Chapter 6 Chemical Reactions Section 1 The Nature
























- Slides: 25

Chapter 6: Chemical Reactions Section 1: The Nature of Chemical Reactions

Chemical Reactions Change Substances • Definition: chemical reaction – a process in which a substance becomes a new substance. • There are often signs that a chemical reaction is taking place: – Color Change – Gas Production (bubbling) – Energy Transfer (getting hot or cold, glowing)

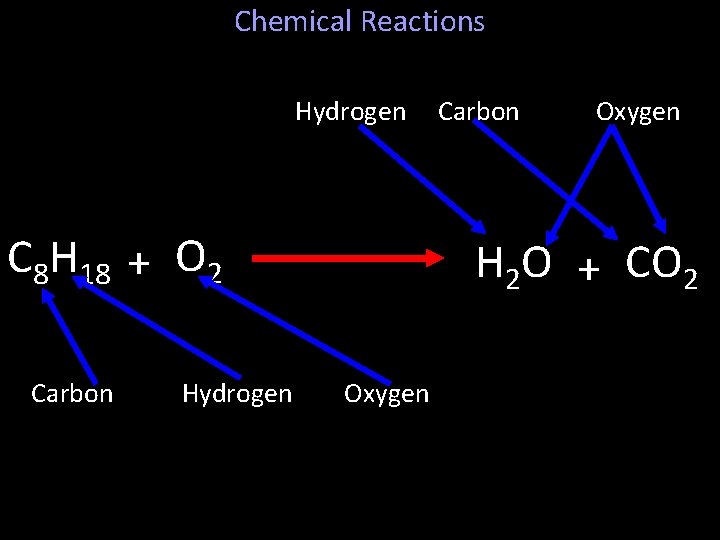
Chemical Reactions • Chemical changes rearrange atoms. – The new substance or substances contain the same atoms. – For Example: – Our cars use gasoline, which has the chemical formula C 8 H 18. – Gasoline burns when it reacts with oxygen, O 2. – When the gas burns…

Chemical Reactions Hydrogen C 8 H 18 + O 2 Carbon Hydrogen Carbon Oxygen H 2 O + CO 2 Oxygen

Reactants & Products • So, in a chemical reaction…things REACT to PRODUCE other things. • Definition: reactants – the substances in a chemical reaction that are changed. • Definition: products - the NEW substances produced in a chemical reaction.

Energy & Reactions • Lets look at the reaction of gasoline and oxygen again… – It would be far to dangerous to use gasoline as a fuel if it immediately reacted with oxygen, right? – What do we need in order to start the reaction? A SPARK!

Energy & Reactions • Energy must be added to some reactants to start the chemical reaction. – The added energy breaks the bonds that holds the reactants together. – Once the bonds are broken, the reactants rearrange into the products.

Mass is CONSERVED • One of the most important things to remember about a chemical reaction is that… • In a chemical reaction, mass is never gained or lost.

Endothermic & Exothermic • When a chemical reaction releases energy, it is called exothermic. – Definition: exothermic reaction – a chem. reaction that releases energy. • Exo. reactions make the air warmer.

Endothermic & Exothermic • When a chemical reaction absorbs energy, it is endothermic. – Definition: endothermic reaction – a chem. reaction that absorbs energy. • Endo reactions make the air colder.

Chapter 6: Chemical Reactions Section 2: Reaction Types

Synthesis Reactions • We can classify reactions based on what happens during the reaction. • There are 4 different types of reactions: – Synthesis – Decomposition – Single Displacement – Double Displacement • Lets talk about Synthesis first…

Synthesis Reactions • When things combine together in a chemical reaction, we call it “synthesis”. • Definition: synthesis reaction – a reaction in which 2 or more substances combine to form a new compound.

Synthesis reactions • A synthesis reaction has the equation: –A + B C – A & B represent the reactants. – C represents the product.

Decomposition Reactions • When a chemical reaction breaks substances apart, it is called “decomposition”. • Definition: decomposition reaction – a reaction where a compound breaks down into 2 or more substances.

Decomposition Reactions • A decomposition reaction has the equation: –C A + B –C represents the reactant compound. –The compounds breaks into A and B.

Single Displacement • In displacement reactions, substances change places. • Definition: single replacement reaction – a reaction where one element takes place of another.

Single Displacement • In displacement reactions, substances change places. • Definition: single replacement reaction – a reaction where one element takes place of another. • AX + B BX + A

Double. Displacement – Definition: Double Displacement reaction – a reaction where 2 elements trade places. – AX + BY AY + BX • Notice that X takes the place of Y and Y takes the place of X.

Balancing Chemical Equations A Beginner’s Tutorial

Chemical Equations • Whenever a chemical reaction happens, one thing must always be true: • A chemical reaction never creates or destroys atoms. – This is the Law of Conservation of Mass.

Chemical Equations • Lets look at the reaction that makes Water: H 2 + O 2 H 2 O • Something’s Wrong here… – If we look at the left side of the reaction, we find that there are… • 2 Hydrogen and 2 Oxygen – The right side of the reaction has… • 2 Hydrogen and 1 Oxygen

Chemical Equations • This reaction is NOT correct!! • It needs to be “balanced”. • To balance it, we can add coefficients to the elements & compounds. H 2 + O 2 H 2 O The subscripts can’t be changed, though.

Chemical Equations: Adding Coefficients (2) H 2 + (1) O 2 (2) H 2 O Adding the coefficients added more atoms to the reaction. Now on the left side, we have… 4 Hydrogen and 2 Oxygen And on the right side, we have… 4 Hydrogen and 2 Oxygen

Chemical Equations • (2) H 2 + (1) O 2 (2) x 2 = 4 H (2) H 2 O (1) x 2 = 2 O (2) x 2 = 4 H (2) x 1 = 1 O • Multiply the Coefficient by the Subscript.