Chapter 6 Chemistry in Biology Section 1 Atoms


Chapter 6 Chemistry in Biology Section 1: Atoms, Elements, and Compounds Section 2: Chemical Reactions Section 3: Water and Solutions Section 4: The Building Blocks of Life Click on a lesson name to select.

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Water’s Polarity § Molecules that have an unequal distribution of charges are called polar molecules. § Polarity is the property of having two opposite poles. § A hydrogen bond is a weak interaction involving a hydrogen atom and a fluorine, oxygen, or nitrogen atom.
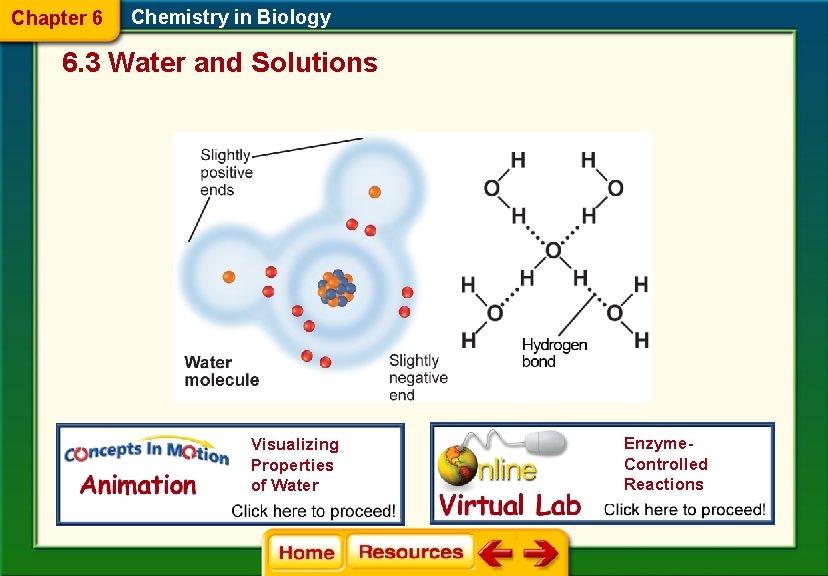
Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Visualizing Properties of Water Enzyme. Controlled Reactions

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Homogenous Mixtures § A mixture that has a uniform composition throughout § A solvent is a substance in which another substance is dissolved. § A solute is the substance that is dissolved in the solvent. Food coloring dissolved in water forms a homogenous mixture.

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Heterogeneous Mixtures § In a heterogeneous mixture, the components remain distinct. A salad is a heterogeneous mixture.

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions Acids and Bases § Substances that release hydrogen ions (H+) when dissolved in water are called acids. § Substances that release hydroxide ions (OH–) when dissolved in water are called bases.

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions p. H and Buffers § The measure of concentration of H+ in a solution is called p. H. § Acidic solutions have p. H values lower than 7. § Basic solutions have p. H values higher than 7.

Chapter 6 Chemistry in Biology 6. 3 Water and Solutions § Buffers are mixtures that can react with acids or bases to keep the p. H within a particular range.

Chapter 6 Chemistry in Biology 6. 3 Formative Questions Why is water able to dissolve a wide variety of solutes? A. It acts as a catalyst. B. Its p. H is neutral. C. It is a polar molecule. D. It is an ionic compound.

Chapter 6 Chemistry in Biology 6. 3 Formative Questions What type of bonds attracts water molecules to each other and to other substances? A. covalent bonds B. double bonds C. hydrogen bonds D. ionic bonds

Chapter 6 Chemistry in Biology 6. 3 Formative Questions Which ion, when released in water, causes a solution to be basic? A. Cl– B. OH– C. H+ D. Na+
Chapter 6 Chemistry in Biology 6. 3 Formative Questions What is the name for a substance that keeps the p. H in cells within the 6. 5 to 7. 5 p. H range? A. alkali B. antacid C. buffer D. neutralizer

Chapter 6 Chemistry in Biology Standardized Test Practice Which fruit contains a higher concentration of hydrogen ions? A. tomatoes B. bananas
Chapter 6 Chemistry in Biology Vocabulary Section 3 polar molecule base hydrogen bond mixture solution solvent solute acid p. H buffer
- Slides: 15