Chapter 6 How Cells Harvest Chemical Energy Biology

Chapter 6 How Cells Harvest Chemical Energy Biology IIH Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Anderson County High School
Introduction

How Is a Marathoner Different from a Sprinter?

How Is a Marathoner Different from a Sprinter? § Individuals inherit various percentages of the two main types of muscle fibers, slow and fast – The difference between the two is the process each uses to make ATP – Slow fibers use oxygen (aerobically) – Fast fibers do not use oxygen (anaerobic)

How Is a Marathoner Different from a Sprinter? § Percentage of slow and fast muscle fibers determines the difference between track athletes – More slow fibers; best long-distance runners – More fast fibers; good sprinters § All cells harvest chemical energy (ATP) from food

• Dark meat of a cooked turkey is slow fiber muscle – Leg muscles support sustained activity • White meat consists of fast fibers – Wing muscles allow for quick bursts of flight

Think About It

Life is Work! • Energy flows into an ecosystem as sunlight and leaves as heat (entropy) • Photosynthesis generates O 2 and organic molecules, which are used in cellular respiration • Cells use chemical energy stored in organic molecules to regenerate ATP • ATP powers work
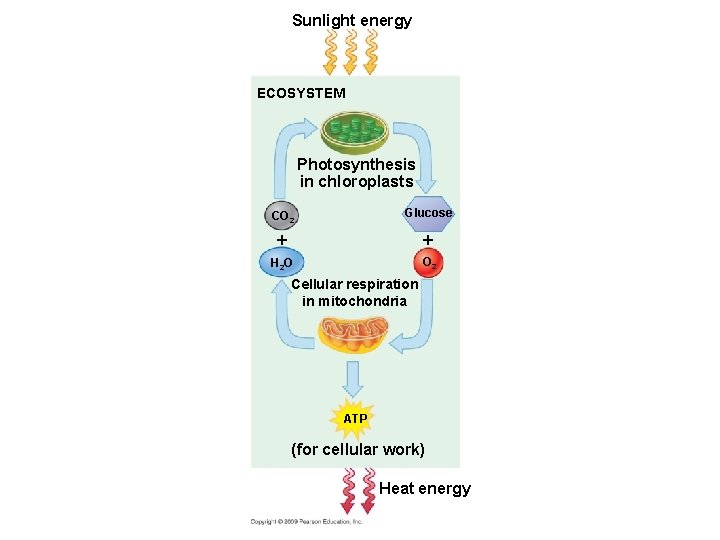
Sunlight energy ECOSYSTEM Photosynthesis in chloroplasts CO 2 Glucose H 2 O O 2 Cellular respiration in mitochondria ATP (for cellular work) Heat energy

CR and Related Pathways • Fermentation is a partial degradation of sugars that occurs without O 2 • Aerobic respiration consumes organic molecules and O 2 and yields ATP • Anaerobic respiration is similar to aerobic respiration but consumes compounds other than O 2

Often Missed Points! • Plants and animals carry out cellular respiration (have mitochondria). • Cellular respiration includes both aerobic and anaerobic respiration but is often used to refer to aerobic respiration. • Carbohydrates, fats, and proteins are all used as fuel. We trace cellular respiration with using glucose to make the study easier: C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + Energy (ATP + heat)
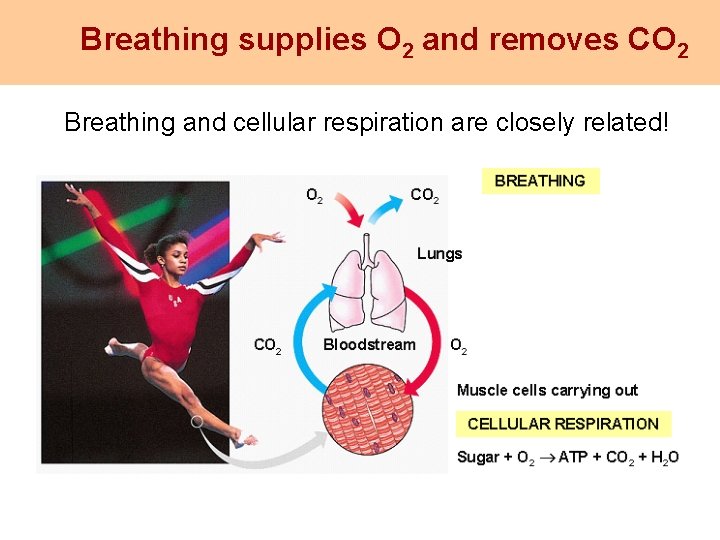
Breathing supplies O 2 and removes CO 2 Breathing and cellular respiration are closely related!

Glucose Breakdown • • • Reactants – carbohydrate & O 2 Products – H 2 O & CO 2 ATP Produced Exergonic Oxidation-reduction reaction – Glucose oxidized – O 2 reduced • “Controlled” (slow)
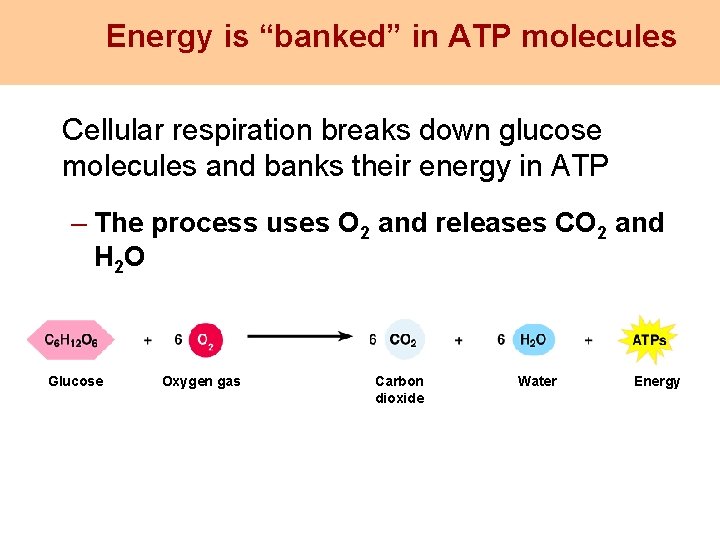
Energy is “banked” in ATP molecules Cellular respiration breaks down glucose molecules and banks their energy in ATP – The process uses O 2 and releases CO 2 and H 2 O Glucose Oxygen gas Carbon dioxide Water Energy

ATP Supplies the Energy § Average adult human needs ~ 2, 200 kcal per day – kilocalorie (kcal) - the quantity of heat required to raise the temperature of 1 kilogram (kg) of water by 1 o. C – This energy is used for body maintenance and for voluntary activities
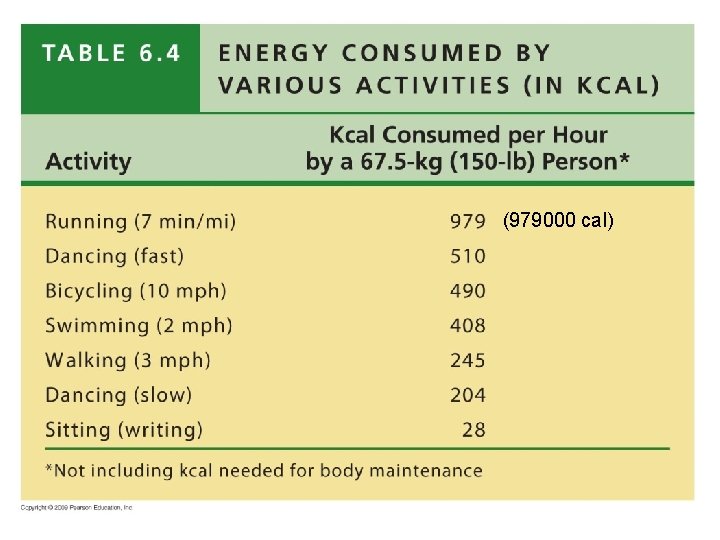
(979000 cal)

Electrons “fall” From Food to O 2 § Energy is contained in electrons in chemical bonds in organic molecules § Bonds are broken & electrons are transferred to oxygen – Oxygen has a strong tendency to attract electrons § Energy can be released from glucose by simply burning it § The energy is dissipated as heat and light and is not available to living organisms

Burning Glucose to Release Energy • Uncontrolled release of energy • Energy is dissipated as heat and light • Energy not available to living organisms

Cellular Respiration is Controlled – Energy from electrons is released in steps and in small amounts that are captured and stored in ATP – Series of reactions involves oxidation and reduction (e. g. coupled reactions)

Oxidation-Reduction Reactions Electrons flow from one molecule to another Oxidation – loss n Reduction – gain n Occur at same time
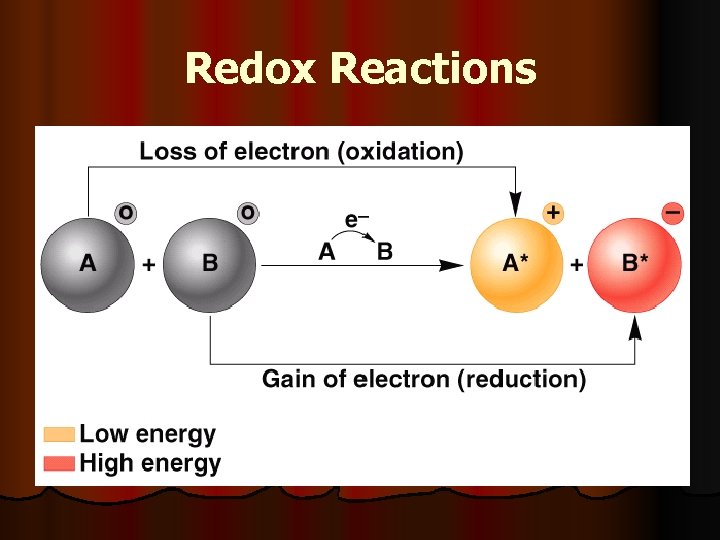
Redox Reactions
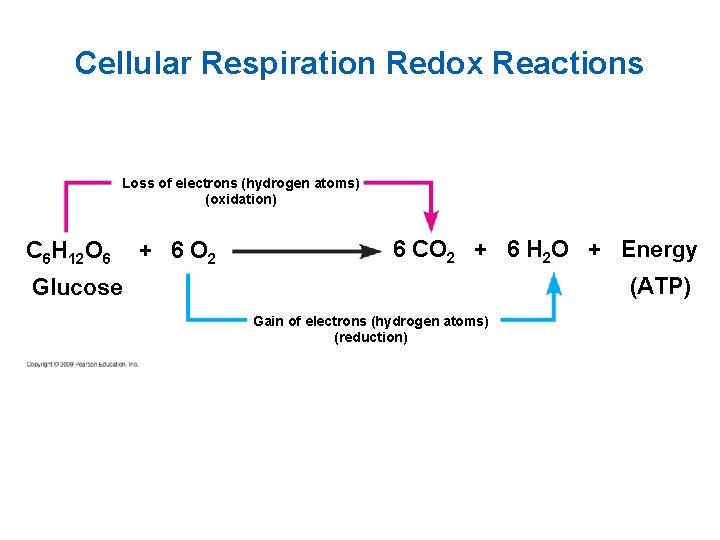
Cellular Respiration Redox Reactions Loss of electrons (hydrogen atoms) (oxidation) C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + Energy (ATP) Glucose Gain of electrons (hydrogen atoms) (reduction)

Enzyme Involvement § Enzymes are necessary – dehydrogenase - removes hydrogen (electrons) – NAD+ (nicotinamide adenine dinucleotide) - a coenzyme required by dehydrogenase to shuttle electrons – Reduced when it accepts electrons and oxidized when it gives them up Reduction Oxidation
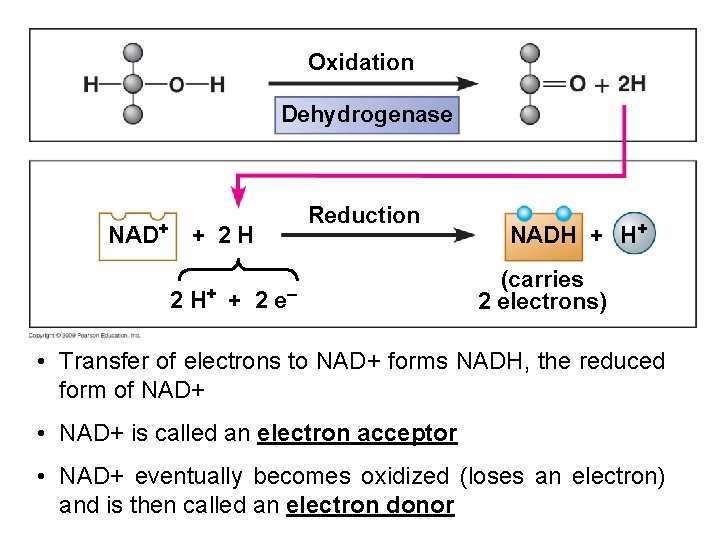
Oxidation Dehydrogenase NAD+ Reduction + 2 H 2 H+ + 2 e– NADH + H+ (carries 2 electrons) • Transfer of electrons to NAD+ forms NADH, the reduced form of NAD+ • NAD+ is called an electron acceptor • NAD+ eventually becomes oxidized (loses an electron) and is then called an electron donor

Electron Transport Chain § Other electron “carrier” molecules that function like NAD+ § Similar to a staircase where the electrons pass from one step to the next down the staircase § These electron carriers collectively are called the electron transport chain § as electrons are transported down the chain, ATP is generated
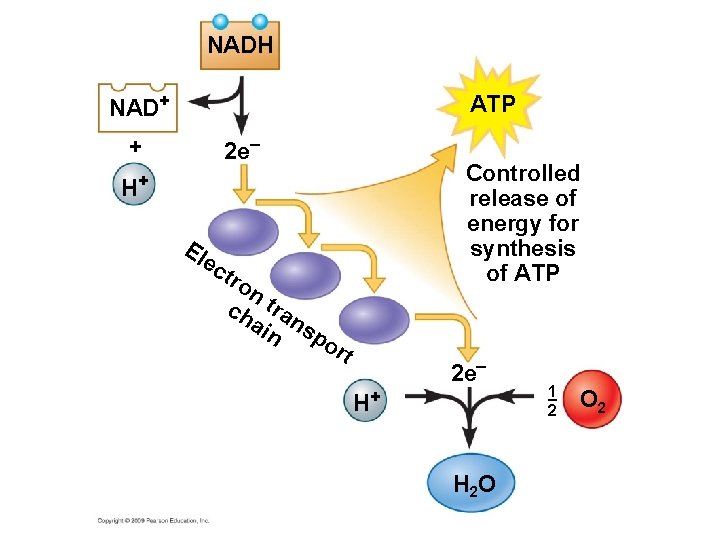
NADH NAD+ ATP + 2 e– Controlled release of energy for synthesis of ATP H+ El ec tro nt ch ra ain ns po rt 2 e– H+ H 2 O 1 2 O 2

ATP Production • Produced gradually • 39% of energy in glucose is converted to ATP • Two Methods – Substrate level phosphorylation – Oxidative phosphorylation (aerobic respiration) • Endergonic

Substrate-level Phosphorylation The formation of ATP through the transfer of a phosphate from a metabolic substrate to ADP. Metabolic substrate P ADP P ATP P P
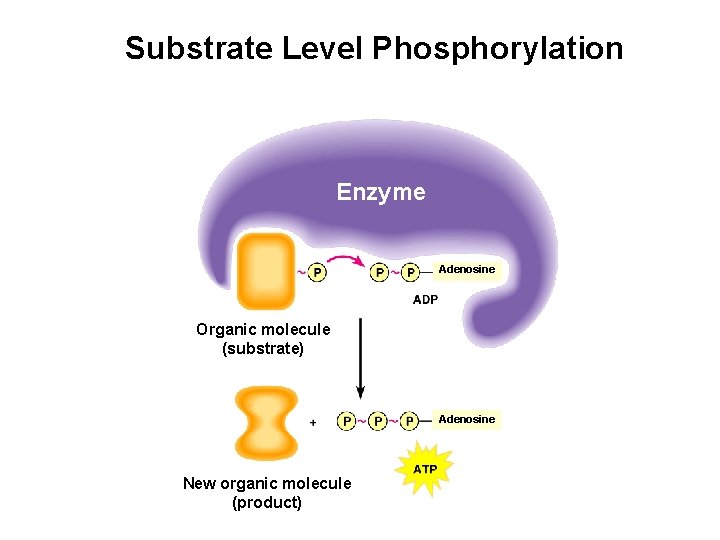
Substrate Level Phosphorylation Enzyme Adenosine Organic molecule (substrate) Adenosine New organic molecule (product)

Cellular Respiration Details

Cellular Respiration Involves 3 Steps 1. 2. 3. Glycolysis Krebs Cycle Oxidative Phosphorylation (Electron Transport & chemiosmosis)
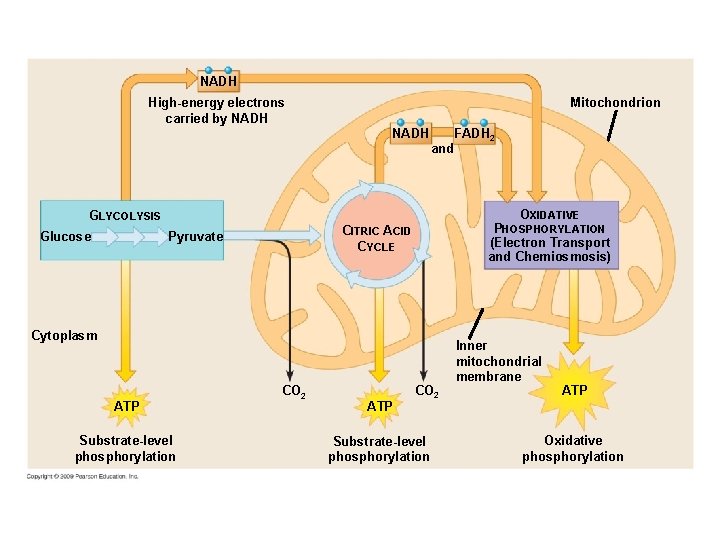
NADH Mitochondrion High-energy electrons carried by NADH FADH 2 and OXIDATIVE GLYCOLYSIS Glucose PHOSPHORYLATION (Electron Transport and Chemiosmosis) CITRIC ACID CYCLE Pyruvate Cytoplasm ATP Substrate-level phosphorylation CO 2 ATP CO 2 Substrate-level phosphorylation Inner mitochondrial membrane ATP Oxidative phosphorylation

Glycolysis

Glycolysis • Universally found in organisms • Evolved before Krebs Cycle • • • Occurs in cytosol Anaerobic ATP produced by substrate level phosphorylation

Glycolysis Single molecule of glucose is enzymatically cut in half to produce two molecules of pyruvate – Two molecules of NAD+ are reduced to two molecules of NADH – Two molecules of ATP produced by substrate-level phosphorylation – This ATP can be used immediately, but NADH must be transported through the electron transport chain to generate additional ATP
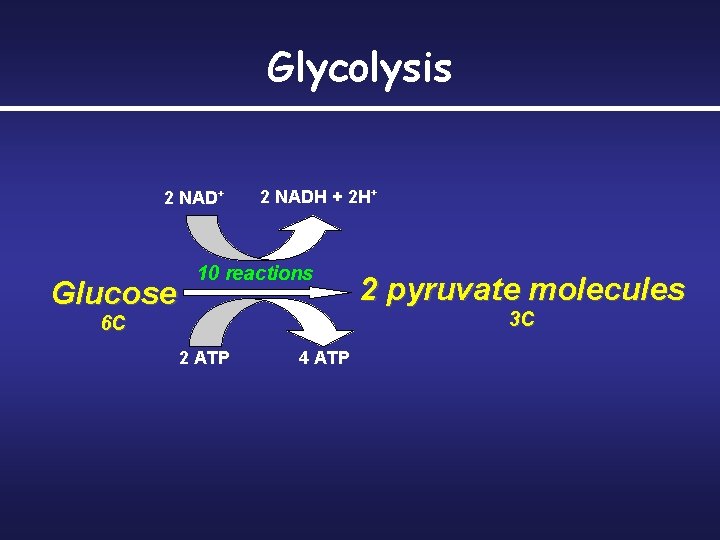
Glycolysis 2 NAD+ Glucose 2 NADH + 2 H+ 10 reactions 2 pyruvate molecules 3 C 6 C 2 ATP 4 ATP

Pyruvate is groomed for the citric acid cycle Transported to the mitochondria – First step: removal of a carboxyl group to form CO 2 – Second step: oxidization of the two-carbon compound remaining. – Third Step: coenzyme A binds to the two-carbon fragment forming acetyl coenzyme A
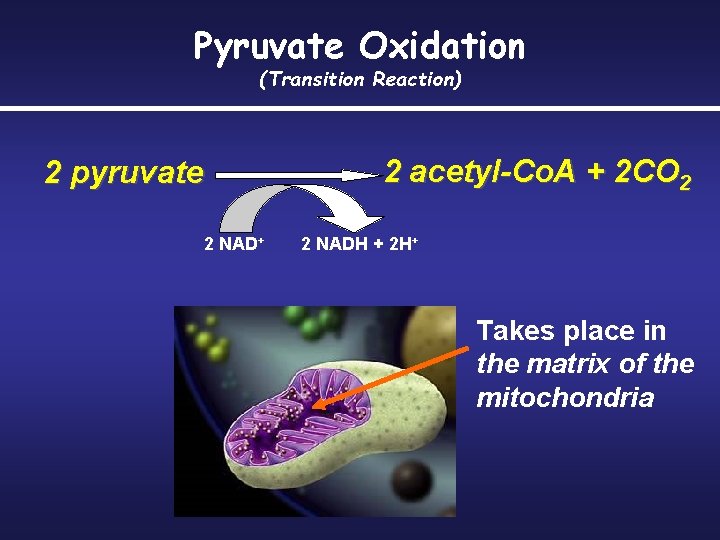
Pyruvate Oxidation (Transition Reaction) 2 pyruvate 2 NAD+ 2 acetyl-Co. A + 2 CO 2 2 NADH + 2 H+ Takes place in the matrix of the mitochondria

Krebs (Citric Acid) Cycle

Citric Acid Cycle (aka Krebs cycle) • • Cyclic Mitochondrial matrix 2 cycles for each glucose molecule 2 ATP produced by substrate level phosphorylation • NADH and FADH 2 carry electrons to ETC

Krebs Cycle Inputs • • 2 acetyl groups 2 ADP + 2 Pi 6 NAD+ 2 FAD Outputs • • 2 CO 2 2 ATP 6 NADH 2 FADH 2

What Happens in the Citric Acid Cycle § Acetyl Co. A (two-carbon compound) enters – The acetyl group associates with a four-carbon molecule forming a six-carbon molecule – The six-carbon molecule then passes through a series of redox reactions that regenerate the four-carbon molecule (thus the “cycle” designation)
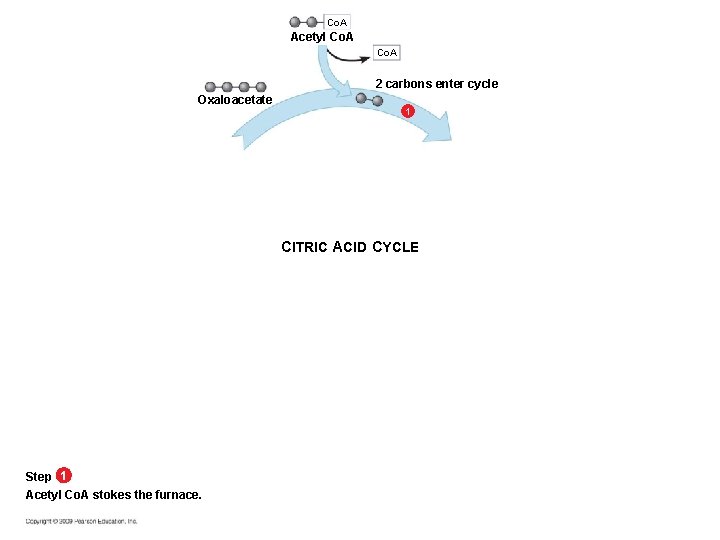
Co. A Acetyl Co. A 2 carbons enter cycle Oxaloacetate 1 CITRIC ACID CYCLE Step 1 Acetyl Co. A stokes the furnace.
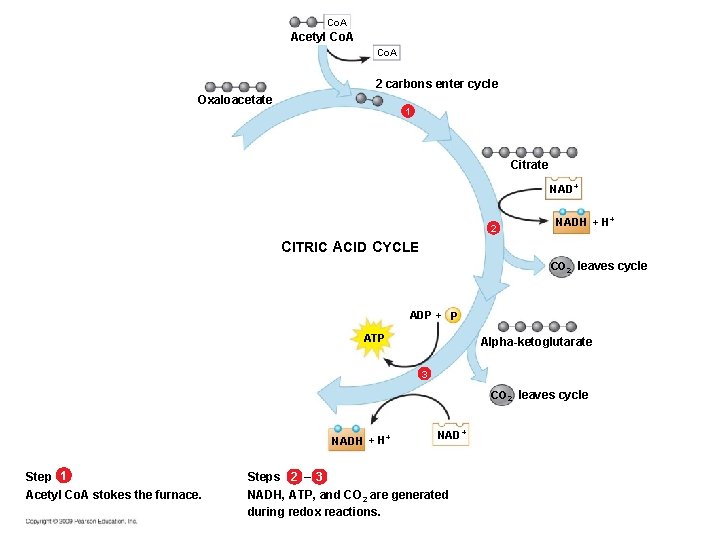
Co. A Acetyl Co. A 2 carbons enter cycle Oxaloacetate 1 Citrate NAD+ 2 NADH + H+ CITRIC ACID CYCLE CO 2 leaves cycle ADP + P ATP Alpha-ketoglutarate 3 CO 2 leaves cycle NADH + H+ Step 1 Acetyl Co. A stokes the furnace. NAD+ Steps 2 – 3 NADH, ATP, and CO 2 are generated during redox reactions.
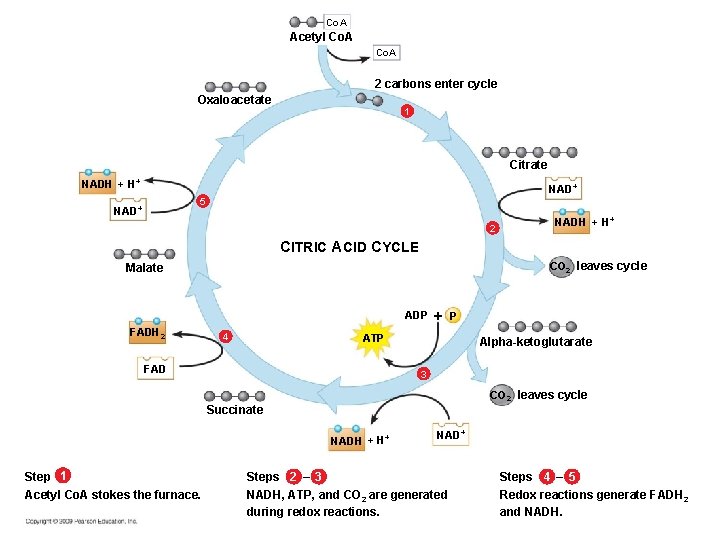
Co. A Acetyl Co. A 2 carbons enter cycle Oxaloacetate 1 Citrate NADH + H+ NAD+ 5 NAD+ 2 NADH + H+ CITRIC ACID CYCLE CO 2 leaves cycle Malate ADP P FADH 2 4 ATP FAD Alpha-ketoglutarate 3 CO 2 leaves cycle Succinate NADH + H+ Step 1 Acetyl Co. A stokes the furnace. NAD+ Steps 2 – 3 NADH, ATP, and CO 2 are generated during redox reactions. Steps 4 – 5 Redox reactions generate FADH 2 and NADH.

Oxidative Phosphorylation

Oxidative Phosphorylation The formation of ATP through the release of energy by the electron transport chain. Involves Electron Transport Chain Chemiosmosis Requires oxygen.
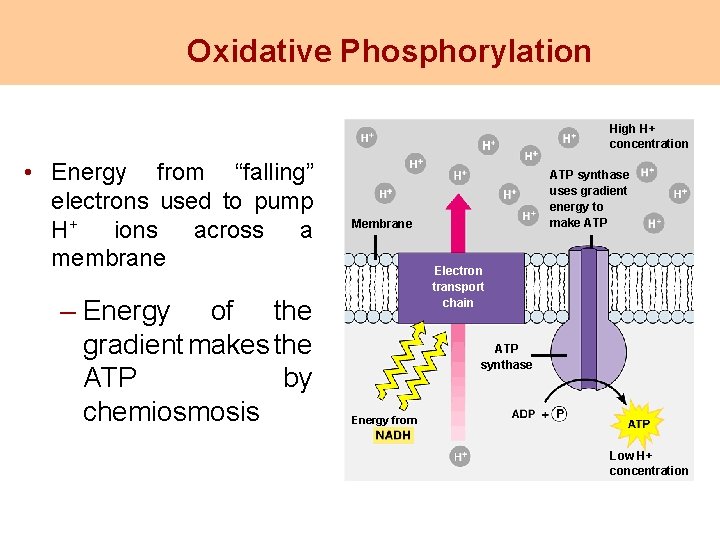
Oxidative Phosphorylation High H+ concentration • Energy from “falling” electrons used to pump H+ ions across a membrane – Energy of the gradient makes the ATP by chemiosmosis ATP synthase uses gradient energy to make ATP Membrane Electron transport chain ATP synthase Energy from Low H+ concentration

Electron Transport Chain • In cristae (inner membrane) of mitochondria • High energy electrons enter, low energy electrons leave • Carriers are NADH, FADH 2 and cytochrome molecules • The ETC creates a proton gradient used to make ATP.

Chemiosmosis An energy coupling mechanism that uses energy stored in the form of a hydrogen ion gradient across a membrane to drive cellular work, such as the synthesis of ATP. Most ATP synthesis in cells occurs by chemiosmosis.
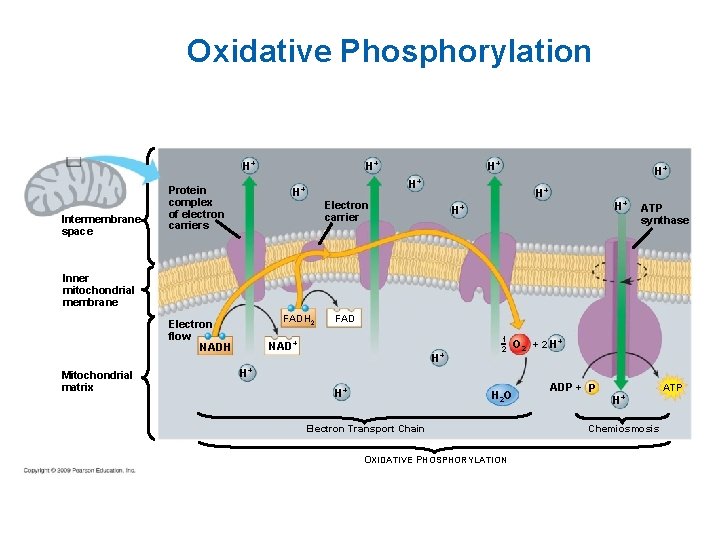
Oxidative Phosphorylation Intermembrane space Protein complex of electron carriers H+ H+ Electron carrier H+ H+ ATP synthase Inner mitochondrial membrane FADH 2 Electron flow NADH Mitochondrial matrix FAD NAD+ H+ 1 2 O 2 + 2 H+ H+ H+ H 2 O Electron Transport Chain OXIDATIVE PHOSPHORYLATION ADP + P H+ Chemiosmosis ATP


Cellular Respiration Energy Yield Summary 36 ATP (Net) (Most eukaryotic cells) Substrate –level phosphorylation net gain 2 ATP from glycolysis 2 ATP Krebs Oxidative phosphorylation 32 ATP

Cellular Respiration Energy Yield Summary 38 ATP (Bacteria & Heart & Liver Cells) Substrate –level phosphorylation net gain 4 ATP from glycolysis matrix 2 ATP Krebs in mitochondria Oxidative phosphorylation 34 ATP
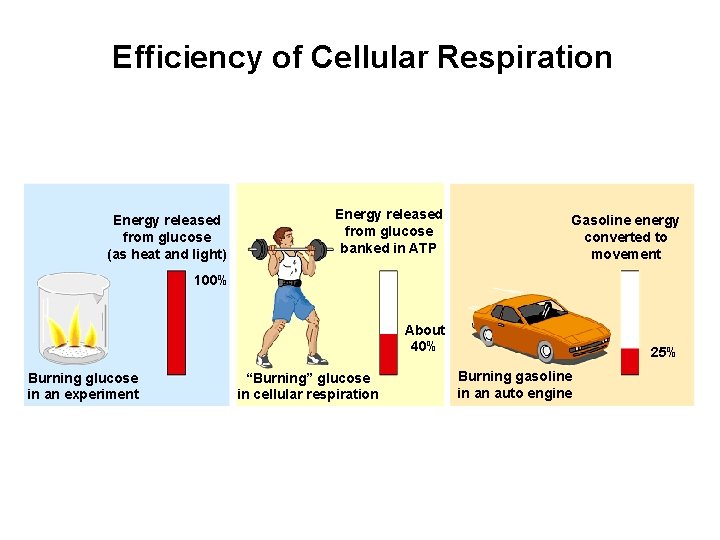
Efficiency of Cellular Respiration Energy released from glucose (as heat and light) Energy released from glucose banked in ATP Gasoline energy converted to movement About 40% 25% 100% Burning glucose in an experiment “Burning” glucose in cellular respiration Burning gasoline in an auto engine

Cellular Respiration Poisons § Mechanisms – Blocking of the electron transport chain (rotenone, cyanide, and carbon monoxide) – Inhibition of ATP synthase (oligomycin) – Making the membrane leaky to hydrogen ions (for example, dinitrophenol)
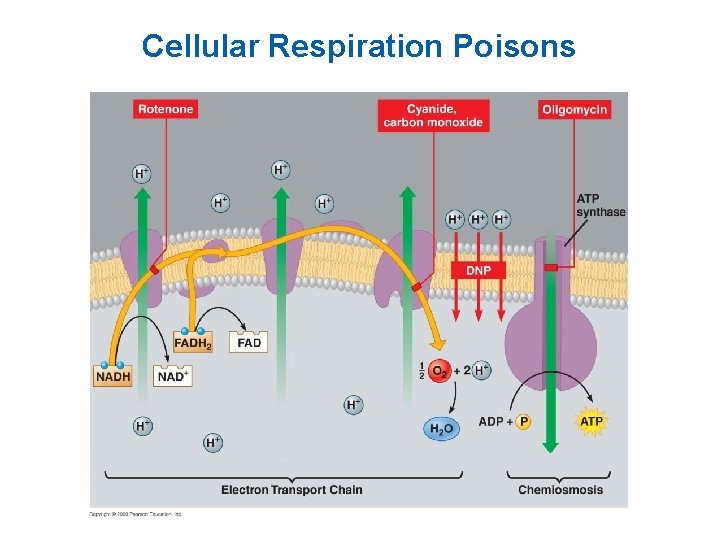
Cellular Respiration Poisons
Cellular Respiration Bioflix Review

Challenge Using a single molecule of glucose, explain what happens in cellular respiration. At the end of your “essay” write the complete, balanced equation for cellular respiration.

Fermentation

Fermentation Basics § An anaerobic (without oxygen) energy-generating process – It takes advantage of glycolysis, producing two ATP molecules and reducing NAD+ to NADH – NADH is oxidized without passing its electrons through the electron transport chain to oxygen
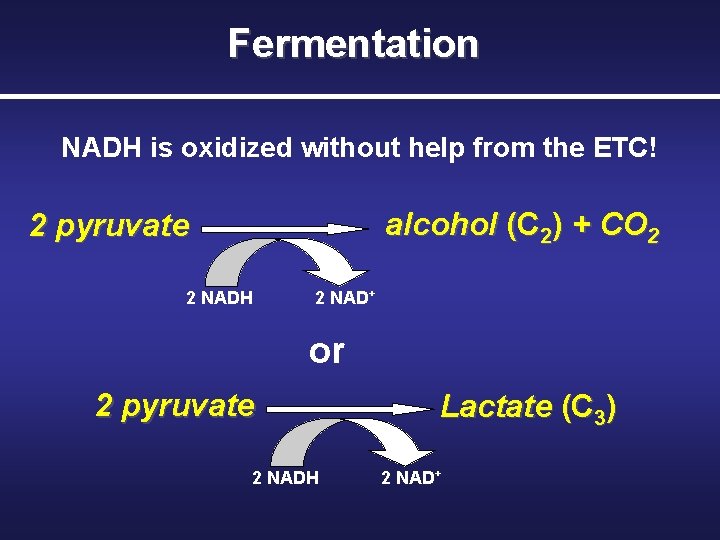
Fermentation NADH is oxidized without help from the ETC! alcohol (C 2) + CO 2 2 pyruvate 2 NADH 2 NAD+ or 2 pyruvate 2 NADH Lactate (C 3) 2 NAD+

Fermentation • Follows glycolysis. . . – In certain anaerobic bacteria – In animals when pyruvate is produced faster that can be oxidized through Krebs Cycle • • Oxygen deficit Muscle Cells Rapid burst of ATP Lactate produced

Lactic Acid Fermentation § Your muscle cells and certain bacteria can oxidize NADH through lactic acid fermentation – Anaerobic – NADH is oxidized to NAD+ when pyruvate is reduced to lactate – In a sense, pyruvate is serving as an “electron sink, ” a place to dispose of the electrons generated by oxidation reactions in glycolysis Animation: Fermentation Overview
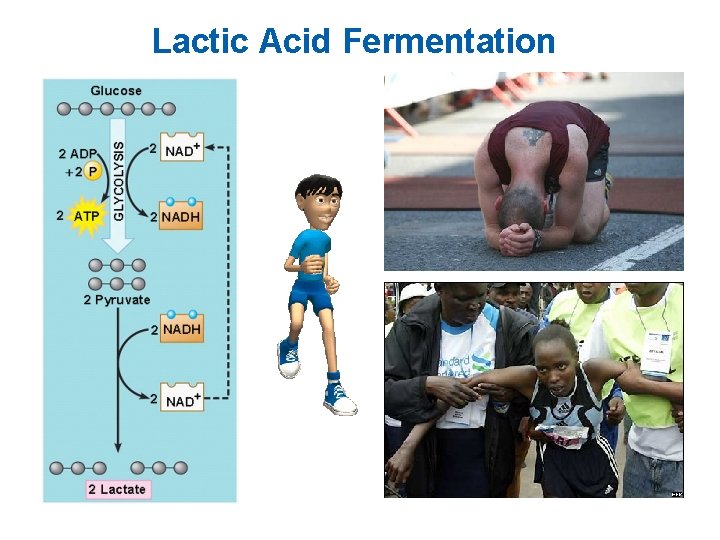
Lactic Acid Fermentation

Alcohol Fermentation § The baking and winemaking industry have used alcohol fermentation for thousands of years – Yeasts are single-celled fungi that not only can use respiration for energy but can ferment under anaerobic conditions – They convert pyruvate to CO 2 and ethanol while oxidizing NADH back to NAD+
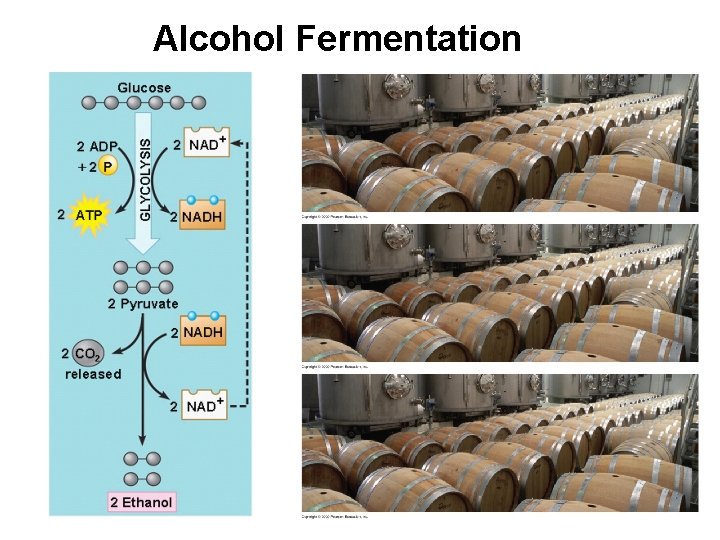
Alcohol Fermentation

Fermentation Efficiency Much less efficient (2. 1%) than complete breakdown of glucose (40%).

• Obligate anaerobes carry out fermentation or anaerobic respiration and cannot survive in the presence of O 2 • Yeast and many bacteria are facultative anaerobes (can survive using either fermentation or cellular respiration) • In a facultative anaerobe, pyruvate is a fork in the metabolic road that leads to two alternative catabolic routes

Fermentation vs. Aerobic Respiration • Both use glycolysis to oxidize glucose and other organic fuels to pyruvate • The processes have different final electron acceptors: an organic molecule (such as pyruvate or acetaldehyde) in fermentation and O 2 in cellular respiration • Cellular respiration produces 36/38 ATP per glucose molecule; fermentation produces 2 ATP per glucose molecule

Metabolic Pool

Metabolic Reactions • Catabolic reactions – Break down molecules – exergonic • Anabolic reactions – Build molecules – Synthetic reactions – Endergonic (driven by ATP)

Catabolic Reactions - Examples • Glucose breakdown • Use of fat molecules for energy • Breakdown of amino acid carbon skeletons

Anabolic Reactions - Examples • Glucose production • Synthesis of fats • Creation of proteins

Metabolic Pool Metabolites that are the product of and / or the substrates for key reactions in cells, allowing one type of molecule to be changed into another type such as the conversion of carbohydrate to fats.

Evolution of Metabolism Degradation Glycolysis Anaerobic Photosynthesis Oxygen-forming Photosynthesis Nitrogen Fixation Aerobic Respiration

Evolutionary Significance of Glycolysis • Glycolysis occurs in nearly all organisms • Glycolysis probably evolved in ancient prokaryotes before there was oxygen in the atmosphere Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Review & Summary

Aerobic Respiration - ATP Yield • Theoretical Yield = 36 • Actual Yield = 30 (32% efficient) – Inner mitochondrial membrane, “leaky” – Proton gradient used for other than ATP production

Regulating Aerobic Respiration • Controlled by ATP and ADP levels – Glycolysis stimulated by High ADP and Low ATP (stimulate phosphofructokinase) – Krebs Cycle inhibited by high Levels of ATP (inhibits citrate synthetase to shut down pathway) • Example of feedback inhibition

The Versatility of Catabolism • Catabolic pathways funnel electrons from many kinds of organic molecules into cellular respiration • Glycolysis accepts a wide range of carbohydrates • Proteins must be digested to amino acids; amino groups can feed glycolysis or the citric acid cycle Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

• Fats are digested to glycerol (used in glycolysis) and fatty acids (used in generating acetyl Co. A) • Fatty acids are broken down by beta oxidation and yield acetyl Co. A • An oxidized gram of fat produces more than twice as much ATP as an oxidized gram of carbohydrate Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Food - Raw Materials for Biosynthesis § Many metabolic pathways are involved in biosynthesis of biological molecules – Cells must be able to biosynthesize molecules not present in its foods – Cell can convert intermediate compounds of glycolysis and the citric acid cycle to molecules not found in food
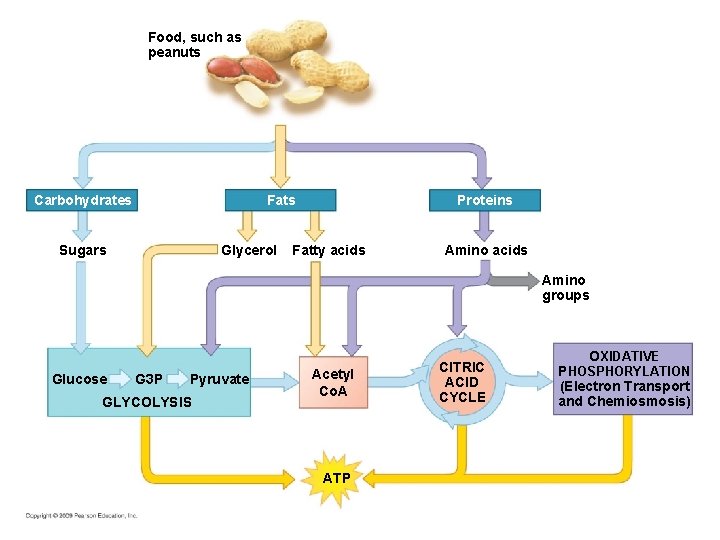
Food, such as peanuts Carbohydrates Fats Glycerol Sugars Proteins Fatty acids Amino groups Glucose G 3 P Pyruvate GLYCOLYSIS Acetyl Co. A ATP CITRIC ACID CYCLE OXIDATIVE PHOSPHORYLATION (Electron Transport and Chemiosmosis)

- Slides: 85