Chapter 6 ABasics of Bonding Chemical Bonds and

Chapter 6 ABasics of Bonding

Chemical Bonds and the Octet Rule • What are chemical bonds? – Attraction made by transferring or sharing electrons that holds atoms together in compounds • Rearrange electrons to maximize stability – Store energy in bonds – Release energy when broken

Chemical Bonds and the Octet Rule • When are atoms most stable? – Full valence shell – Octet Rule- atoms tend to gain, lose, or share electrons until its valence is full

Chemical Bonds and the Octet Rule • BUT!!!! – Octet rule doesn’t apply to transition and inner transition metals – Also, not for H, He, Li, Be, B. Why? • Prefer to have a full 1 s (Li, Be, B) or empty shell (H) instead

Chemical Bonds and the Octet Rule

Examples • How can (2 ways) sodium and oxygen get 8 valence electrons? Which is more likely? – Sodium: lose 1 electron or gain 7 electrons • Losing is more likely – Oxygen: Lose 6 electrons or gain 2 electrons • Gaining is more likely

Types of Chemical Bonds • 3 Types • Ionic Bond – Transfer electrons to make a cation and an anion

Types of Chemical Bonds • 3 Types • Covalent Bonds – Share electrons (usually between nonmetals)

Types of Chemical Bonds • 3 rd type • Metallic Bond – Atoms with weak electron affinity that easily share their valence electrons (usually metals)

Polarity and Bond Character • What if hydrogen and fluorine bonded? – Hydrogen – weak valence – Fluorine- only needs 1 to make valence octet – When they bond, electron is pulled closer to F than H

Polarity and Bond Character • Polarity- tendency of an object to form 2 regions of opposite charge. • Each element has an electronegativity value. – H: 2. 1 – F: 4. 0 • Higher electronegativity = stronger attraction to electrons • So for HF, which atom has higher electronegativity? Which atom attracts the electron more? – Fluorine
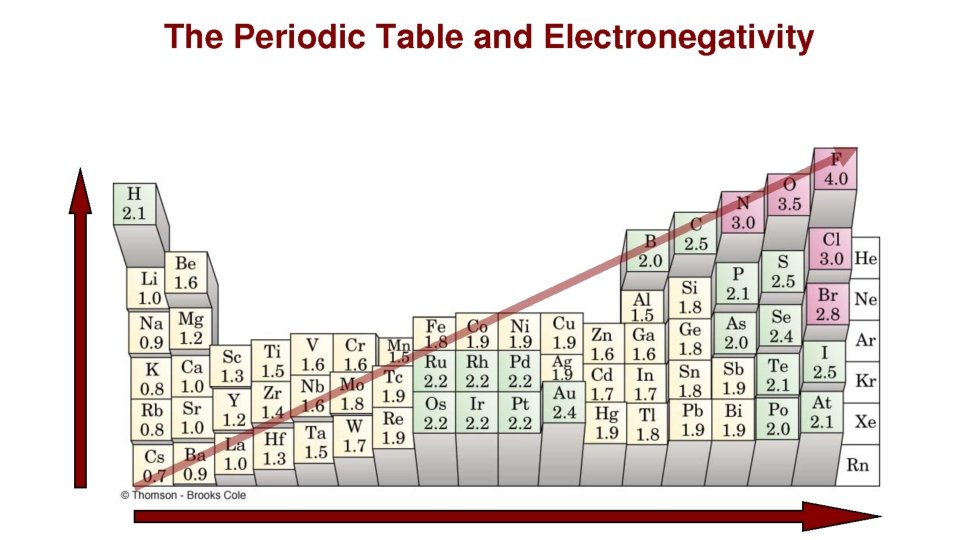
Electronegativity
- Slides: 12