Chapter 6 2 Dissolving and Solubility Lecture PLUS

Chapter 6. 2 • Dissolving and Solubility Lecture. PLUS Timberlake 1

Making process of dissolving FASTER! • Many of same things that make chemical reactions go faster – • But DISSOLVING IS A PHYSICAL CHANGE, NOT A CHEMICAL CHANGE. Lecture. PLUS Timberlake 2

Faster dissolving • When a chunk of something dissolves, you are just pulling it apart into smaller bits (even down to individual molecules or ions) Lecture. PLUS Timberlake 3

Faster dissolving • • heat stirring shaking smaller chunks (larger surface area) Lecture. PLUS Timberlake 4

In a solution, the solute molecules are randomly distributed among the solvent molecules Lecture. PLUS Timberlake 5
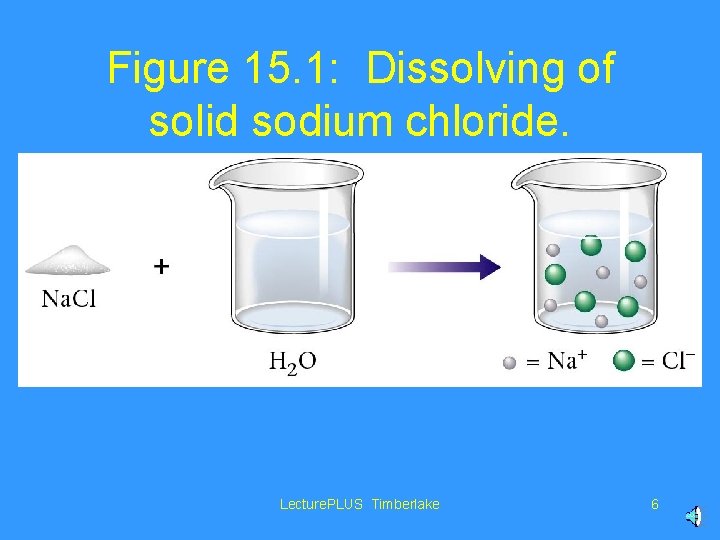
Figure 15. 1: Dissolving of solid sodium chloride. Lecture. PLUS Timberlake 6

• The solute is the substance that dissolves in a solution. • The solvent is the substance that dissolves the solute to make a solution. Lecture. PLUS Timberlake 7

Why is water such a great dissolver (solvent) for other substances? Lecture. PLUS Timberlake 8

Water is polar. . . It has a slightly negative “side” by the oxygen atom, and slightly positive “side” by the hydrogens. Lecture. PLUS Timberlake 9

Water can dissolve ionic compounds, and covalent compounds that are polar (have “slightly negative” and “slightly positive” parts). Lecture. PLUS Timberlake 10
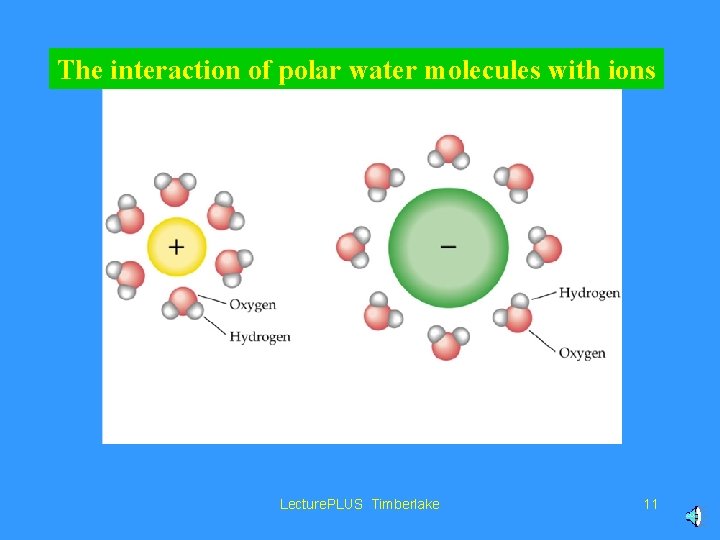
The interaction of polar water molecules with ions Lecture. PLUS Timberlake 11

Representation of the polar hydrogen chloride molecule Lecture. PLUS Timberlake 12

Chlorine hogs the electron blanket, leaving hydrogen partially, but positively, exposed Lecture. PLUS Timberlake 13
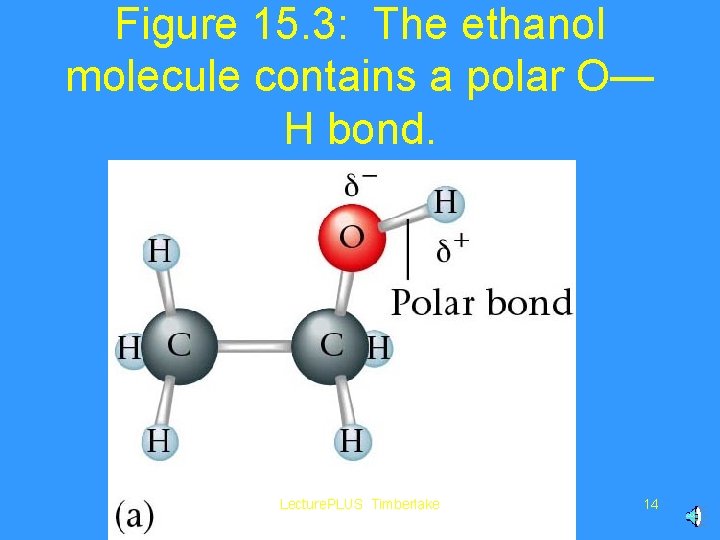
Figure 15. 3: The ethanol molecule contains a polar O— H bond. Lecture. PLUS Timberlake 14
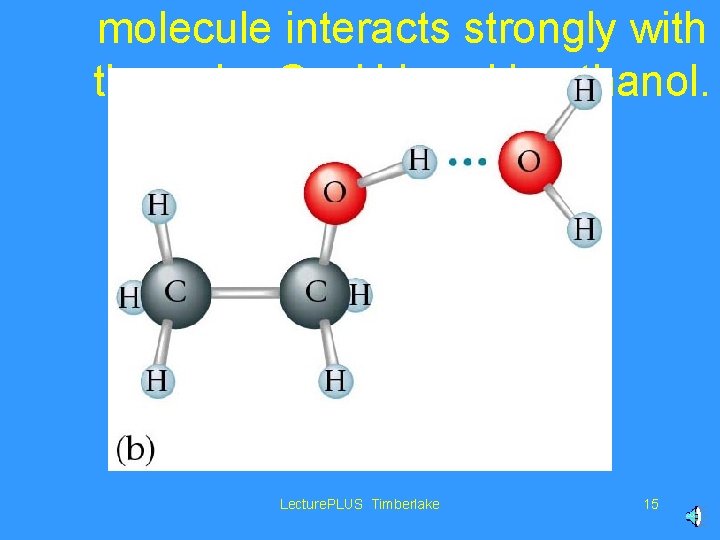
molecule interacts strongly with the polar O—H bond in ethanol. Lecture. PLUS Timberlake 15

Soap • The nonpolar side of a soap molecule is “attached” (attracted to) nonpolar grease and oil, the polar side hangs on to the water, and the dirt slips off your hands and is pulled down the drain, trapped, along with the water molecules, by that pesky soap molecule! Lecture. PLUS Timberlake 16

(a) non-polar gasoline and non-polar oil mix (b) polar vinegar and nonpolar oil do not mix (c) polar water and polar ethyl alcohol mix Lecture. PLUS Timberlake 17
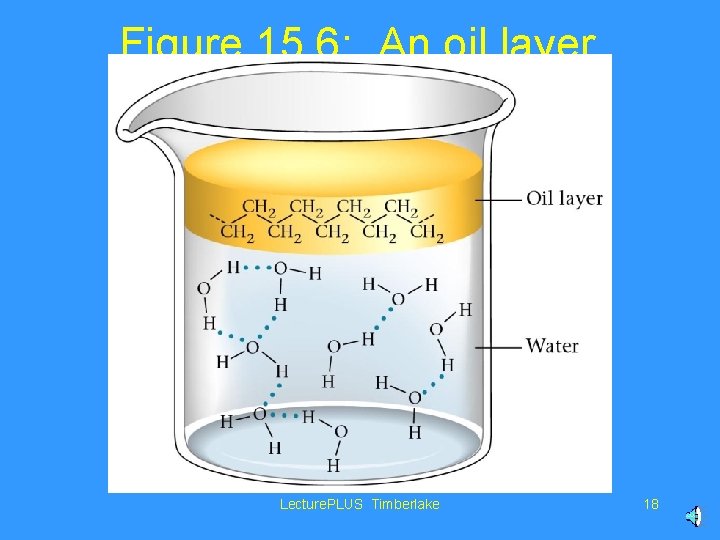
Figure 15. 6: An oil layer floating on water. Lecture. PLUS Timberlake 18

For a more detailed explanation of why polar and nonpolar materials can’t dissolve each other: • http: //www. knoxnews. com/kns/science/ article/0, 1406, KNS_9116_1971484, 00. h tml Lecture. PLUS Timberlake 19

Saturated and Unsaturated l A saturated solution contains the maximum amount of solute that can dissolve. Undissolved solute remains. l An unsaturated solution does not contain all the solute that could dissolve Lecture. PLUS Timberlake 20

Supersaturated Solutions Lecture. PLUS Timberlake 21

To see more of this supersaturation experiment: • http: //www. csudh. edu/oliver/demos/sup ersat/supersat. htm Lecture. PLUS Timberlake 22

To see more exciting supersaturation stuff, go to: • Crystallization from Supersaturated Solutions of Sodium Acetate • Note, you may need to hit “refresh” to get demonstration started and restarted. Lecture. PLUS Timberlake 23

Molarity • Concentration unit of a solution that expresses moles of solute dissolved per liter of solution. • To make a 1 molar solution of a substance, put 1 mole of stuff into a container and add solvent (usually water) up to the “ 1 liter” mark. Lecture. PLUS Timberlake 24

Not this type of Mole Lecture. PLUS Timberlake 25

Or this type of Mole Lecture. PLUS Timberlake 26

Mole • 6. 02 x 1023 is a mole 602, 000, 000 • If you counted paper at the rate of one sheet per second it would take you 19, 089, 294, 774, 226, 281 years to count a mole of paper. • It is a big number because atoms are small. Lecture. PLUS Timberlake 27

• A mole of sugar weighs about one half pound but contains how many molecules of sugar? • 602, 000, 000 • 6. 02 x 1023 molecules. Lecture. PLUS Timberlake 28
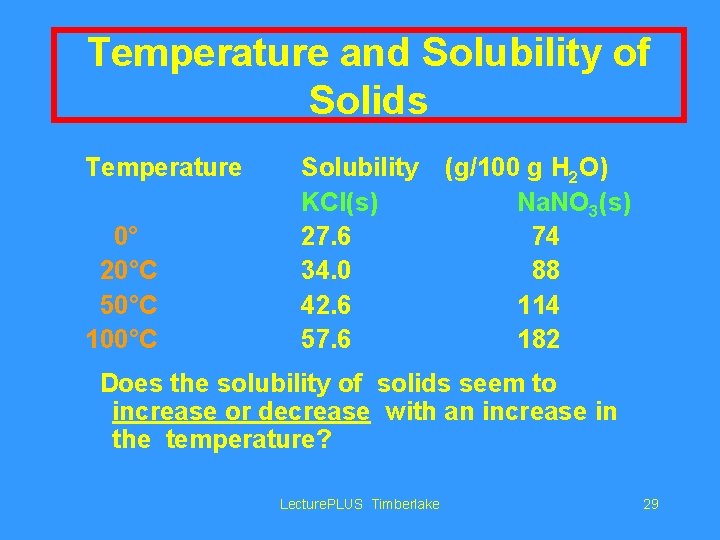
Temperature and Solubility of Solids Temperature 0° 20°C 50°C 100°C Solubility KCl(s) 27. 6 34. 0 42. 6 57. 6 (g/100 g H 2 O) Na. NO 3(s) 74 88 114 182 Does the solubility of solids seem to increase or decrease with an increase in the temperature? Lecture. PLUS Timberlake 29

A. Why would a bottle of carbonated drink possibly burst (explode) when it is left out in the hot sun ? B. Why would fish die in water that gets too warm? Lecture. PLUS Timberlake 30

Solutions A. Gas in the bottle builds up as the gas becomes less soluble in water at high temperatures, which may cause the bottle to explode. B. Because O 2 gas is less soluble in warm water, the fish may not obtain the needed amount of O 2 for their survival. Lecture. PLUS Timberlake 31
- Slides: 31