Chapter 6 Lecture Basic Chemistry Fourth Edition Chapter

Chapter 6 Lecture Basic Chemistry Fourth Edition Chapter 6 Ionic and Molecular Compounds 6. 6 Organic Compounds Learning Goal Identify the properties characteristic of organic and inorganic compounds. © 2014 Pearson Education, Inc.

Organic Compounds Organic compounds • always contain carbon and hydrogen, and sometimes oxygen, sulfur, nitrogen, phosphorus, or a halogen • occur in nature and are also found in fuel, shampoos, cosmetics, perfumes, and foods • are the foundation for understanding biochemistry © 2014 Pearson Education, Inc.

Organic Compounds, Properties Formulas for organic compounds are written with carbon first, followed by hydrogen and then other elements. Organic compounds • have low melting and boiling points • are not soluble in water and are less dense than water • undergo combustion, burning vigorously in air © 2014 Pearson Education, Inc.
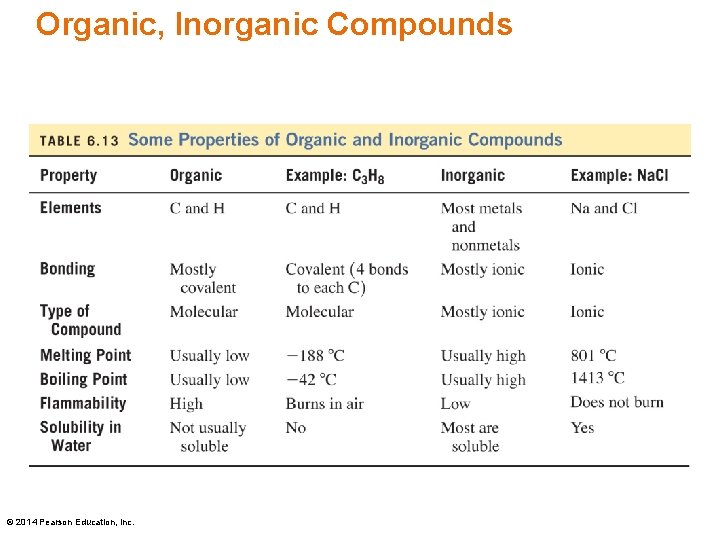
Organic, Inorganic Compounds © 2014 Pearson Education, Inc.

Organic, Inorganic Compounds Propane, C 3 H 8, is an organic compound, whereas sodium chloride, Na. Cl, is an inorganic compound. © 2014 Pearson Education, Inc.

Bonding in Organic Compounds Hydrocarbons are organic compounds that contain only carbon and hydrogen. In the electron-dot formulas for hydrocarbons, each shared pair of electrons represents a single covalent bond. Saturated hydrocarbons contain only single carbon-carbon bonds. Expanded structural formulas are drawn to show the bonds between all the atoms. © 2014 Pearson Education, Inc.
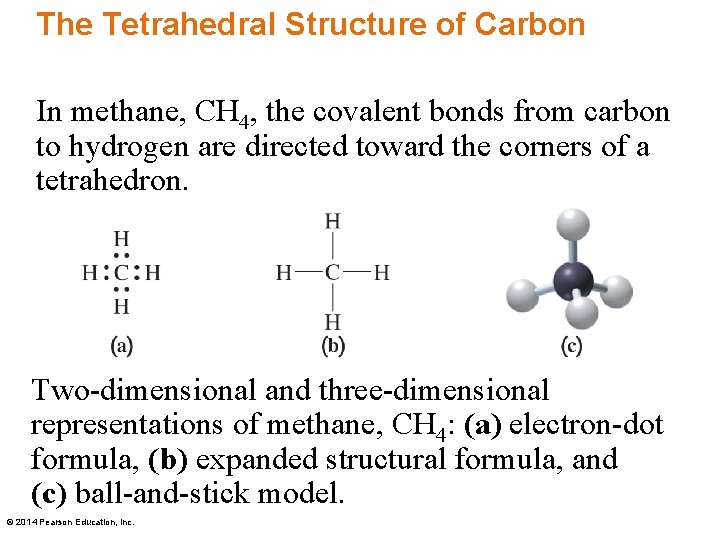
The Tetrahedral Structure of Carbon In methane, CH 4, the covalent bonds from carbon to hydrogen are directed toward the corners of a tetrahedron. Two-dimensional and three-dimensional representations of methane, CH 4: (a) electron-dot formula, (b) expanded structural formula, and (c) ball-and-stick model. © 2014 Pearson Education, Inc.
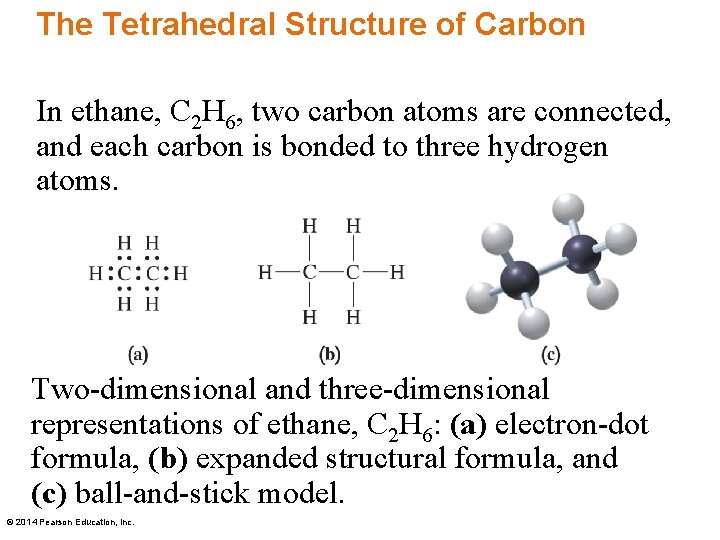
The Tetrahedral Structure of Carbon In ethane, C 2 H 6, two carbon atoms are connected, and each carbon is bonded to three hydrogen atoms. Two-dimensional and three-dimensional representations of ethane, C 2 H 6: (a) electron-dot formula, (b) expanded structural formula, and (c) ball-and-stick model. © 2014 Pearson Education, Inc.

Learning Check Identify each of the following characteristics as most typical of organic or inorganic compounds. A. has a high melting point B. is soluble in water C. has the formula, CH 3─CH 2─CH 3 D. burns easily in air E. has ionic bonds © 2014 Pearson Education, Inc.

Solution Identify each of the following characteristics as most typical of organic or inorganic compounds. A. has a high melting point inorganic B. is insoluble in water organic C. has the formula, CH 3─CH 2─CH 3 organic D. burns easily in air organic E. has ionic bonds inorganic © 2014 Pearson Education, Inc.
- Slides: 10