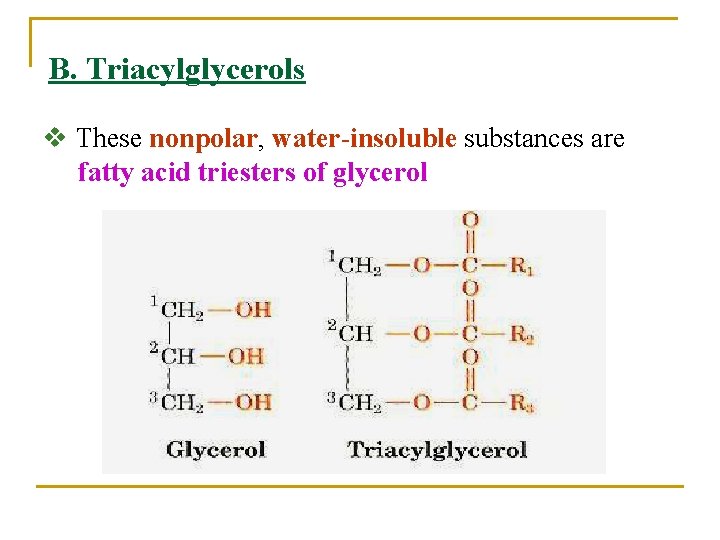
B. Triacylglycerols v These nonpolar, water-insoluble substances are fatty acid triesters of glycerol

v. Fats and oil that occur in plants and animals consist largely of mixtures of Triacylglycerols (also referred to as triglycerides or neutral fats). v Triacylglycerols function as energy reservoirs in animals, therefore v They are NOT components of biological membranes. v Triacylglycerols differ according to the identity and placement of their three fatty acid residues. 1. Simple triacylglycerols contain one type of fatty acid residue and are named accordingly. For example: A) Tristearoylglycerol or tristearin contains three stearic acid residues B) Trioleoylglycerol or triolein has three oleic acid residues.

2. Mixed triacylglycerols contain two or three different types of fatty acid residues and are named according to their placement on the glycerol moiety. v Fat and oils: v Are differ only in that: v Fats are solid v Oils are liquid at room temperature v. Are complex mixtures of simple and mixed triacylglycerols v Plant oils: v Are usually richer in unsaturated fatty acid residues than are animal fats

1. Triacylglycerols are efficient energy reserves 2. • Fats are highly efficient form in which to store metabolic energy: • This is because fats are less oxidized than are carbohydrates or proteins and hence yield significantly more energy on oxidation • Fats, being nonpolar substances, are stored in anhydrous form: • Whereas glycogen bind about twice its weight of water

• Fats therefore provide about six times: • The metabolic energy of an equal weight of hydrates glycogen. • In animals, • Adipocytes (fat cells) are specialized for the synthesis and storage of triacylglycerols.

• Adipose tissue is most abundant in a subcutaneous layer and in the abdominal cavity • Fat content of normal human (21% for men, 26% for women) enables them to survive starvation for 2 to 3 months. • In contrast, the body’s glycogen supply, which functions are a short-term energy store, can provide for the body’s metabolic need for less than a day. • The subcutaneous fat layer also provides thermal insulation.

C. Glycerophospholipids are major lipid components of biological membranes • Glycerophospholipids (or phosphoglycerides) they consist of: 1. Glycerol-3 -phosphate (Figure 6. 2 a) • Esterified at its C 1 and C 2 positions to fatty acids • Phosphoryl group connect to a group (X) to form the class of substances as in Figure 6. 2 b. • Glycerophospholipids are therefore amphiphilic molecules with: • Nonpolar aliphatic tails • Polar Phosphoryl -X heads
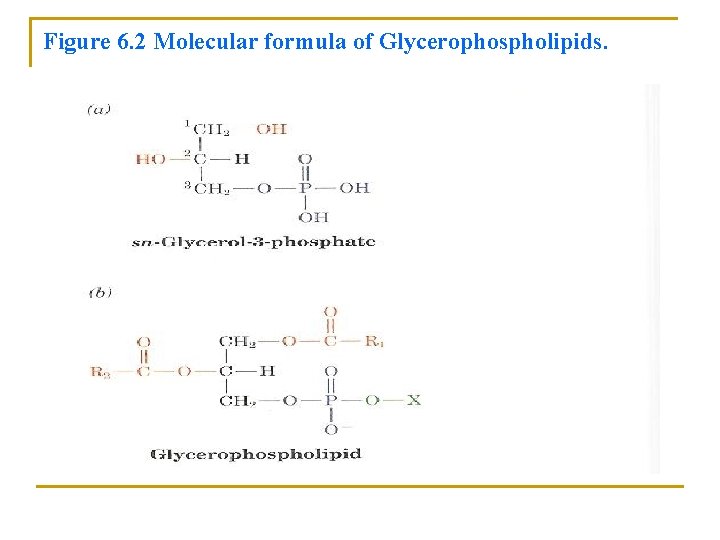
Figure 6. 2 Molecular formula of Glycerophospholipids.
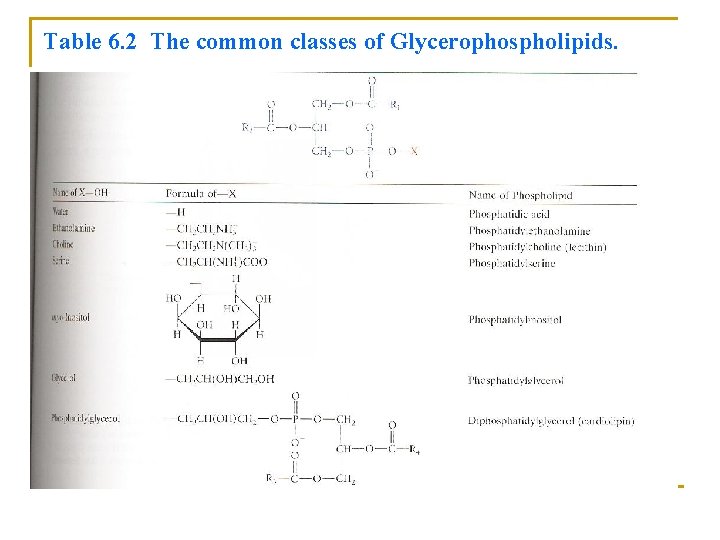
Table 6. 2 The common classes of Glycerophospholipids.

• The simplest Glycerophospholipids, in which X=H, are phosphatidic acids; they are present only in small amounts in biological membranes (Table 6. 2). • Glycerophospholipids are named according to the identities of these fatty acid residues. • Some glycerophospholipids have common names: 1. Phosphatidylcholines: known as lecithins 2. Diphosphtidylglycerols: known as cardiolipins (first isolated from heart muscle)

D. Sphingolipids comprise a second class of molecules in biological membranes ØSphingolipids are also major membrane components ØAre derivatives of the C 18 amino alcohols sphingosine, dihydrosphingosine (Fig. 6. 3), and their C 16, C 17, C 19, and C 20 homologs. Ø Their N-acyl fatty acid derivatives 1. Ceramides a) Occur only in small amounts in plant and animal tissues but form the parent compounds of more abundant Sphingolipids:

Figure 6. 3 Molecular formulas of sphingosine and dihydrosphingosine. The chiral centers at C 2 and C 3 of sphingosine and dihydrosphingosine have the configurations shown in Fisher projection. The double bond in sphingosine has the trans configuration.

1. Sphingomyelins • Are the most common Sphingolipids • Are ceramides bearing either a phospocholine (Fig. 6. 4) or a phosphoethanolamine moiety • They can also be classified as sphingophposholipids • Sphingomyelines differ chemically from phosphataidylcholine and phosphatidylethanolamine but their conformations and charge distributions are quite similar
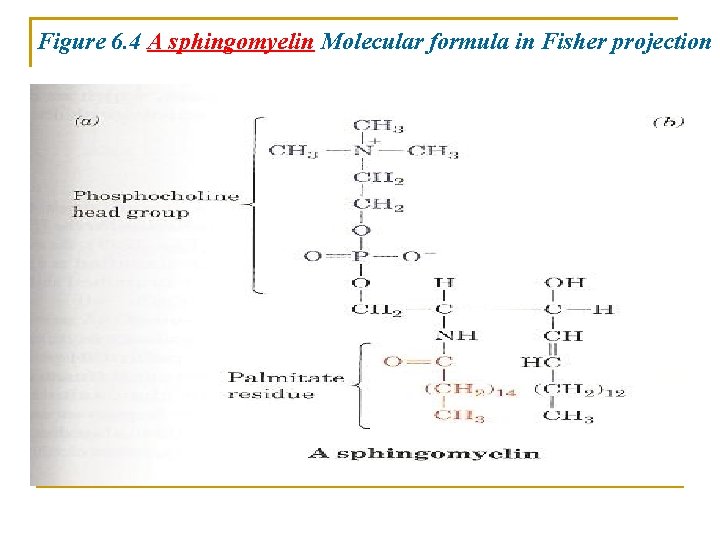
Figure 6. 4 A sphingomyelin Molecular formula in Fisher projection
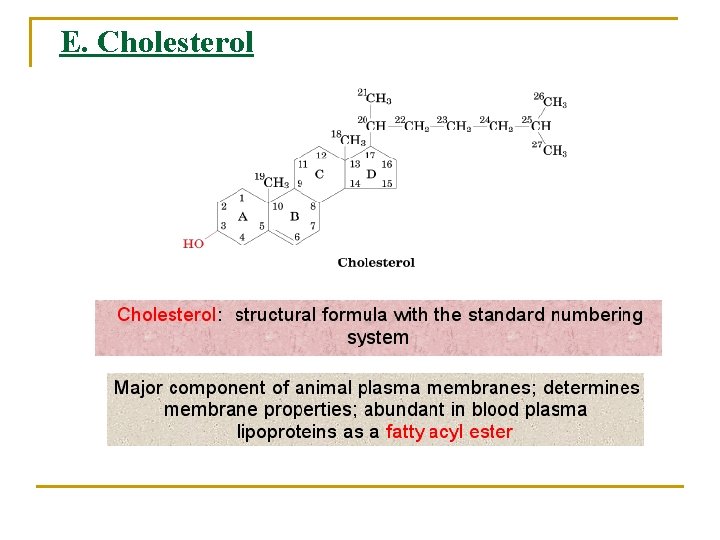
E. Cholesterol

Ø Its polar OH group gives it a weak amphiphilic character, whereas its fused ring system provides it with greater rigidity than other membrane lipids Ø It is also abundant in blood plasma lipoproteins Ø Cholesterol is the metabolic precursor of steroid hormones: Ø Substances that regulate a great variety of physiological functions including: Ø Sexual development Ø Carbohydrate metabolism.

Ø Plants contain little cholesterol. Rather, the most common sterol components of their membranes are which differ form cholesterol only in their aliphatic side chains. Ø Yeast and fungi have yet other membrane sterols such as ergosterol, which has a C 7 to C 8 double bond. Ø Prokaryotes: with the exception of mycoplasmas, contain little, if any, sterol.

Membrane Glycerophospholipids and Sphingolipids Spontaneously Form Bilayers v Soaps are the salts of fatty acids 1. Are produced by treating fatty acids with alkali (Figure 6. 5). 2. These soaps are the simplest types of molecules that behave like lipids in aqueous solution. 3. They can form ordered aggregates known as monolayers, and bilayers. v A monolayers form when soap is gently spread on the surface of a water (Figure 6. 6).

Figure 6. 5: production of soup molecule. Treatment of a fatty acid such as palmitic acid with sodium hydroxide [Na. OH(aq)] produces the sodium salt of the carboxylate anion, a molecule called a soap. The negative charge of carboxylate group is balanced by the positively charged Na+ ions.
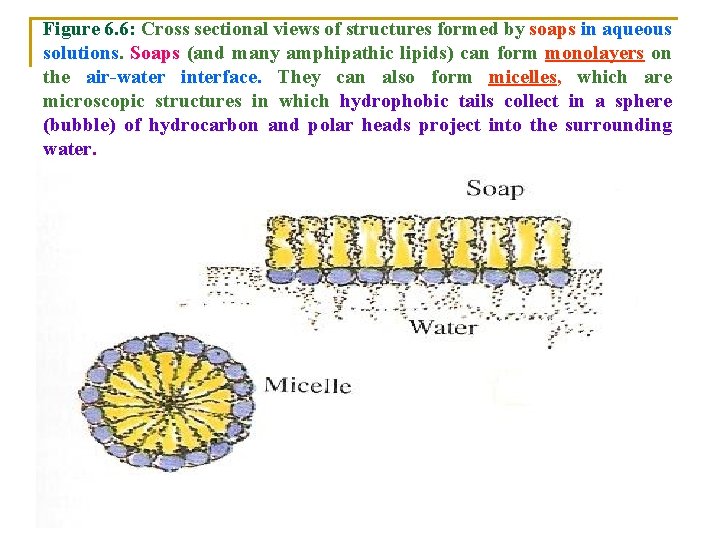
Figure 6. 6: Cross sectional views of structures formed by soaps in aqueous solutions. Soaps (and many amphipathic lipids) can form monolayers on the air-water interface. They can also form micelles, which are microscopic structures in which hydrophobic tails collect in a sphere (bubble) of hydrocarbon and polar heads project into the surrounding water.

v In 1)Monolayers: v The hydrophilic polar heads dip into the water. v Thus, only one side of the soap molecules is exposed to water. v 2)Micelles: v. Are Groups of soap molecules that are surrounded by water its v Polar heads: form an outer rim (frame), which is exposed to and interacts with water. v Nonpolar fatty acid tails: associate with one another in the interior of the structure.

v Soap bubbles are the 3)bilayers of soap molecules (Figure 5. 7). v In a Soap bubble: v A thin layer of water is packed into the center of the Bilayers v Surrounded by the hydrophilic polar heads of the soap molecules which face each other within the interface of the bilayer. v. The hydrophobic hydrocarbon tails are: v Beside one another v Extend into the hydrophobic air on each side of the bubble.
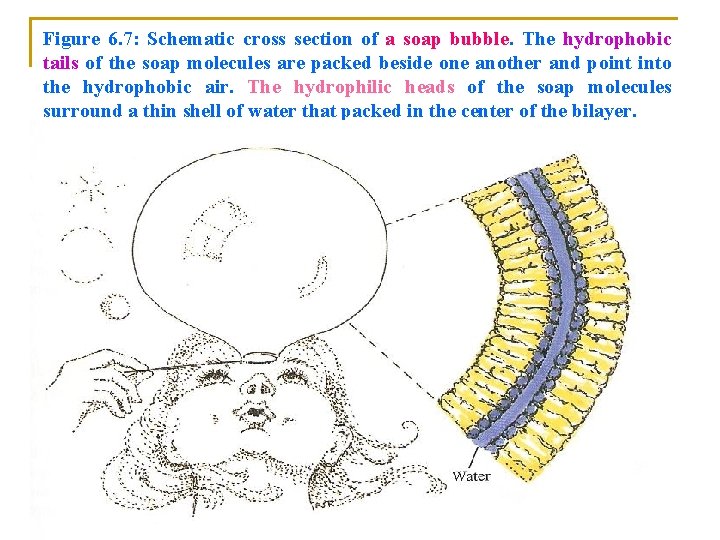
Figure 6. 7: Schematic cross section of a soap bubble. The hydrophobic tails of the soap molecules are packed beside one another and point into the hydrophobic air. The hydrophilic heads of the soap molecules surround a thin shell of water that packed in the center of the bilayer.

v Glycerophospholipids and Sphingolipids, like soaps, form monolayers. v They spontaneously form lipid bilayers, which are the bases of living biological membranes. v The two hydrocarbon chains of these lipids are: v Too bulky to pack well into micelles v But they fit nicely into bilayers

n n n Unlike soap bilayers: q Lipid bilayers are oriented: n With their hydrophobic tails inside the bilayers n While the hydrophilic heads are in contact with the aqueous solution on each side (Figure 6. 8). Charged polar head group of two layers: q Provides both surfaces with an ionic coat. Liposomes are: q Phospholipids bilayers that : n Enclose (put in) an aqueous compartment can be formed in high yield in the laboratory under carefully controlled conditions (Figure 6. 9).
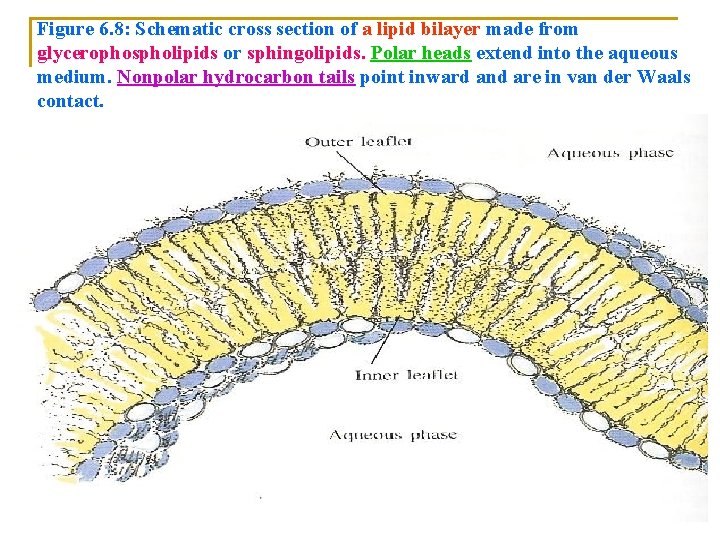
Figure 6. 8: Schematic cross section of a lipid bilayer made from glycerophospholipids or sphingolipids. Polar heads extend into the aqueous medium. Nonpolar hydrocarbon tails point inward and are in van der Waals contact.
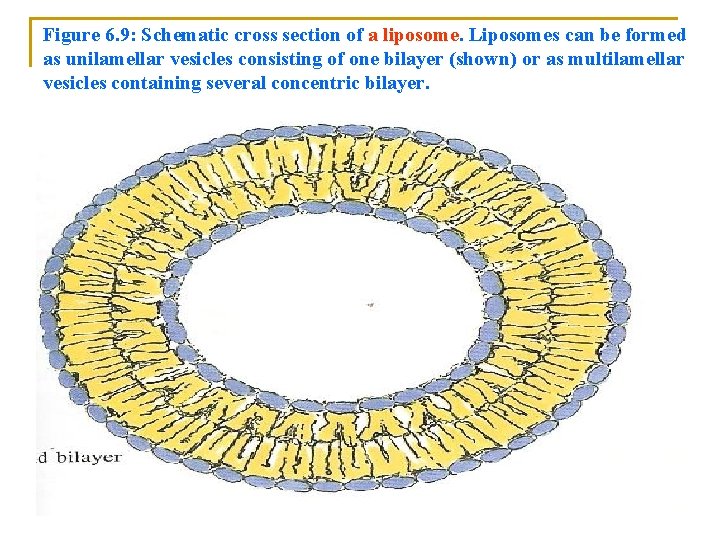
Figure 6. 9: Schematic cross section of a liposome. Liposomes can be formed as unilamellar vesicles consisting of one bilayer (shown) or as multilamellar vesicles containing several concentric bilayer.

v Not all lipids form bilayers. v. A lipid bilayer can form only: when the cross-sectional areas of the hydrophobic tail and the polar head are about equal. v Glycerophospholipids and Sphingolipids form bilayers, v Lysophospholipids, which have only one fatty acyl group, do NOT form bilayers because the heads are too large. v Cholesterol do NOT form bilayers because the rigid fused systems and additional nonpolar tails are too large.

Lipid Bilayers Are Fluid Ø A lipid bilayer is about 6 mm across. This is so thin that we can regard the membrane as a twodimensional fluid. Ø Lipid molecules in a bilayer are highly oriented. Ø Lipids DO NOT readily cross from one leaflet of the bilayer to the other; thus, little exchange (change over) occurs between lipids in the outer and inner leaflets.

Ø Once within the hydrocarbon phase, the polar head would experience no stabilizing interactions. Ø There is large potential energy barrier to movement of the lipid across the bilayer. Ø This strong orientation of lipids within each leaflet gives the bilayer some of the properties of a crystal.

n Diffusion of a phospholipids: n Laterally (side way) n Parallel to the plane of the bilayer n is rapid For example: n q In a bacterial membrane about 2μm long, a lipid diffuses from one end to the other in one second. n As a result of this free lateral diffusion, a lipid bilayer can be described as n A liquid crystal.

Lipid Bilayers Are Selective. Permeability Barriers § The hydrophobic hydrocarbon chains in the lipid bilayer of a sealed vesicle form a phase boundary that separates the aqueous compartment inside the vesicle from its aqueous surroundings. § This thin layer of hydrocarbon acts as a selectivepermeability barrier, which allows hydrophobic molecules to diffuse across the bilayer at relatively rapid rates while serving as an almost impenetrable barrier to hydrophilic molecules.

§ Liposomes: § Can be used to study the permeability of lipid bilayers to various substances. § These synthetic vesicles provide a model fit the study of passive diffusion, also called simple diffusion, which can be defined as unaided spontaneous diffusion across a lipid bilayer. § The direction in which net diffusion across a bilayer occurs depends upon the concentration of a solute inside and outside the vesicle.

§ The difference in the concentration of a substance on the two sides of the bilayer is called the concentration gradient. § Spontaneous diffusion occurs down the concentration gradient from the side with the higher concentration to the side with the lower concentration. § The rate of diffusion of a substance across a lipid bilayer parallels the solubility of that substance in a nonpolar solvent relative to its solubility water.

§ Hydrophobic molecules are more soluble in nonpolar solvents (which have properties similar to those of the hydrocarbon interior of the bilayer) than they are in water § Therefore, they diffuse across lipid bilayers fairly rapidly.

§ Polar and ionic substances, On the other hand, are much less soluble in nonpolar solvents than they are in water. § As a result, Polar and ionic substances diffuse slowly across the hydrophobic hydrocarbon interior of the bilayer. § Polar molecules such as glucose and small ions such as Na+ lose their solvation spheres as they move through gaps in lipid molecules. § These gaps open infrequently for short periods of time as a result of the random motion of the lipid molecules.

§ Once the polar molecule or ion reaches the other side of the bilayer, it regains a solvent coat. § Water is a surprising exception to this rule that polar molecules diffuse slowly across lipid bilayer.

§ The high concentration of water (55. 5 M) surrounding the bilayer facilitates the diffusion of water molecules. § Therefore, even though water is a relatively polar substance, it diffuses more rapidly than other polar substances as a result of its abundance.

Biological Membranes are Fluid Mosaic of Protein in a Phospholipid Bilayer v Lipids give biological membranes their basic structural character, but proteins are necessary for the function of these membranes. v Functions performed by membrane proteins are 1) Transportation of molecules across the lipid bilayer 2) Transduction of signals across the membrane.

v The existence of two broad classes of membrane proteins: 1) Integral (internal) membrane proteins üIntegral membrane proteins are embedded in lipid bilayers üPortions of these proteins are in van der Waals contact with the hydrophobic region of the membrane üIntegral membrane proteins may be exposed either to the exterior surface of the membrane or to the interior compartment, or they may span (cross) the bilayer completely.

ü Singer and Nicolson further hypothesized that: ü Integral membrane proteins diffuse freely parallel to the plan of the lipid bilayer like icebergs in lipid sea. ü This model of membrane structure is called the fluid mosaic model (Figure 6. 10).

2) Peripheral membrane proteins üPeripheral membrane proteins may be weakly bound to the surface of the lipid bilayer by: a) Ionic interactions b) Hydrogen bonds that form between the protein and the polar heads of the membrane lipids. üThey may also interact with integral membrane proteins.

v These membrane proteins may be attached to either the interior or exterior surface of the bilayer.
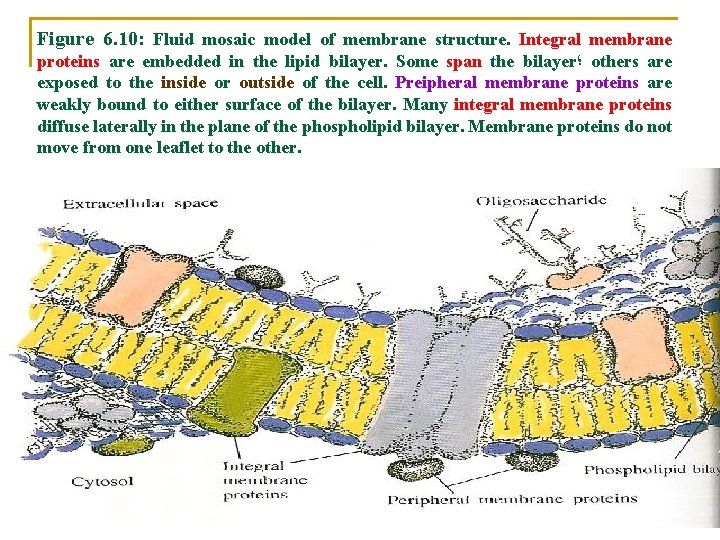
Figure 6. 10: Fluid mosaic model of membrane structure. Integral membrane proteins are embedded in the lipid bilayer. Some span the bilayer ؛ others are exposed to the inside or outside of the cell. Preipheral membrane proteins are weakly bound to either surface of the bilayer. Many integral membrane proteins diffuse laterally in the plane of the phospholipid bilayer. Membrane proteins do not move from one leaflet to the other.
- Slides: 56