Chapter 6 Chemical Reactions Section 8 Oxidation Reduction



















- Slides: 20

Chapter 6: Chemical Reactions Section 8: Oxidation. Reduction Reactions

Learning Goals �Identify redox reactions. �Identify and write equations for combustion reactions.

Oxidation-Reduction Reactions �A reaction can be classified as a redox reaction if it meets any one of these requirements. A substance reacts with elemental oxygen (combustion). A metal reacts with a nonmetal. During single displacement reactions. More generally, one substance transfers electrons to another substance.

Oxidation-Reduction Reactions � 2 Na(s) + Cl 2(g) 2 Na. Cl(s) Na + Cl e- + Na + Cl

Oxidation-Reduction Reactions �Combustion reactions are a type of redox reaction. �Combustion reactions are characterized by the reaction of a substance with O 2 to form one or more oxygen-containing compounds, often including water.

Oxidation-Reduction Reactions �Combustion reactions are exothermic (they emit heat). �Compounds containing carbon and hydrogen always form carbon dioxide and water upon combustion.

Example � CH 4(g) + O 2(g)

Oxidation-Reduction Reactions � 2 Mg(s) + O 2(g) 2 Mg. O(s) Mg + O e- e- 2+ Mg + 2 O ! ! N O I T C A E R S I S E H T N Y S A O S L A

Oxidation-Reduction Reactions � 2 Al(s) + Fe 2 O 3(s) 2 Fe(s) + Al 2 O 3(s) Al + Fe 3+ Fe + Al 3+ ee- -e. L G N I S A O ALS ! ! N O I T C A E R T N E M E E REPLAC

Practice � For each reaction, show electrons are gained and lost: 2 Na (s) + Br 2(l) 2 Na. Br (s) 2 Ca (s) + O 2(g) 2 Ca. O (s)

Practice �Predict the products for the following combustion reactions: C 6 H 14(l) + O 2(g) Cu(s) + O 2(g)

Chapter Review

Review � Classify each of the following reactions in as many ways possible. 2 K (s) + Cl 2(g) 2 KCl (s) 2 Mg (s) + O 2(g) 2 Mg. O (s)

Review � Classify each of the following reactions in as many ways possible. Fe 2 O 3(s) + 2 Al(s) Al 2 O 3(s) + 2 Fe(s) HNO 3(aq) + Na. OH(aq) → H 2 O(l) + Na. NO 3 (aq)

Review �Ba(NO 3)2(aq) + (NH 4)2 SO 4(aq) �HCl(aq) + KOH(aq)

Review �Na(s) + Cl 2(g) �Fe. Cl 3(aq) + Na. OH(aq)

Review �Zn(s) + HNO 3(aq) �C 6 H 14(l) + O 2(g)

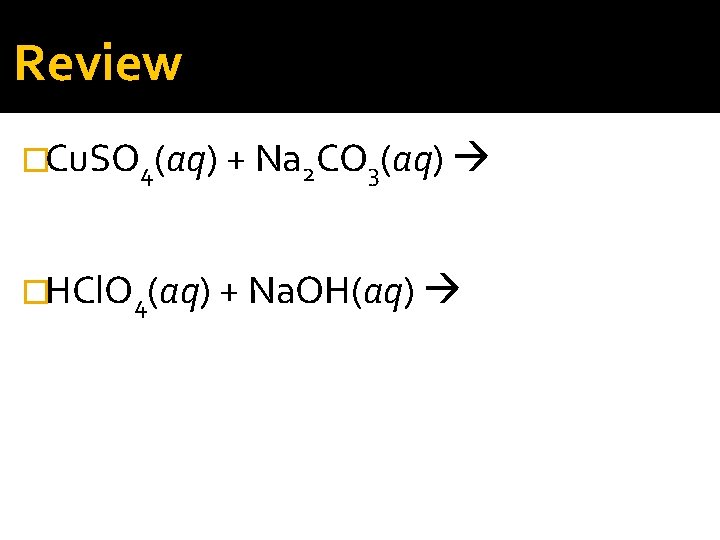
Review �Cu. SO 4(aq) + Na 2 CO 3(aq) �HCl. O 4(aq) + Na. OH(aq)

Review �NI 3(s) �Zn. Cl 2(aq) + Mg(s)

Review �H 2 SO 4(aq) + Ba(OH)2(aq) �KNO 3(aq) + Ba. Cl 2(aq)