Chapter 6 Atoms Elements and Compounds What are















- Slides: 16

Chapter 6 Atoms, Elements and Compounds

What are we going to be studying? • Transfer of Energy – In every chemical reaction, there is a change in energy • What we are made up of – Atoms are the foundation of biological chemistry and the building blocks for all living organisms – Matter is composed of tiny particles called atoms

Atoms • Chemistry is the study of matter – Matter is anything that has mass and takes up space – All living organisms are made up of matter • Atoms are the building blocks of matter – Atoms are made up of 3 particles • Protons • Neutrons • Electrons

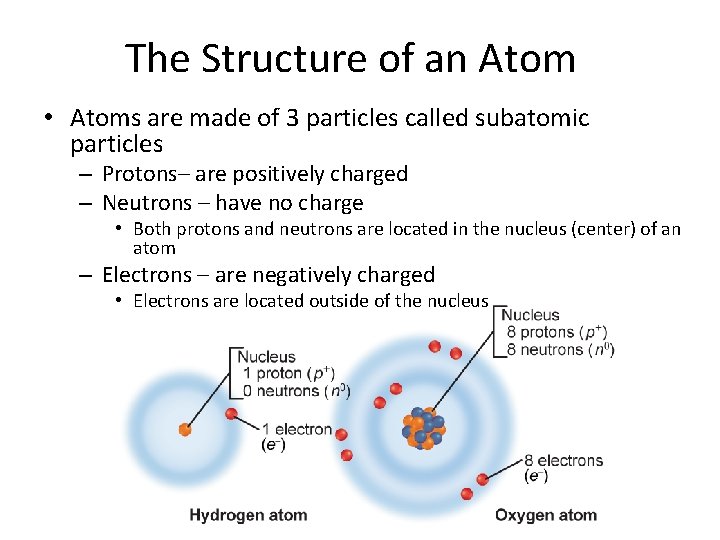
The Structure of an Atom • Atoms are made of 3 particles called subatomic particles – Protons– are positively charged – Neutrons – have no charge • Both protons and neutrons are located in the nucleus (center) of an atom – Electrons – are negatively charged • Electrons are located outside of the nucleus

Elements • An Element is a PURE substance that cannot be broken down into other substances • Elements cannot even be broken down by physical or chemical means. • There are over 100 known elements • 92 Elements occur naturally • Each element has a unique name and symbol

Non-naturally occurring elements • When we create new elements by combining 2 other elements, called bombarding – Cyclotron - there is one at MSU – The nuclei are fused together – These only last for a fraction of a second – Used for research

The Periodic Table of Elements • Horizontal rows are called periods • Vertical columns are called groups • Number of valence electron by groups

Chapter 6 RFC 1 #19 - Chart ATOMIC SYMBOL ELEMENT ATOMIC # MASS # # OF PROTONS # OF NEUTRONS # OF ELECTRONS # OF VALENCE ELECTRONS HYDROGEN H 1 1 1 0 1 1 CARBON C 6 12 6 6 6 4 NITROGEN N 7 14 7 7 7 5 OXYGEN O 8 16 8 8 8 6 PHOSPHORUS P 15 31 15 16 15 5 SULFUR S 16 32 16 16 16 6

# of subatomic particles • # protons = atomic # • # electrons = # of protons (for neutral atoms) • # neutrons = rounded mass # - atomic # – When we round we use the. 5 rule

Isotopes • Isotopes are atoms of the same element that have the same number of protons and electrons, but have a different number of neutrons. • Because they have a different number of neutrons they have different masses.

Radioactive Isotopes • Although atoms with different number of neutrons have the same overall charge, the stability of these atoms differ. • When a nucleus decays, or breaks apart, it gives of radiation that can be detected and used for many applications.

Radioactive Isotopes • The half-life, or the amount of time it take for half of the radioactive isotopes to decay, can be used to determine the age of an object • Carbon dating • How old are rocks/fossils • Tracing molecules – cancer diagnosis

Compounds • A compound is a pure substance formed when two or more different elements combine. – Are always formed from a specific combination of elements in a fixed ratio – Ex - water

Compounds • Compounds are chemically and physically different from the elements that make them up. – Its like a family • mom, dad, and child • are physically and chemically different

Compounds • Compounds cannot be broken down into simpler compounds or elements by physical means, however they can be broken down by chemical means. – Chemical means – chemical reaction

Compounds • Each compound has a chemical formula – This formula is made up of the chemical symbols for each element they contain – They also include the number of atoms of those element