Chapter 6 Gases 6 1 Properties of Gases










- Slides: 11

Chapter 6 Gases 6. 1 Properties of Gases 6. 2 Gas Pressure 1

Kinetic Theory of Gases A gas consists of small particles that • move rapidly in straight lines. • have essentially no attractive (or repulsive) forces. • are very far apart. • have very small volumes compared to the volume of the container they occupy. • have kinetic energies that increase with an increase in temperature. 2

Properties That Describe a Gases are described in terms of four properties: pressure (P), volume(V), temperature(T), and amount(n). 3

Gas Pressure Gas pressure • is described as a force acting on a specific area. Pressure (P) = Force Area • Has units of atm, mm Hg, torr, lb/in. 2 and kilopascals(k. Pa). 1 atm = = 760 mm Hg (exact) 760 torr 14. 7 lb/in. 2 101. 325 k. Pa 4

Learning Check A. What is 475 mm Hg expressed in atm? 1) 475 atm 2) 0. 625 atm 3) 3. 61 x 105 atm B. The pressure in a tire is 2. 00 atm. What is this pressure in mm Hg? 1) 2. 00 mm Hg 2) 1520 mm Hg 3) 22 300 mm Hg 5

Solution A. What is 475 mm Hg expressed in atm? 2) 0. 625 atm 475 mm Hg x 1 atm = 0. 625 atm 760 mm Hg B. The pressure of a tire is measured as 2. 00 atm. What is this pressure in mm Hg? 2) 1520 mm Hg 2. 00 atm x 760 mm Hg = 1520 mm Hg 1 atm 6

Atmospheric Pressure Atmospheric pressure is the pressure exerted by a column of air from the top of the atmosphere to the surface of the Earth. 7

Altitude and Atmospheric Pressure Atmospheric pressure • is about 1 atmosphere at sea level. • depends on the altitude and the weather. • is lower at high altitudes where the density of air is less. • is higher on a rainy day than on a sunny day. 8

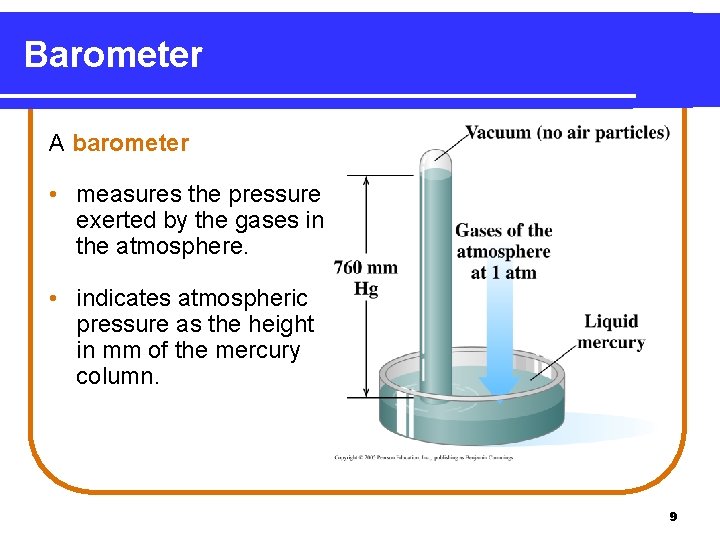
Barometer A barometer • measures the pressure exerted by the gases in the atmosphere. • indicates atmospheric pressure as the height in mm of the mercury column. 9

Learning Check A. The downward pressure of the Hg in a barometer is _____ than (as) the pressure of the atmosphere. 1) greater 2) less 3) the same B. A water barometer is 13. 6 times taller than a Hg barometer (DHg = 13. 6 g/m. L) because 1) H 2 O is less dense than mercury. 2) H 2 O is heavier than mercury. 3) air is more dense than H 2 O. 10

Solution A. The downward pressure of the Hg in a barometer is 3) the same (as) the pressure of the atmosphere. B. A water barometer is 13. 6 times taller than a Hg barometer (DHg = 13. 6 g/m. L) because 1) H 2 O is less dense than mercury. 11