Chapter 6 Equilibrium I Dynamic Equilibrium II Equilibrium

Chapter 6: Equilibrium I. Dynamic Equilibrium II. Equilibrium Constant; Kc and Kp III. Homogenous vs. Heterogeneous Equilibrium IV. Calculation of K from Equilibrium Concentrations

Equilibrium is a state in which there are no observable changes as time goes by. Chemical equilibrium is achieved when: • the rates of the forward and reverse reactions are equal and they are not zero. • the concentrations of the reactants and products remain constant Chemical equilibrium N 2 O 4 (g) 2 NO 2 (g)

Law of Mass Action • At the particular temperature, the rate of a chemical reaction is directly proportional to the product of the effective concentrations of each participating molecule. (effective concentrations of each participating molecule is the concentration of reactant or product express by the number molecular weight dissolved in one liter. a. A + b. B V 1 V 2 c. C + d. D where V 1 is rate of forward reaction, V 2 id the rate of backward reaction, and [A]a, [B]b, [C]c, [D]d are the concentration of each participating molecules. The rate of forward reaction is v 1 α [A]a. [B]b The rate of forward reaction is v 2 α [C]c, [D]d In case of equilibrium, rate of reaction forward and backward reaction are equal, V 1 = V 2, the equilibrium constant Kc for the reaction is: Kc = [C]c, [D]d [A]a. [B]b
Types of chemical equilibrium Homogenous equilibria Heterogeneous equilibria

Homogenous equilibria • Applies to reactions in which all reacting species are in the same phase. • N 2 O 4 (g) 2 NO 2 (g) Kc = [NO 2]2 [N 2 O 4] • The concentrations of reactant and products in gaseous reactions can also expressed in terms of partial pressures P, P 2 NO 2 KP = PN O 2 4 KP = Kc (0. 0821 T)Δn Where Δn = moles of product - moles of reactant, T = (o. C + 273)

Example • Write expressions for Kc and KP , if applicable for these reactions in equilibrium: • • A) 2 NO (g) + O 2 (g) 2 NO 2 (g) B) CH 3 COOH (aq) + C 2 H 5 OH (aq) • a) b) Kc = [NO 2]2 CH 3 COOC 2 H 5 (aq) + H 2 O(I) KP = [NO]2 [O 2] P 2 NO 2 P 2 NO PO 2 [CH 3 COOC 2 H 5] [H 2 O] [CH 3 COOH] [C 2 H 5 OH] [CH 3 COOC 2 H 5] [CH 3 COOH] [C 2 H 5 OH] • Write expressions for Kc and KP , if applicable for these reactions in equilibrium: • 2 N 2 O 5 (g) + 4 NO 2 (g) + O 2 (g)

Example • The equilibrium constant KP for the decomposition of PCl 5 (g) PCl 3(g) + Cl 2 (g) is found to be 1. 05 at 25 o. C. If the equilibrium partial pressure of PCl 5 and PCl 3 are 0. 875 atm and 0. 463 atm, respectively, what o is the equilibrium partial pressure of Cl 2 at 25 C? P PCl 3 2 KP = P PCl 5 1. 05 = (0. 463)(PCl ) 2 (0. 875) • The equilibrium constant KP for the decomposition of 2 NO 2 (g) 2 NO (g) + O 2 (g) is found to be 158 at 1000 K. If the equilibrium partial pressure of NO 2 and NO are 0. 400 atm and 0. 270 atm, orespectively, what is the equilibrium partial pressure of O 2 at 25 C?

Example • For the reaction N 2 (g) + H 2(g) 2 NH (g) KP is 4. 3 X 10 -4 at 375 o. C. Calculate Kc for the reaction? K c= KP (0. 0821 T)Δn Because T = 375 + 273 = 648 K Δn = 2 – 4 = _ 2 Kc = 4. 3 X 10 -4 (0. 821 X 648)-2

Heterogeneous equilibria • Applies to reactions in which all reacting species are in the different phase. • Ca. CO 3 (s) Ca. O (s) + 2 CO 2 (g) Kc = [Ca. O]. [CO 2] [Ca. CO 3] • The concentrations of solid, like its density, does not depend on how much of the substance is present, so Kc =[CO 2] KP = PCO 2
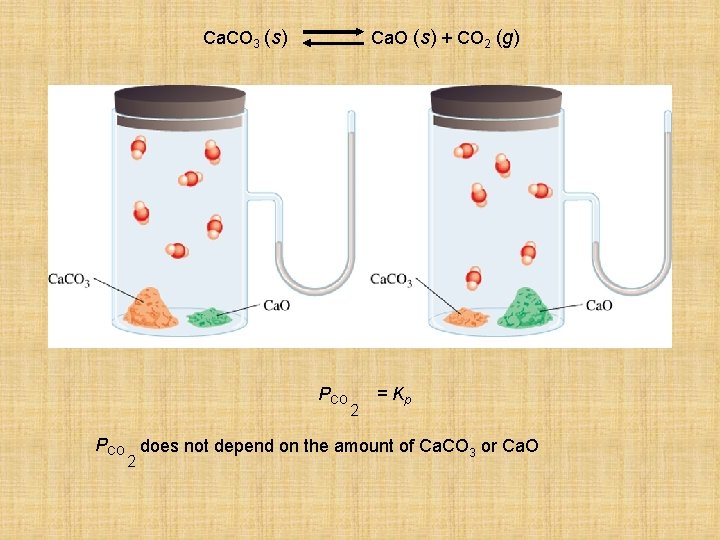
Ca. CO 3 (s) Ca. O (s) + CO 2 (g) PCO 2 = Kp PCO does not depend on the amount of Ca. CO 3 or Ca. O 2

LE CHATELIER'S PRINCIPLE If a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium moves to counteract the change.
Factors affecting Chemical Equilibrium Reactions Pressure Heat Concentrations

Effect of Pressure • This only applies to reactions involving gases in reactants or products: N 2 + 3 H 2 1 molecules 3 molecules 2 NH 3 + heat (24 k. Cal) 2 molecules • What would happen if you changed the conditions by increasing the pressure? • • According to Le Chatelier, the position of equilibrium will move in such a way as to counteract the change. That means that the position of equilibrium will move so that the pressure is reduced again. Pressure is caused by gas molecules hitting the sides of their container. The more molecules you have in the container, the higher the pressure will be. The system can reduce the pressure by reacting in such a way as to produce fewer molecules. In this case, there are 3 molecules on the left-hand side of the equation, but only 2 on the right. By forming more 2 molecules, the system causes the pressure to reduce. Increasing the pressure on a gas reaction shifts the position of equilibrium towards the side with fewer molecules.

• What would happen if you changed the conditions by decreasing the pressure? • The equilibrium will move in such a way that the pressure increases again. It can do that by producing more molecules. In this case, the position of equilibrium will move towards the left-hand side of the reaction.

• What happens if there are the same number of molecules on both sides of the equilibrium reaction? • In this case, increasing the pressure has no effect whatsoever on the position of the equilibrium. Because you have the same numbers of molecules on both sides, the equilibrium can't move in any way that will reduce the pressure again.

Effect of Heat (Temperature) • For most chemical reactions, by increases the temperature the rate of reaction increases, but in case of chemical equilibrium reactions, it depends on the type of chemical reaction whether it is exothermic reactions (heat evolved), or endothermic reactions (heat absorbed). I 2 + H 2 2 HI + heat (Δ E = _ 12. 8 K. Cal. )

• What would happen if you changed the conditions by increasing the temperature? • According to Le Chatelier, the position of equilibrium will move in such a way as to counteract the change. That means that the position of equilibrium will move so that the temperature is reduced again. • Suppose the system is in equilibrium at 300°C, and you increase the temperature to 500°C. How can the reaction counteract the change you have made? How can it cool itself down again? • To cool down, it needs to absorb the extra heat that you have just put in. In the case we are looking at, the back reaction absorbs heat. The position of equilibrium therefore moves to the left. The new equilibrium mixture contains more A and B, and less C and D.

• What would happen if you changed the conditions by decreasing the temperature? • The equilibrium will move in such a way that the temperature increases again. • Suppose the system is in equilibrium at 500°C and you reduce the temperature to 400°C. The reaction will tend to heat itself up again to return to the original temperature. It can do that by favoring the exothermic reaction. • The position of equilibrium will move to the right. More A and B are converted into C and D at the lower temperature.

Effect of Concentrations • What would happen if you changed the conditions by increasing the concentration of A? • According to Le Chatelier, the position of equilibrium will move in such a way as to counteract the change. That means that the position of equilibrium will move so that the concentration of A decreases again - by reacting it with B and turning it into C + D. The position of equilibrium moves to the right.

• What would happen if you changed the conditions by decreasing the concentration of A? • According to Le Chatelier, the position of equilibrium will move so that the concentration of A increases again. That means that more C and D will react to replace the A that has been removed. The position of equilibrium moves to the left.
- Slides: 20