Chapter 6 Chemistry in Biology Section 1 Atoms


Chapter 6 Chemistry in Biology Section 1: Atoms, Elements, and Compounds Section 2: Chemical Reactions Section 3: Water and Solutions Section 4: The Building Blocks of Life Click on a lesson name to select.

Objective: • Identify the particles that makeup atoms. Diagram the particles that make up an atom. Click on a lesson name to select.

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Atoms § Chemistry is the study of matter. § Matter is anything that has mass and occupies space. § Atoms are the building blocks of matter. § Neutrons and protons are located at the center of the atom in the nucleus. § Protons are positively charged particles. § Neutrons are particles that have no charge.

Name Location Charge Proton Nucleus Positive Neutron Nucleus Neutral Electron Energy levels negative

• The first energy level holds 2 electrons. • 2 nd energy level holds 8 electrons
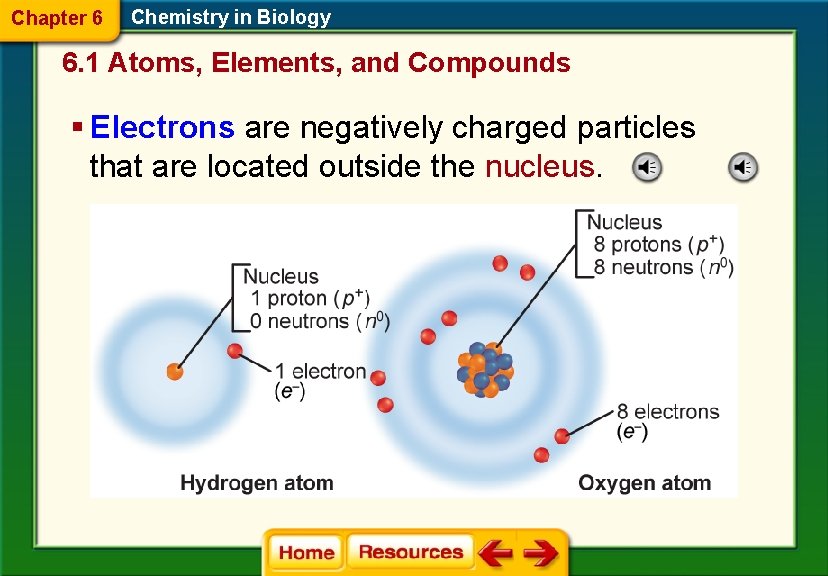
Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds § Electrons are negatively charged particles that are located outside the nucleus.

• The number of neutrons is calculated by subtracting the atomic number from the atomic mass of the element. • Ex. Carbon has the atomic number of 6 and an atomic mass of 12. • Therefore, it has 6 neutrons (12 -6=6).

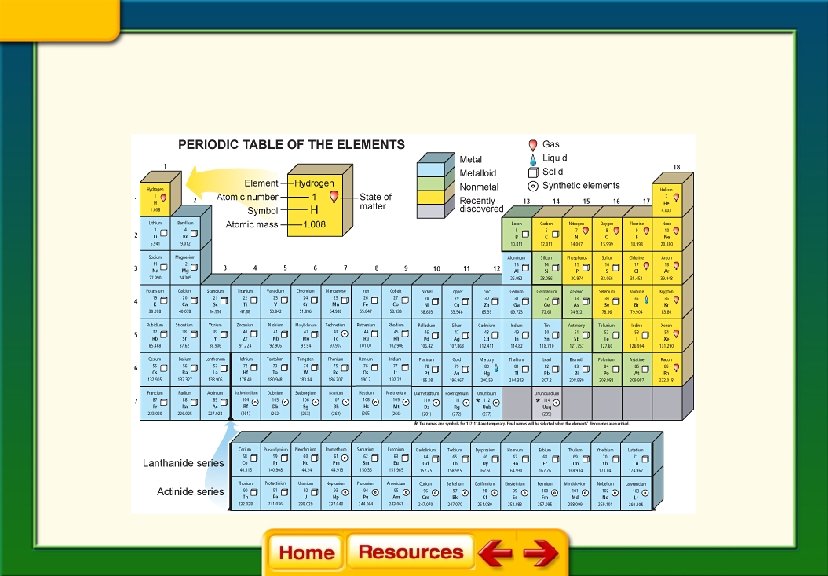
Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Elements § An element is a pure substance that cannot be broken down into other substances by physical or chemical means. § There are over 100 known elements, 92 of which occur naturally. § Each element has a unique name and symbol.

• The elements are arranged on the periodic table by their atomic number which is the number of protons for that element.

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds The Periodic Table of Elements § Horizontal rows are called periods. § Vertical columns are called groups. § Elements in a group have similar properties.
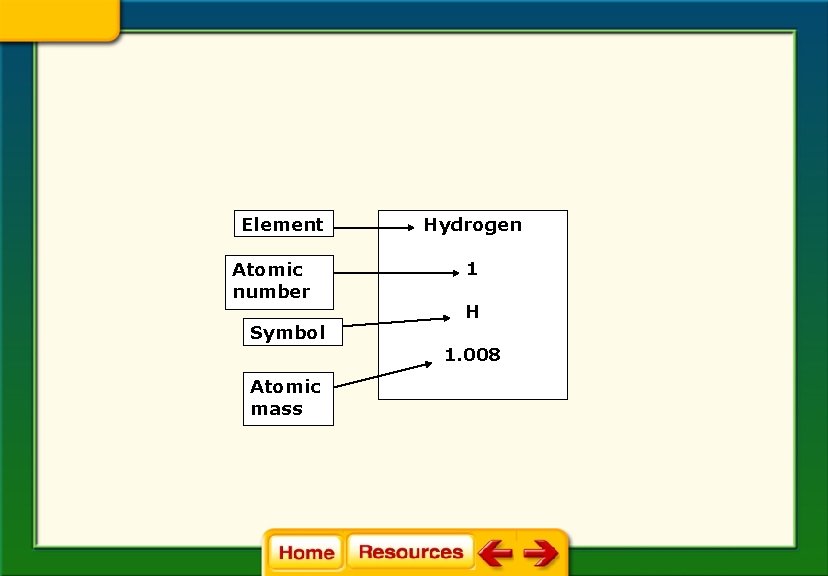
Element Atomic number Symbol Atomic mass Hydrogen 1 H 1. 008

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Isotopes § Isotopes are atoms of the same element that have the same number of protons and electrons but have a different number of neutrons.
Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Radioactive Isotopes § Radioisotopes give off radiation when the nucleus splits. This radiation can be detected and used for many applications.
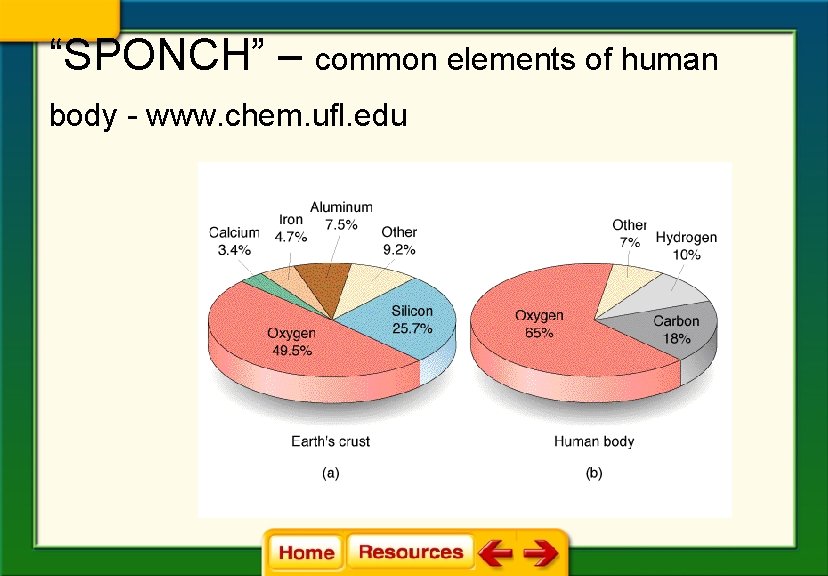
“SPONCH” – common elements of human body - www. chem. ufl. edu

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds § A compound is formed when two or more different elements combine. § The combination of elements will be in a fixed ratio. Ex. water – H 2 O § Compounds cannot be broken down into simpler compounds or elements by physical means.

Objective • Compare and contrast covalent bonds and ionic bonds.

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Chemical Bonds § Covalent bonds form when electrons are shared. A molecule is formed. A molecule is a compound in which the atoms are held together by covalent bonds.

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Ionic Bonds § Ionic bonds form when two oppositely charged atoms are attracted to each other. Electrons are transferred.

• Metals donate electrons. • Nonmetals accept electrons. • An ion is an atom that has lost or gained one or more electrons.
Chapter 6 Chemistry in Biology

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds § Most ionic compounds are crystalline at room temperature and have higher melting points than molecular compounds formed by covalent bonds. § Ionic liquids are more environmentally friendly.

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds van der Waals Forces § van der Waals forces are attractive forces between molecules. When molecules come close together, the attractive forces between slightly positive and negative regions pull on the molecules and hold them together. § The strength of the attraction depends on the size of the molecule, its shape, and its ability to attract electrons.

Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions Reactants and Products § A chemical reaction is the process by which atoms or groups of atoms in substances are reorganized into different substances by breaking or forming chemical bonds. Ex. rust § Clues that a chemical reaction has taken place include the production of heat or light, and formation of a gas, liquid, or solid. Chemical reaction Physical reaction

• A physical change alters the substance’s appearance not composition. • A chemical change involves a change in composition of a substance.

Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions Chemical Equations § Chemical formulas describe the substances in the reaction and arrows indicate the process of change. § Reactants are the starting substances, on the left side of the arrow. § Products are the substances formed during the reaction, on the right side of the arrow. § The arrow means yields.

Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions § Glucose and oxygen react to form carbon dioxide and water.

Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions Balanced Equations § The law of conservation of mass states matter cannot be created or destroyed. § All chemical reactions must balance. The number of atoms of each element on the reactant side must equal the number of atoms of the same element on the product side.

• The coefficient is the number in front of the reactant or product. • The subscript is the number of atoms. • The coefficient times the subscript equals the total number of atoms.
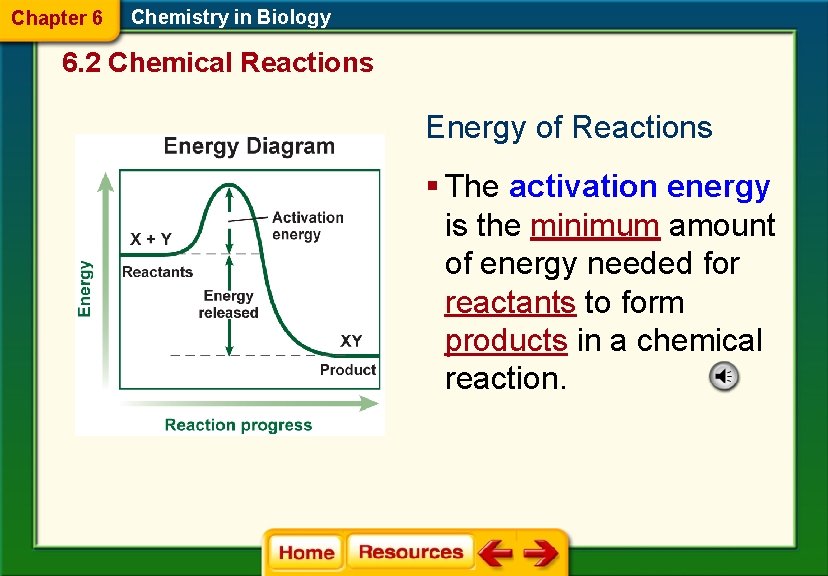
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions Energy of Reactions § The activation energy is the minimum amount of energy needed for reactants to form products in a chemical reaction.
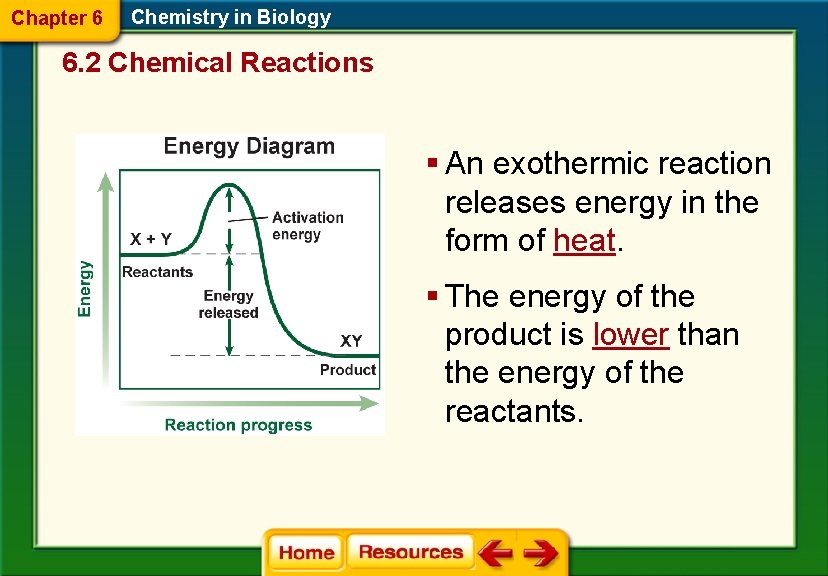
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions § An exothermic reaction releases energy in the form of heat. § The energy of the product is lower than the energy of the reactants.
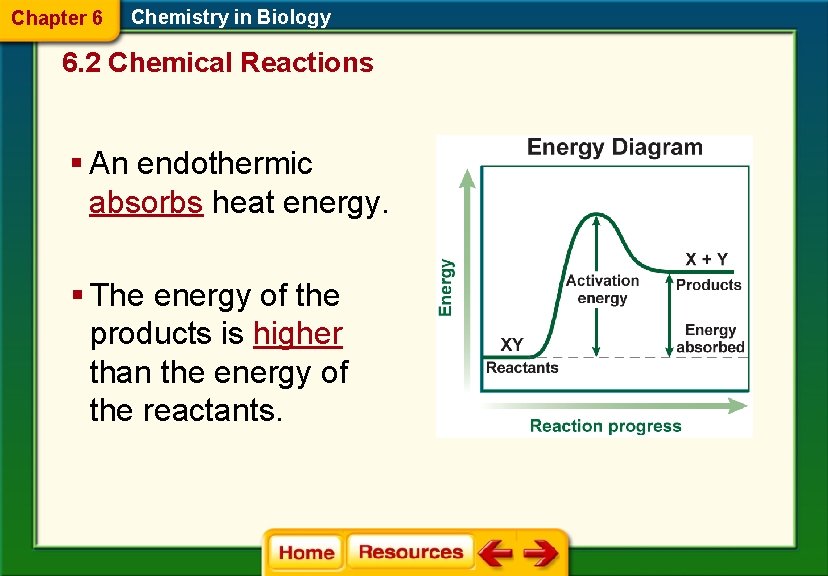
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions § An endothermic absorbs heat energy. § The energy of the products is higher than the energy of the reactants.
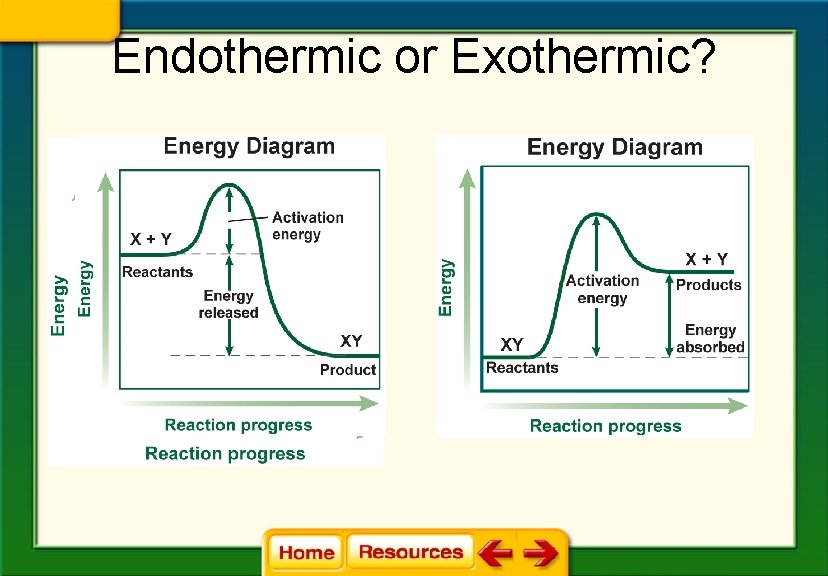
Endothermic or Exothermic?
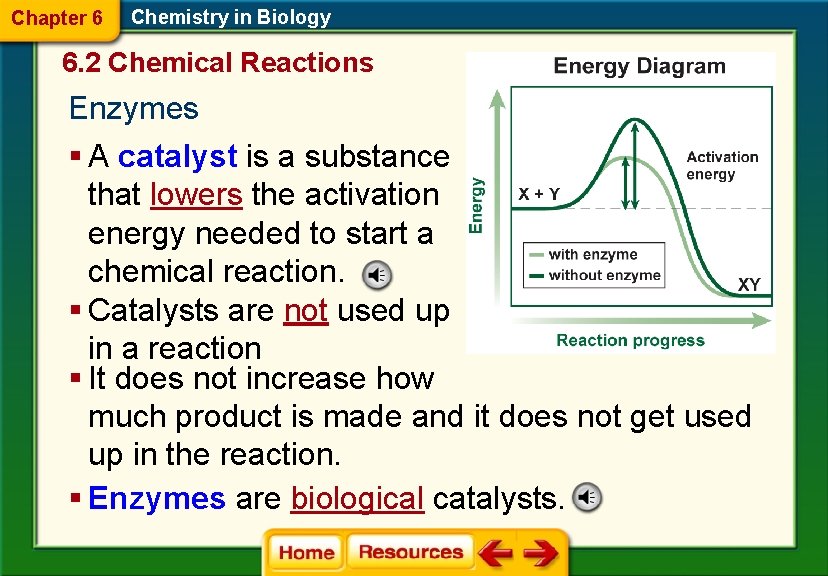
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions Enzymes § A catalyst is a substance that lowers the activation energy needed to start a chemical reaction. § Catalysts are not used up in a reaction § It does not increase how much product is made and it does not get used up in the reaction. § Enzymes are biological catalysts.
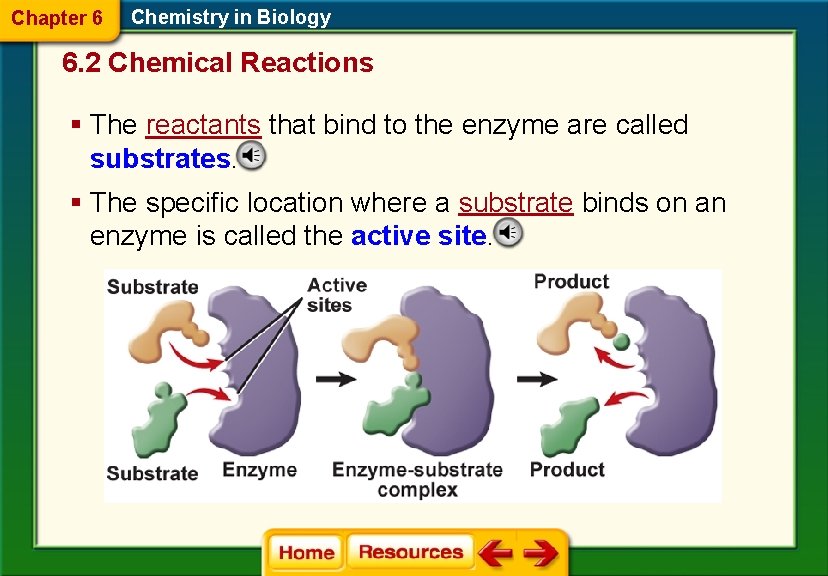
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions § The reactants that bind to the enzyme are called substrates. § The specific location where a substrate binds on an enzyme is called the active site.

Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions § The active site changes shape and forms the enzyme-substrate complex, which helps chemical bonds in the reactants to be broken and new bonds to form. § Factors such as p. H, temperature, and other substances affect enzyme activity.
Chapter 6 Chemistry in Biology

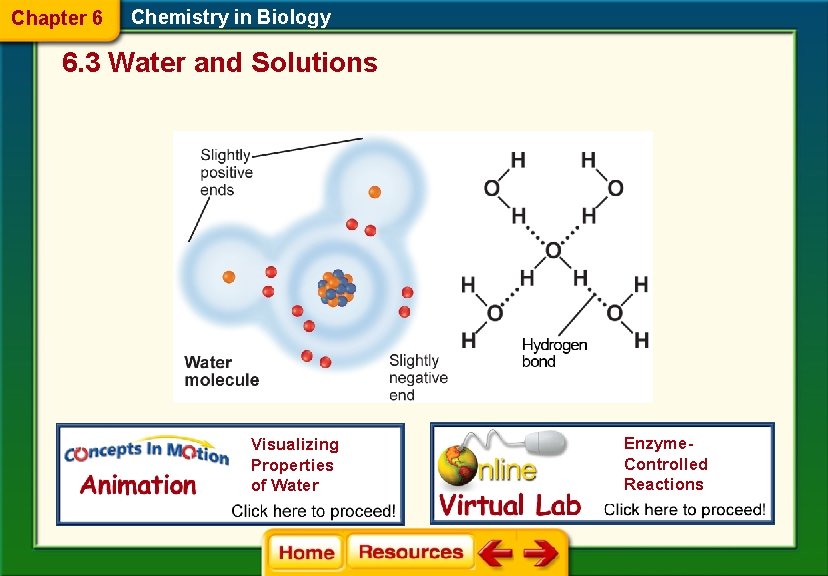
Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Water’s Polarity § Molecules that have an unequal distribution of charges are called polar molecules. § Polarity is the property of having two opposite poles. Water is a polar compound. § A hydrogen bond is a weak interaction involving a hydrogen atom and a fluorine, oxygen, or nitrogen atom. (#3)
Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Visualizing Properties of Water Enzyme. Controlled Reactions

Mixtures • A mixture is a combination of two or more substances in which each substance retains its individual characteristics. • There are two types: – Heterogeneous mixtures – Homogeneous mixtures

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Homogenous Mixtures § A homogenous mixture has a uniform composition throughout the mixture. Ex. food coloring in water § Solvent + solute = solution § A solvent is a substance in which another substance is dissolved. § A solute is the substance that is dissolved in the solvent. #10 Food coloring dissolved in water forms a homogenous mixture.

• Water is the universal solvent because many substances dissolve in it. (#7) • Water is adhesive (sticks to surfaces) which allows it to travel up the stem of a plant. (#8) • Water is cohesive (molecules stick together) which causes water to form droplets. (#9) § A solute is the substance that is dissolved in the solvent. (#10)

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Heterogeneous Mixtures § In a heterogeneous mixture, the components remain distinct. Ex. _____ A salad is a heterogeneous mixture.

• A suspension is a heterogeneous mixture in which the particles settle out over time. • Ex. sand water

• A colloid is a heterogeneous mixture in which the particles do not settle out over time. • Ex. milk, mayonnaise, paint, blood.

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Acids and Bases § Substances that release hydrogen ions (H+) when dissolved in water are called acids. § Substances that release hydroxide ions (OH–) when dissolved in water are called bases.

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions p. H and Buffers § The measure of concentration of H+ in a solution is called p. H. § Acidic solutions have p. H values lower than 7. § Basic solutions have p. H values higher than 7.

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions § Buffers are mixtures that can react with acids or bases to keep the p. H within a particular range.

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Organic Chemistry § The element carbon is a component of almost all biological molecules.

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life § Carbon has four electrons in its outermost energy level. § One carbon atom can form four covalent bonds with other atoms. This enables carbon atoms to bond to each other which results in a variety of important organic compounds. (#2) § Carbon compounds can be in the shape of straight chains, branched chains, and rings.

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Macromolecules, also called polymers § Carbon atoms can be joined to form carbon molecules. § Macromolecules are large molecules formed by joining smaller organic molecules together. § Polymers are molecules made from monomers (repeating units of identical or nearly identical compounds linked together by a series of covalent bonds). (#4)

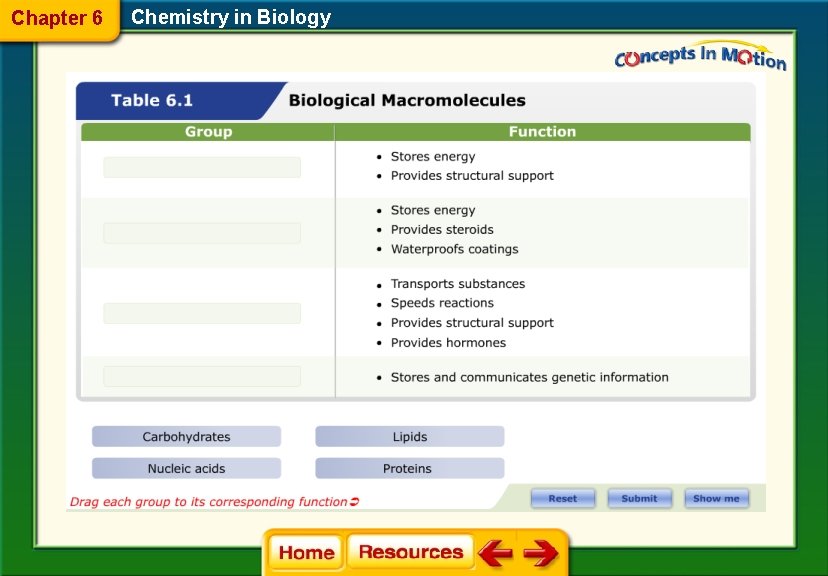
• The four major categories of macromolecules are carbohydrates, lipids, proteins, and nucleic acids.

Chapter 6 Chemistry in Biology
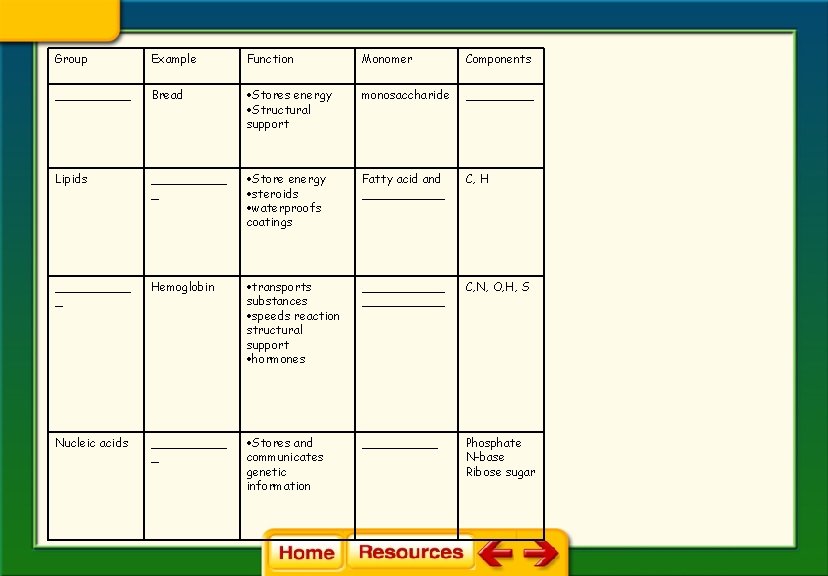
Group Example Function Monomer Components _____ Bread Stores energy Structural support monosaccharide _____ Lipids _____ _ Store energy steroids waterproofs coatings Fatty acid and ______ C, H _____ _ Hemoglobin transports substances speeds reaction structural support hormones ___________ C, N, O, H, S Nucleic acids _____ _ Stores and communicates genetic information _____ Phosphate N-base Ribose sugar

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Carbohydrates § Compounds composed of carbon, hydrogen, and oxygen in a ratio of one oxygen and two hydrogen atoms for each carbon atom—(CH 2 O)n

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life § Values of n ranging from three to seven are called simple sugars, or monosaccharides. § Two monosaccharides joined together form a disaccharide. § Longer carbohydrate molecules are called polysaccharides.

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Lipids § Molecules made mostly of carbon and hydrogen § A triglyceride is a fat if it is solid at room temperature and an oil if it is liquid at room temperature.
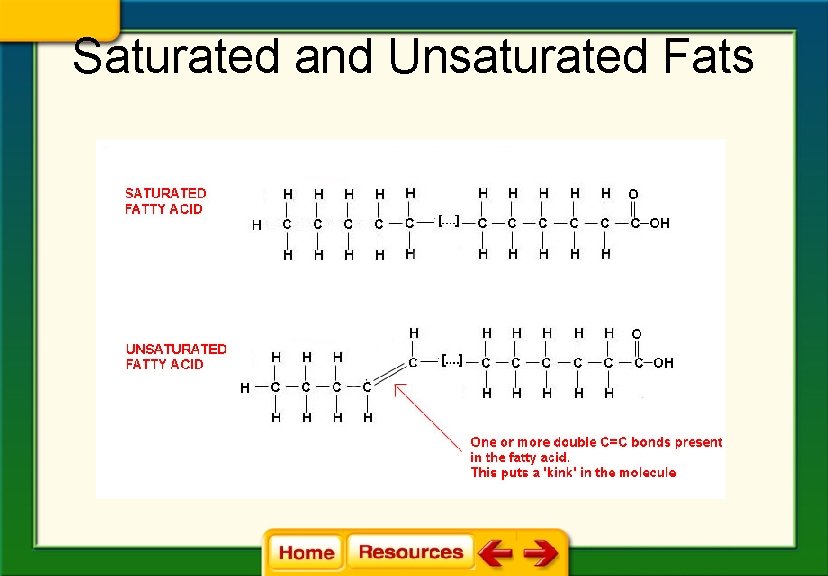
Saturated and Unsaturated Fats

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life § Saturated fats (lipids) have tail chains made up of only single bonds between the carbon atoms. § Unsaturated fats have tail chains with at least one double bond between the carbon atoms. § Fats with more than one double bond in the tail are called polyunsaturated fats.

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Proteins § Proteins are made of amino acids. § Amino acids are small compounds that are made of carbon, nitrogen, oxygen, hydrogen, and sometimes sulfur.

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life § Amino acids have a central carbon atom. § One of the four carbon bonds is with hydrogen. § The other three bonds are with an amino group (–NH 2), a carboxyl group (–COOH), and a variable group (–R).
Chapter 6 Chemistry in Biology
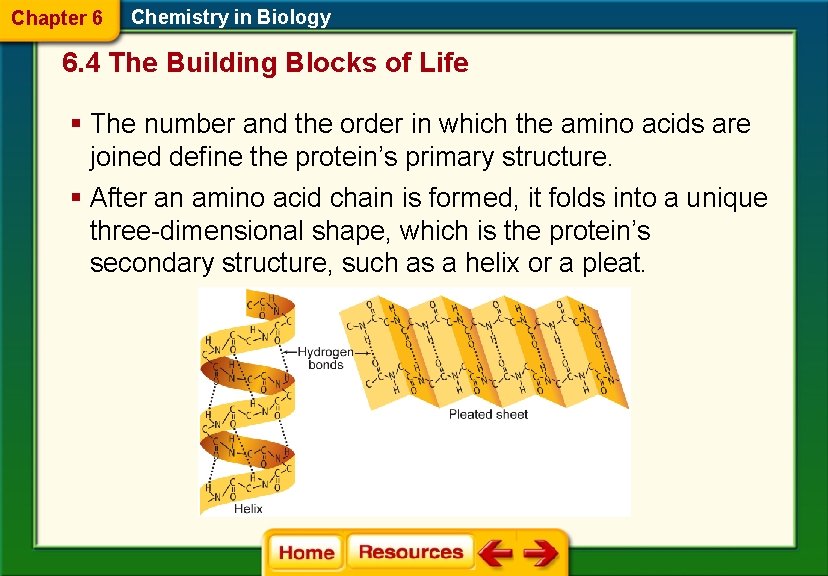
Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life § The number and the order in which the amino acids are joined define the protein’s primary structure. § After an amino acid chain is formed, it folds into a unique three-dimensional shape, which is the protein’s secondary structure, such as a helix or a pleat.
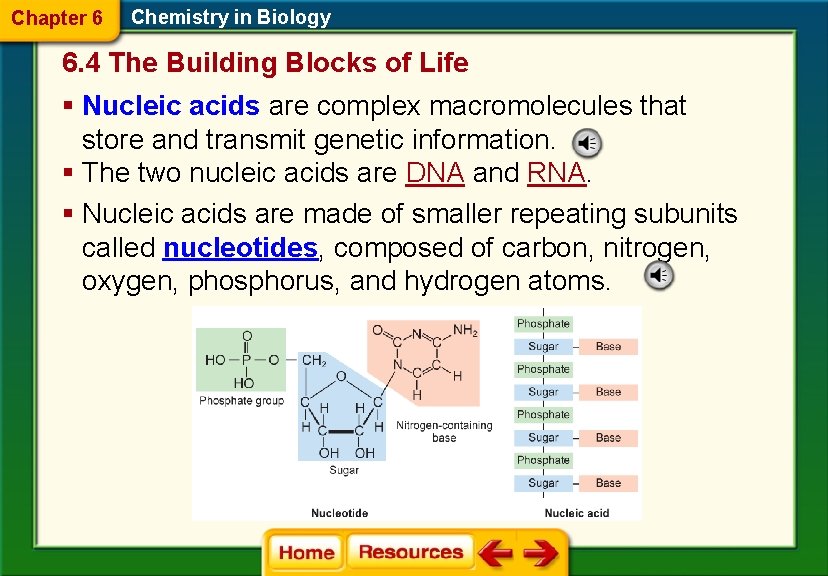
Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life § Nucleic acids are complex macromolecules that store and transmit genetic information. § The two nucleic acids are DNA and RNA. § Nucleic acids are made of smaller repeating subunits called nucleotides, composed of carbon, nitrogen, oxygen, phosphorus, and hydrogen atoms.
Chapter 6 Chemistry in Biology Chapter Resource Menu Chapter Diagnostic Questions Formative Test Questions Chapter Assessment Questions Standardized Test Practice biologygmh. com Glencoe Biology Transparencies Image Bank Vocabulary Animation Click on a hyperlink to view the corresponding lesson.
Chapter 6 Chemistry in Biology Chapter 6 Diagnostic Questions (1 -31) 1. Which of the following particles is 2. negatively charged? A. electron B. isotope C. neutron D. proton
Chapter 6 Chemistry in Biology Chapter 6 Diagnostic Questions 2. Isotopes are created by a change in the number of what particle of an atom? A. electrons B. neutrons C. protons D. ions

Chapter 6 Chemistry in Biology Chapter 6 Diagnostic Questions 3. Identify the proteins that speed up the rate of chemical reactions. A. substrates B. enzymes C. ions D. reactants

Chapter 6 Chemistry in Biology 6. 1 Formative Questions 4. What particles are in an atom’s nucleus? A. neutrons and electrons B. protons and electrons C. protons and neutrons D. protons, neutrons and electrons

Chapter 6 Chemistry in Biology 6. 1 Formative Questions 5. What causes the overall charge of an atom to be zero? A. an equal number of protons and neutrons B. an equal number of protons and electrons C. an equal number of neutrons and electrons D. an unequal number of protons and electrons

Chapter 6 Chemistry in Biology 6. 1 Formative Questions 6. What type of substance is water? A. a compound B. an element C. an isotope D. a mixture

Chapter 6 Chemistry in Biology 6. 1 Formative Questions 7. What provides the energy for all living processes? A. chemical bonds B. ionic compounds C. radioactive isotopes D. van der Waals forces
Chapter 6 Chemistry in Biology 6. 2 Formative Questions 8. Which is a chemical reaction? A. a match burning B. salt dissolving C. water boiling D. gasoline evaporating
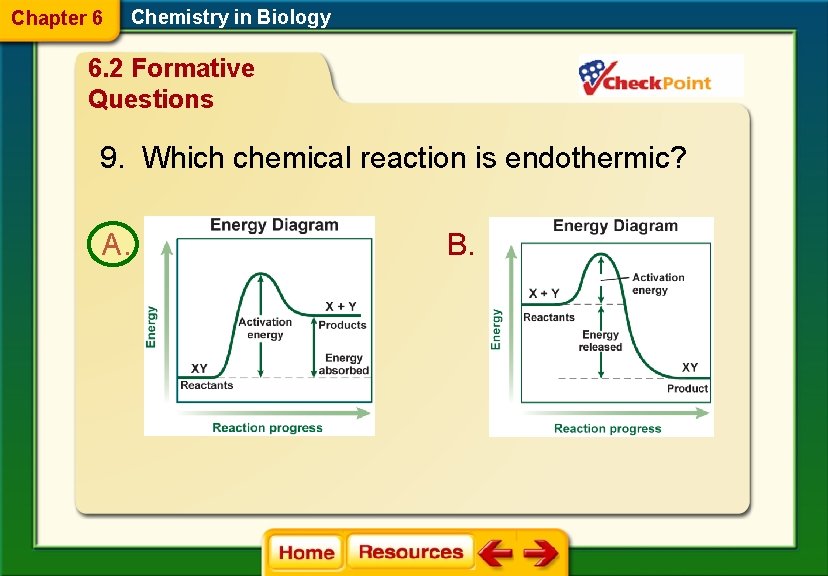
Chapter 6 Chemistry in Biology 6. 2 Formative Questions 9. Which chemical reaction is endothermic? A. B.

Chapter 6 Chemistry in Biology 6. 2 Formative Questions 10. How does an enzyme increase the rate of a chemical reaction? A. It acts as a reactant. B. It reduces the amount of heat produced. C. It increases the amount of product. D. It lowers the activation energy.
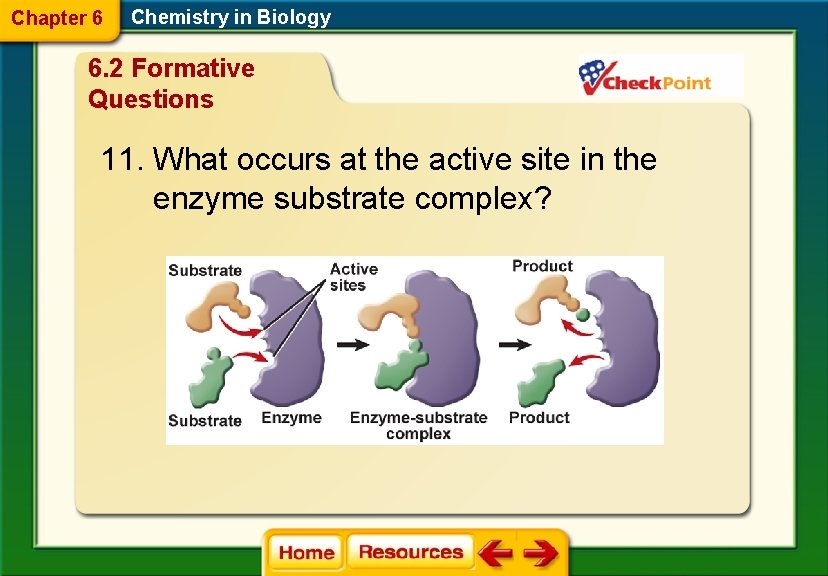
Chapter 6 Chemistry in Biology 6. 2 Formative Questions 11. What occurs at the active site in the enzyme substrate complex?

Chapter 6 Chemistry in Biology 6. 2 Formative Questions A. An exothermic chemical reaction takes place. B. Chemical bonds are broken and new bonds are formed. C. The enzyme gets used up in the reaction. D. The substrates provide energy for the enzyme.

Chapter 6 Chemistry in Biology 6. 3 Formative Questions 12. Why is water able to dissolve a wide variety of solutes? A. It acts as a catalyst. B. Its p. H is neutral. C. It is a polar molecule. D. It is an ionic compound.

Chapter 6 Chemistry in Biology 6. 3 Formative Questions 13. What type of bonds attracts water molecules to each other and to other substances? A. covalent bonds B. double bonds C. hydrogen bonds D. ionic bonds

Chapter 6 Chemistry in Biology 6. 3 Formative Questions 14. Which ion, when released in water, causes a solution to be basic? A. Cl– B. OH– C. H+ D. Na+
Chapter 6 Chemistry in Biology 6. 3 Formative Questions 15. What is the name for a substance that keeps the p. H in cells within the 6. 5 to 7. 5 p. H range? A. alkali B. antacid C. buffer D. neutralizer
Chapter 6 Chemistry in Biology 6. 4 Formative Questions 16. Which element do almost all biological molecules contain? A. carbon B. nitrogen C. phosphorus D. sodium

Chapter 6 Chemistry in Biology 6. 4 Formative Questions 17. How many covalent bonds can carbon 18. form with other atoms? A. 1 B. 2 C. 4 D. 8
Chapter 6 Chemistry in Biology 6. 4 Formative Questions 18. What type of biological molecule is an enzyme? A. hormone B. nucleic acid C. protein D. steroid
Chapter 6 Chemistry in Biology 6. 4 Formative Questions 19. What are fats, oils, and waxes composed of? A. lipids B. nucleotides C. polypeptides D. sugars

Chapter 6 Chemistry in Biology 6. 4 Formative Questions 20. Which biological molecule transports substances between cells? A. carbohydrate B. lipid C. nucleic acid D. protein
Chapter 6 Chemistry in Biology 6. 4 Formative Questions 21. What are the monomers that make up proteins? A. amino acids B. fatty acids C. glycerols D. nucleotides
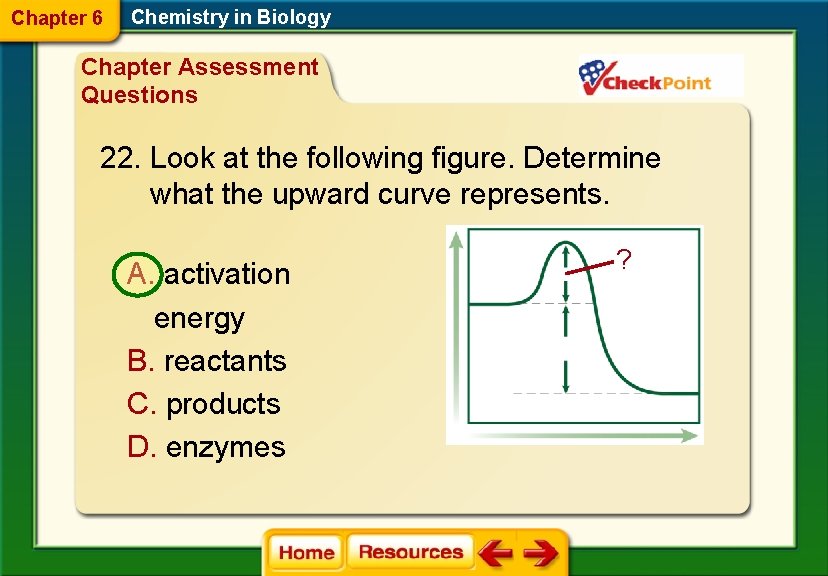
Chapter 6 Chemistry in Biology Chapter Assessment Questions 22. Look at the following figure. Determine what the upward curve represents. A. activation energy B. reactants C. products D. enzymes ?

Chapter 6 Chemistry in Biology Chapter Assessment Questions 23. Look at the energy levels in the atom. What is the maximum number of electrons energy level two can hold? A. 2 B. 4 C. 6 D. 8

Chapter 6 Chemistry in Biology Chapter Assessment Questions 24. Explain why chemical equations must be balanced. Short answer. Answer: Chemical reactions require balance of mass. Therefore, the number of atoms of each element on the reactant side must equal the number of atoms of the same element on the product side.

Chapter 6 Chemistry in Biology Standardized Test Practice 25. Which is a result of van der Waals forces? A. atoms sharing electrons B. table salt dissolving in water C. ionic compounds forming crystals D. water molecules forming droplets
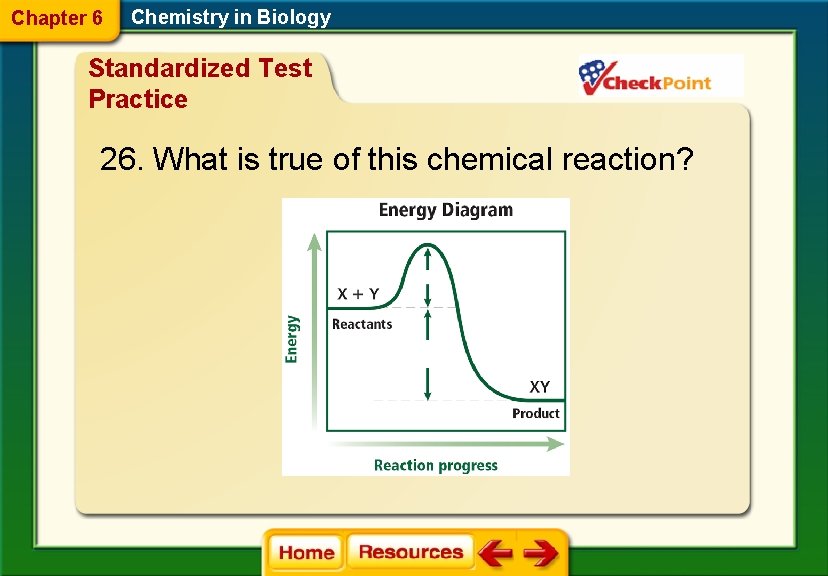
Chapter 6 Chemistry in Biology Standardized Test Practice 26. What is true of this chemical reaction?

Chapter 6 Chemistry in Biology Standardized Test Practice A. Energy is not needed to start the chemical reaction. B. Heat and/or light energy are released in this reaction. C. The activation energy is greater than the energy released. D. The energy of the products and the reactants is the same.

Chapter 6 Chemistry in Biology Standardized Test Practice 27. Which fruit contains a higher concentration of hydrogen ions? A. tomatoes B. bananas

Chapter 6 Chemistry in Biology Standardized Test Practice 28. What do cellulose and chitin have in common? A. They are energy-storing polymers. B. They are found in the cells of animals. C. They are structural polysaccharides. D. They are composed of repeating sucrose units.
Chapter 6 Chemistry in Biology Standardized Test Practice 29. Which polysaccharide stores energy in muscle and liver tissue? A. gluten B. glycogen C. starch D. sucrolose

Chemistry in Biology Chapter 6 Standardized Test Practice 30. What is the function of this biological macromolecule? A. B. C. D. communicate signals between cells produce vitamins and hormones provide support and protection store and transmit genetic information

Chapter 6 Chemistry in Biology Standardized Test Practice 31. Which is a characteristic of all lipids? A. They are saturated triglycerides. B. They do not dissolve in water. C. They are liquid at room temperature. D. They store less energy than carbohydrates.

Chapter 6 Chemistry in Biology Glencoe Biology Transparencies
Chapter 6 Chemistry in Biology Image Bank
Chapter 6 Chemistry in Biology Image Bank
Chapter 6 Chemistry in Biology Vocabulary Section 1 atom compound nucleus proton neutron electron element isotope covalent bond molecule ionic bond van der Waals force
Chapter 6 Chemistry in Biology Vocabulary Section 2 chemical reaction substrate reactant product activation energy catalyst enzyme active site
Chapter 6 Chemistry in Biology Vocabulary Section 3 polar molecule base hydrogen bond mixture solution solvent solute acid p. H buffer
Chapter 6 Chemistry in Biology Vocabulary Section 4 macromolecule nucleic acid polymer carbohydrate lipid protein amino acid nucleotide

Chapter 6 Chemistry in Biology Animation § Ionic Bonds § Enzymes § Visualizing Properties of Water § Polypeptides
- Slides: 109