CHAPTER 6 BONDING II MOLECULAR GEOMETRY BONDING THEORIES

CHAPTER 6: BONDING II – MOLECULAR GEOMETRY & BONDING THEORIES ADVANCE CHEMISTRY

Purpose � Learn how to determine the electron-domain geometry and molecular geometry of a molecule. � Understand what are molecular polarity and dipole moment. � Be able to tell what kind of bond the molecule have. � Understand hybridization. � Be able to calculate the bond order. � Learn about the molecular orbital.
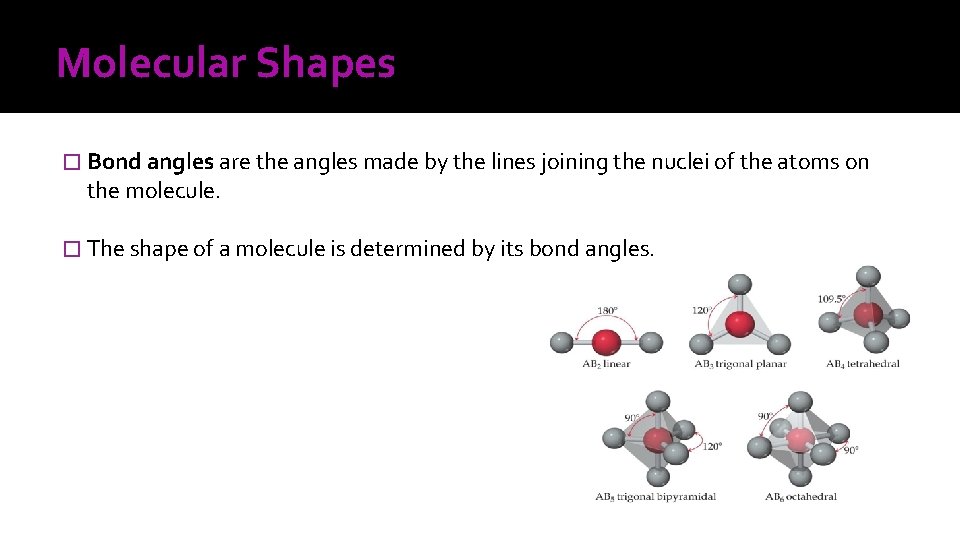
Molecular Shapes � Bond angles are the angles made by the lines joining the nuclei of the atoms on the molecule. � The shape of a molecule is determined by its bond angles.

VSERP Model � Valence-shell electron-pair repulsion (VSERP) model is the geometry of the molecule is determined by the number of electrons in the valence shell. � “The best arrangement of a given number of electron domains is the one that minimizes the repulsions among them. ” � Electron domain defines a region in which electrons are likely to be found. � We can refer to the electron pairs as bond pairs.

Electron-Domain Geometry � The arrangement of electron domains about the central atom of an ABn molecule or ion is called its Electron. Domain Geometry. � The molecular geometry is the arrangement of only the atoms in a molecule or ion- any nonbonding pairs in the geometry are non part of the description of the molecular geometry.

Bonding � Nonbonding Pairs and Bond Angle Nonbonding pairs are physically larger than bonding pairs. � Multiple Bonds and Bond Angles Double and triple bonds place greater electron density on one side of the central atom than do single bonds.
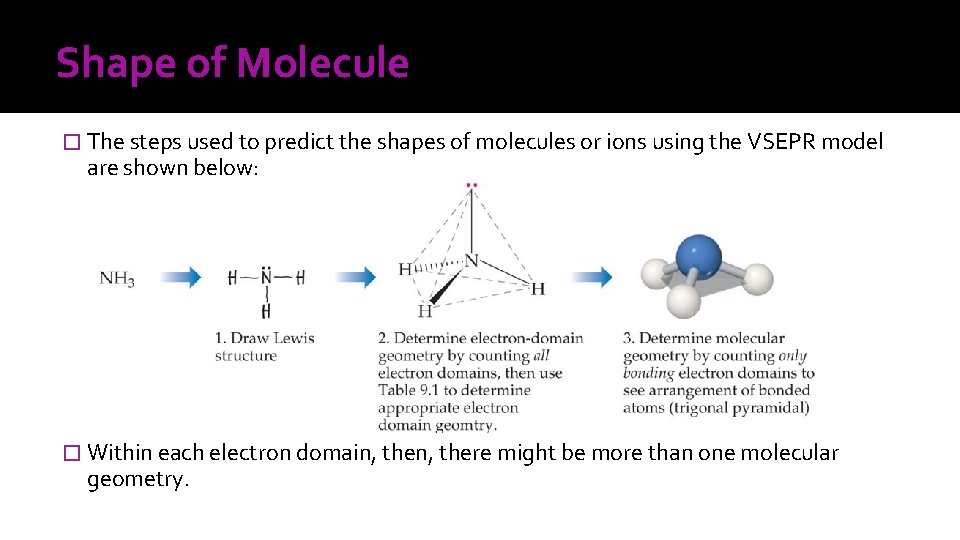
Shape of Molecule � The steps used to predict the shapes of molecules or ions using the VSEPR model are shown below: � Within each electron domain, there might be more than one molecular geometry.
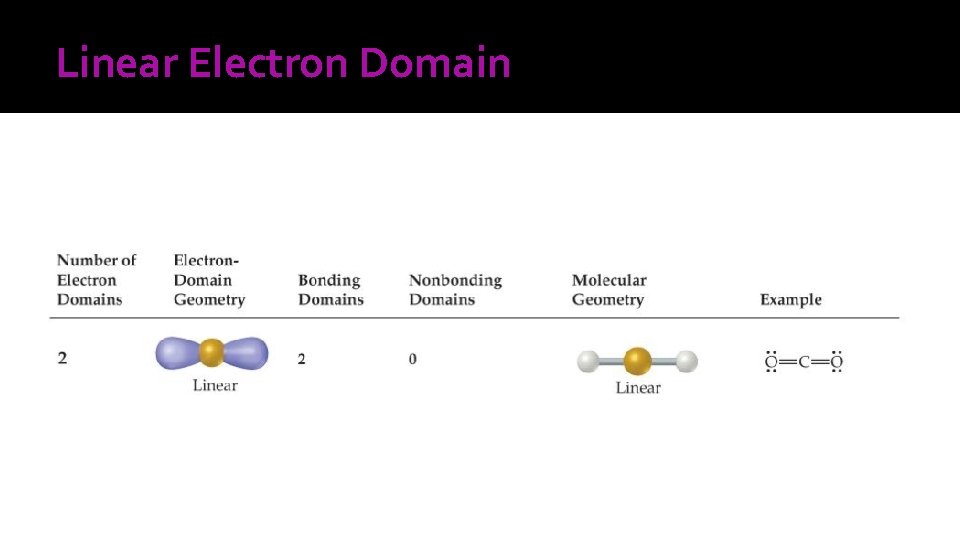
Linear Electron Domain

Trigonal Planar Electron Domain

Tetrahedral Electron Domain

Trigonal Bipyramidal Electron Domain

Octahedral Electron Domain
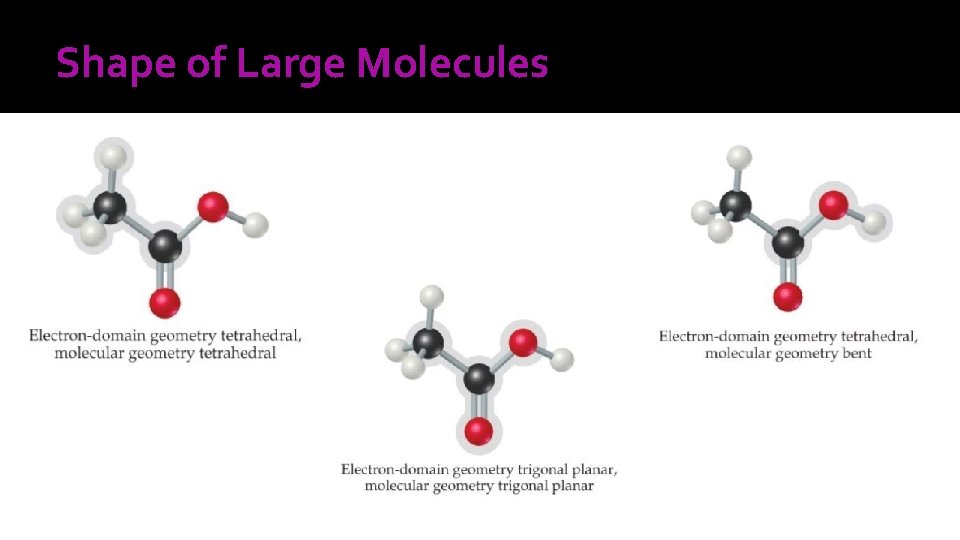
Shape of Large Molecules

Molecular Shape & Molecular Polarity � Just because a molecule possesses polar bonds does not mean the molecule as a whole will be polar.
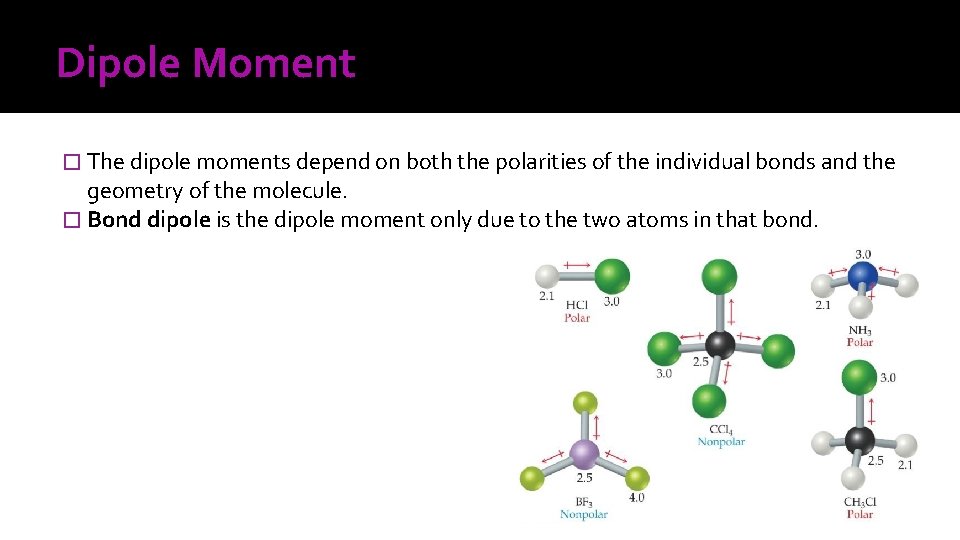
Dipole Moment � The dipole moments depend on both the polarities of the individual bonds and the geometry of the molecule. � Bond dipole is the dipole moment only due to the two atoms in that bond.

Valence Bond Theory � Molecular orbital theory assumes the formation of molecular orbitals. Both theories have a place, and neither provides the full picture, but complement each other. In valence bond theory, two atoms have their orbitals overlap, forming a bond. The two electron of the one atom is attracted to the nucleus of the other, and vice versa. When the bonding distance is optimal, then attractive forces balance the nuclear-nuclear, and electron-electron repulsion. Energy is given off when a bond forms. The differences in orbital types, accounts for the different bond strengths, not accounted for by the Lewis structures.

Bond Overlap � Increased overlap brings the electrons and nuclei closer together while simultaneously decreasing electron– electron repulsion. However, if atoms get too close, the internuclear repulsion greatly raises the energy.

Hybrid Orbital � The shape of any hybrid orbital is different from the shapes of original atomic orbitals. � The process of mixing orbitals is a mathematical operation called hybridization. � The total number of atomic orbitals remains constant, so the number of hybrid orbitals on an atom equals the number of atomic orbitals that are mixed.
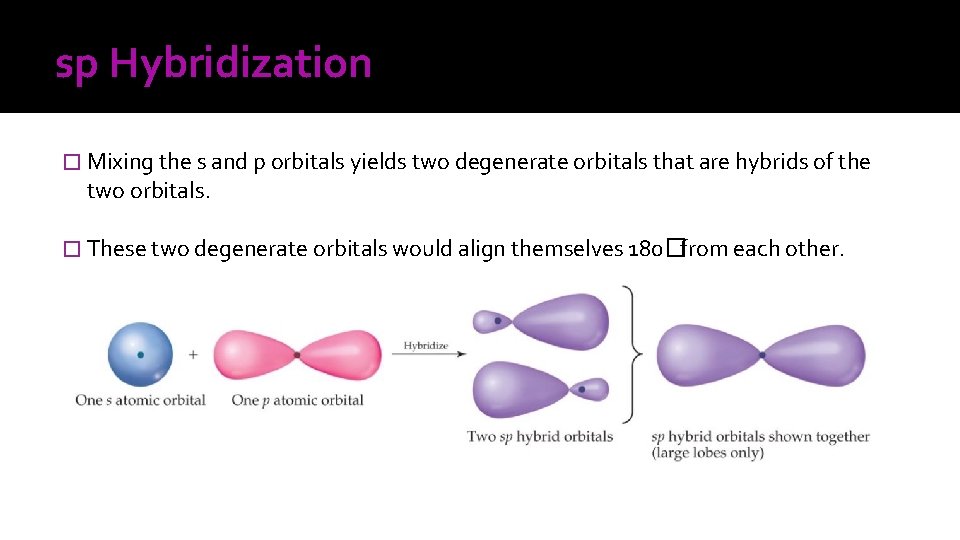
sp Hybridization � Mixing the s and p orbitals yields two degenerate orbitals that are hybrids of the two orbitals. � These two degenerate orbitals would align themselves 180�from each other.

sp² Hybridization

sp³ Hybridization

Procedure for Hybridising atomic orbitals: 1. 2. 3. Draw the Lewis structure for a molecule or an ion. Predict the overall arrangement by VSEPR. Meaning, use VSEPR model to determine the electron-domain geometry around the central atom. Deduce the hybridisation of the central atom.
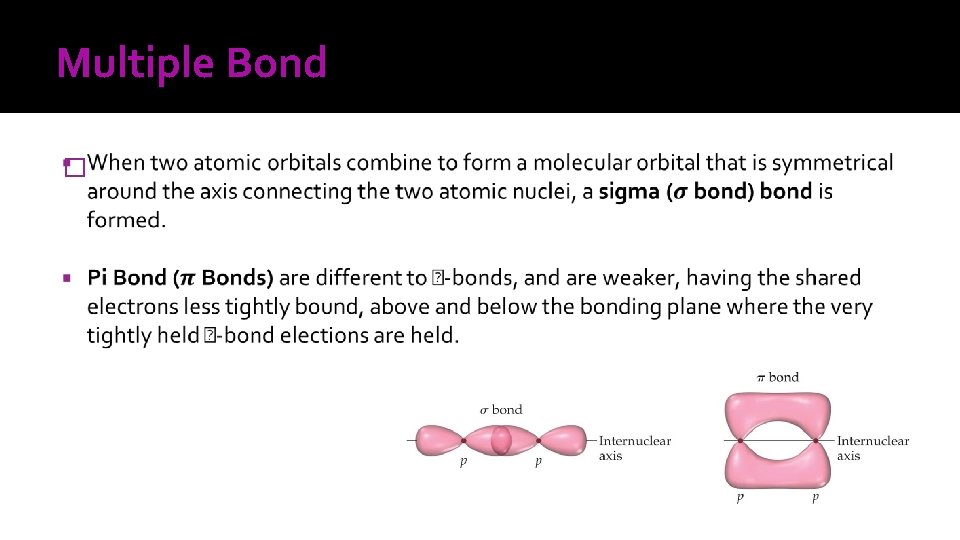
Multiple Bond �
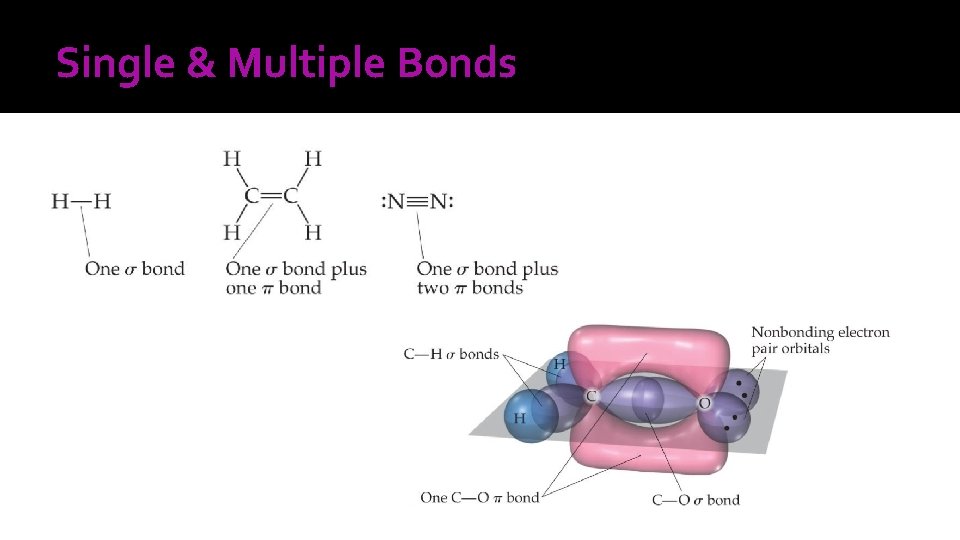
Single & Multiple Bonds
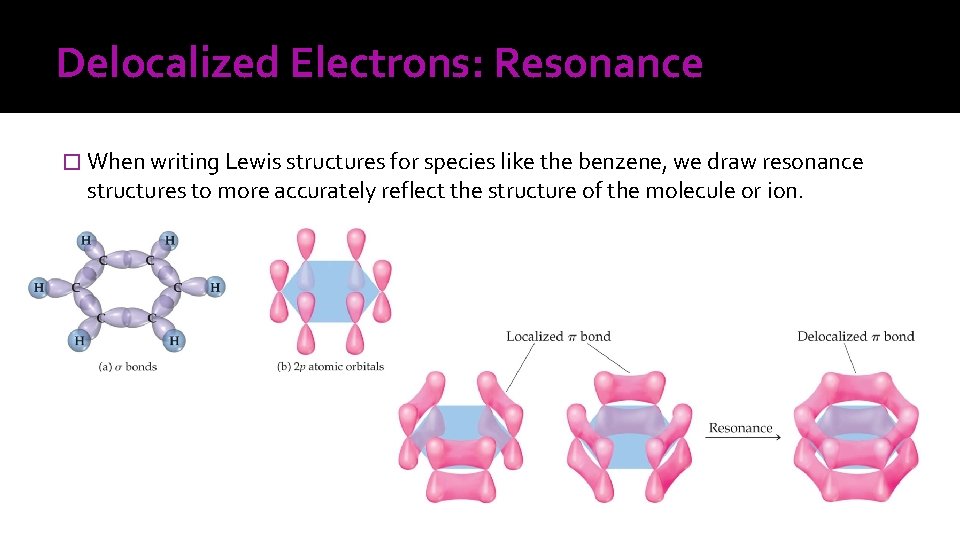
Delocalized Electrons: Resonance � When writing Lewis structures for species like the benzene, we draw resonance structures to more accurately reflect the structure of the molecule or ion.

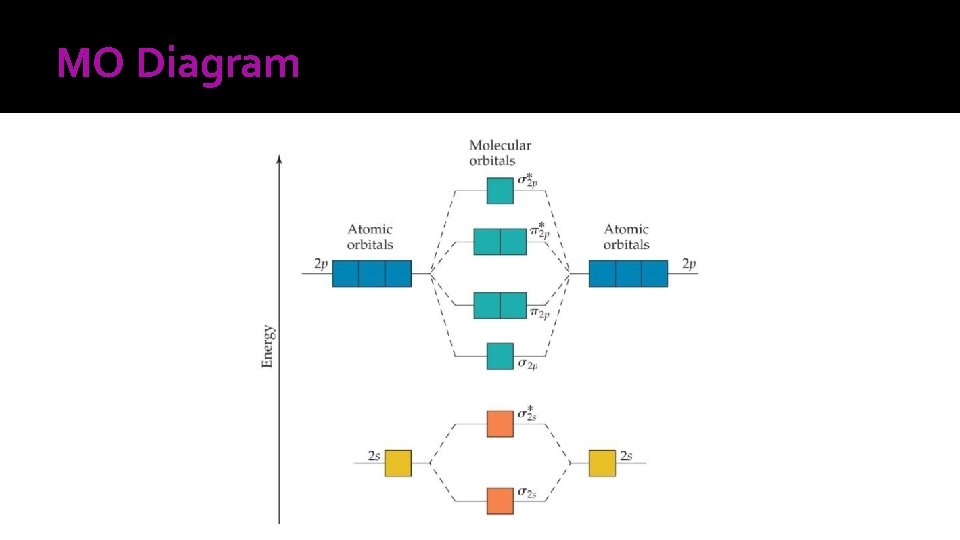
Molecular Orbital Theory � In MO theory, we invoke the wave nature of electrons. Whenever two atomic orbitals overlap, two molecular orbitals form. One MO is formed by the adding of wave functions of the two orbitals (1 s orbitals in H₂). This is referred to as constructive combination. If waves interact constructively, the resulting orbital is lower in energy and is called a bonding molecular orbital. � If waves interact destructively, the resulting orbital is higher in energy and is called an antibonding molecular orbital.



Bond Order � Bond order is the number of chemical bonds between a pair of atoms. � Bond order = ½ (no. of bonding electrons – no. of antibonding electrons) � Bond order of 1 represents a single bond, a bond order of 2 represents a double bond, and a bond order of 3 represents a triple bond. � A bond order of 0 means no bond exists. � Fraction bond orders are also possible because MO theory takes into account molecule containing old number of electrons.

Diatomic Molecule � The following are the rules that summarize the formation of MOs and how they are populated with electrons: 1. 2. 3. 4. 5. 6. The number of MOs formed equals the number of atomic orbitals combined. The more stable the bonding MO, the less stable the antibonding MO. The MOs accommodates, at most, two elections with paired spins (Pauli’s exclusion principle). Orbitals fill up from bottom to top in the energy diagram. The electrons in the bonding orbitals must always exceed those in the antibonding orbitals. The numbers of electrons in the MO’s are equal to the sum of all the electrons in the bonding atoms. Electrons fill orbitals of equal energy with one electron each first, with parallel spin (Hund’s rule)

MO Diagram

Effects of Interactions between 2 s and 2 p Atomic Orbitals. The more unpaired the electrons in the species, the stronger the attractive force, and this type of behavior is called paramagnetism. Substances with unpaired electrons are weakly repelled by the magnetic field, and this type of behavior is called diamagnetism.

References 1. 2. 3. 4. 5. 6. 7. Lecture: Theodore E. B. , Eugene, H. L. H. , Bruce E. B. , Catherine M. , Patrick W. , (2011). Chemistry: The Central Science (12 Ed). Prentice Hall. USA. Laboratory: Theodore E. B. , John H. N. , Kenneth C. K. , Matthew S. (2011). Laboratory Experiments for Chemistry: The Central Science (12 Ed). Prentice Hall. USA. Theodore E. B. , (2011). Solutions to Exercises for Chemistry: The Central Science. Prentice Hall. USA. John M. , Robert C. F. (2010). Chemistry (4 Ed): Prentice Hall Companion Website. http: //wps. prenhall. com/esm_mcmurry_chemistry_4/9/2408/616516. cw/index. html Chemistry Online at http: //preparatorychemistry. com/Bishop_Chemistry_First. htm Chemistry and You at http: //www. saskschools. ca/curr_content/science 9/chemistry/index. html Teachers Notes

END
- Slides: 35