Chapter 54 Ecosystems Power Point Lectures for Biology

Chapter 54 Ecosystems Power. Point Lectures for Biology, Seventh Edition Neil Campbell and Jane Reece Lectures by Chris Romero Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Overview: Ecosystems, Energy, and Matter • An ecosystem consists of all the organisms living in a community – As well as all the abiotic factors with which they interact Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Ecosystems can range from a microcosm, such as an aquarium – To a large area such as a lake or forest Figure 54. 1 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Regardless of an ecosystem’s size – Its dynamics involve two main processes: energy flow and chemical cycling • Energy flows through ecosystems – While matter cycles within them Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Concept 54. 1: Ecosystem ecology emphasizes energy flow and chemical cycling • Ecosystem ecologists view ecosystems – As transformers of energy and processors of matter Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Ecosystems and Physical Laws • The laws of physics and chemistry apply to ecosystems – Particularly in regard to the flow of energy • Energy is conserved – But degraded to heat during ecosystem processes Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Trophic Relationships • Energy and nutrients pass from primary producers (autotrophs) – To primary consumers (herbivores) and then to secondary consumers (carnivores) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
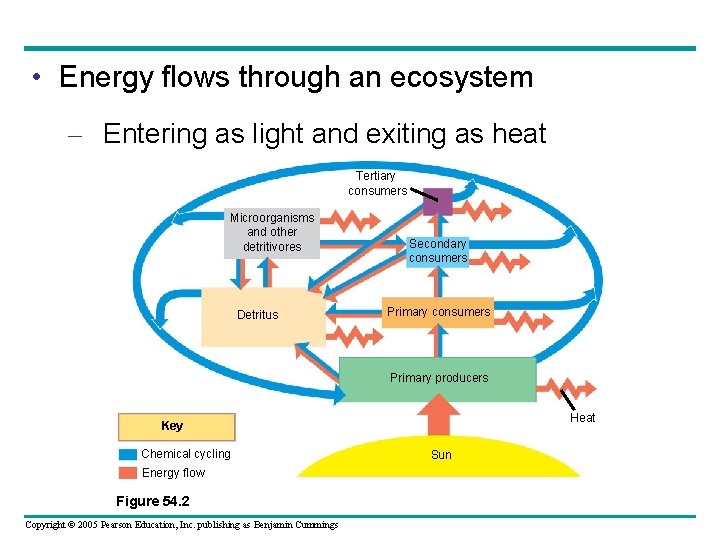
• Energy flows through an ecosystem – Entering as light and exiting as heat Tertiary consumers Microorganisms and other detritivores Detritus Secondary consumers Primary producers Heat Key Chemical cycling Energy flow Figure 54. 2 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Sun

• Nutrients cycle within an ecosystem Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Decomposition • Decomposition – Connects all trophic levels Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Detritivores, mainly bacteria and fungi, recycle essential chemical elements – By decomposing organic material and returning elements to inorganic reservoirs Figure 54. 3 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Concept 54. 2: Physical and chemical factors limit primary production in ecosystems • Primary production in an ecosystem – Is the amount of light energy converted to chemical energy by autotrophs during a given time period Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Ecosystem Energy Budgets • The extent of photosynthetic production – Sets the spending limit for the energy budget of the entire ecosystem Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

The Global Energy Budget • The amount of solar radiation reaching the surface of the Earth – Limits the photosynthetic output of ecosystems • Only a small fraction of solar energy – Actually strikes photosynthetic organisms Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Gross and Net Primary Production • Total primary production in an ecosystem – Is known as that ecosystem’s gross primary production (GPP) • Not all of this production – Is stored as organic material in the growing plants Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Net primary production (NPP) – Is equal to GPP minus the energy used by the primary producers for respiration • Only NPP – Is available to consumers Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
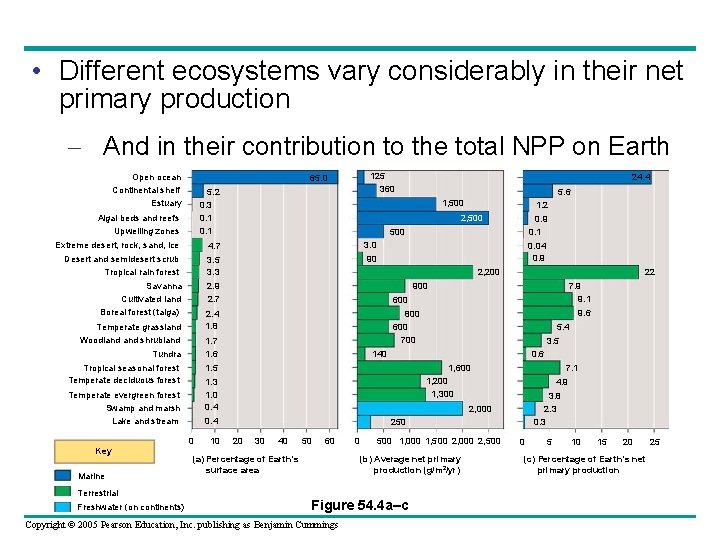
• Different ecosystems vary considerably in their net primary production – And in their contribution to the total NPP on Earth Open ocean Continental shelf Estuary 5. 2 0. 3 0. 1 Algal beds and reefs Upwelling zones Extreme desert, rock, sand, ice 4. 7 Desert and semidesert scrub Tropical rain forest 3. 5 3. 3 2. 9 2. 7 Savanna Cultivated land Boreal forest (taiga) Tropical seasonal forest Temperate deciduous forest Marine Terrestrial Freshwater (on continents) 3. 0 90 0. 04 0. 9 2, 200 10 22 900 7. 9 9. 1 600 9. 6 800 600 700 5. 4 3. 5 0. 6 140 1, 600 7. 1 1, 200 1, 300 4. 9 3. 8 2. 3 0. 3 2, 000 0. 4 0 0. 9 0. 1 500 1. 5 1. 3 1. 0 0. 4 Temperate evergreen forest Swamp and marsh Lake and stream 1. 2 2, 500 1. 7 1. 6 Tundra 24. 4 5. 6 1, 500 2. 4 1. 8 Temperate grassland Woodland shrubland Key 125 360 65. 0 250 20 30 40 50 60 0 500 1, 000 1, 500 2, 000 2, 500 (b) Average net primary production (g/m 2/yr) (a) Percentage of Earth’s surface area Figure 54. 4 a–c Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 0 5 10 15 20 (c) Percentage of Earth’s net primary production 25
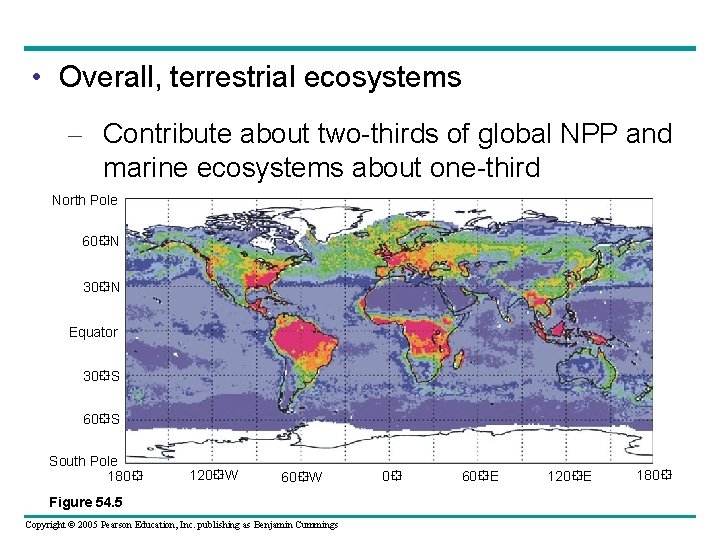
• Overall, terrestrial ecosystems – Contribute about two-thirds of global NPP and marine ecosystems about one-third North Pole 60 N 30 N Equator 30 S 60 S South Pole 180 120 W 60 W Figure 54. 5 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 0 60 E 120 E 180

Primary Production in Marine and Freshwater Ecosystems • In marine and freshwater ecosystems – Both light and nutrients are important in controlling primary production Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Light Limitation • The depth of light penetration – Affects primary production throughout the photic zone of an ocean or lake Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Nutrient Limitation • More than light, nutrients limit primary production – Both in different geographic regions of the ocean and in lakes Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• A limiting nutrient is the element that must be added – In order for production to increase in a particular area • Nitrogen and phosphorous – Are typically the nutrients that most often limit marine production Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Nutrient enrichment experiments – Confirmed that nitrogen was limiting phytoplankton growth in an area of the ocean EXPERIMENT Pollution from duck farms concentrated near Moriches Bay adds both nitrogen and phosphorus to the coastal water off Long Island. Researchers cultured the phytoplankton Nannochloris atomus with water collected from several bays. 30 s I ng Lo Coast of Long Island, New York. The numbers on the map indicate the data collection stations. Figure 54. 6 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 5 4 Bay uth o S reat G 2 21 d lan 15 11 19 Shinnecock Bay Moriches Bay Atlantic Ocean
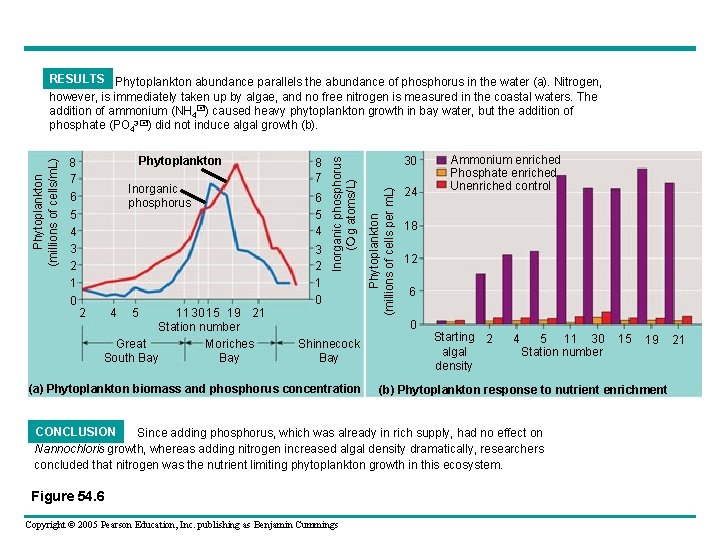
5 4 3 2 1 0 Inorganic phosphorus 2 4 5 11 30 15 19 21 Station number Great Moriches South Bay 8 7 6 5 4 3 2 1 0 30 Phytoplankton (millions of cells per m. L) Phytoplankton 8 7 6 Inorganic phosphorus ( g atoms/L) Phytoplankton (millions of cells/m. L) RESULTS Phytoplankton abundance parallels the abundance of phosphorus in the water (a). Nitrogen, however, is immediately taken up by algae, and no free nitrogen is measured in the coastal waters. The addition of ammonium (NH 4 ) caused heavy phytoplankton growth in bay water, but the addition of phosphate (PO 43 ) did not induce algal growth (b). 24 18 12 6 0 Shinnecock Bay (a) Phytoplankton biomass and phosphorus concentration Ammonium enriched Phosphate enriched Unenriched control Starting 2 algal density 4 5 11 30 Station number Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 19 (b) Phytoplankton response to nutrient enrichment CONCLUSION Since adding phosphorus, which was already in rich supply, had no effect on Nannochloris growth, whereas adding nitrogen increased algal density dramatically, researchers concluded that nitrogen was the nutrient limiting phytoplankton growth in this ecosystem. Figure 54. 6 15 21

• Experiments in another ocean region – Showed that iron limited primary production Table 54. 1 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• The addition of large amounts of nutrients to lakes – Has a wide range of ecological impacts Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• In some areas, sewage runoff – Has caused eutrophication of lakes, which can lead to the eventual loss of most fish species from the lakes Figure 54. 7 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Primary Production in Terrestrial and Wetland Ecosystems • In terrestrial and wetland ecosystems climatic factors – Such as temperature and moisture, affect primary production on a large geographic scale Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• The contrast between wet and dry climates – Can be represented by a measure called actual evapotranspiration Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
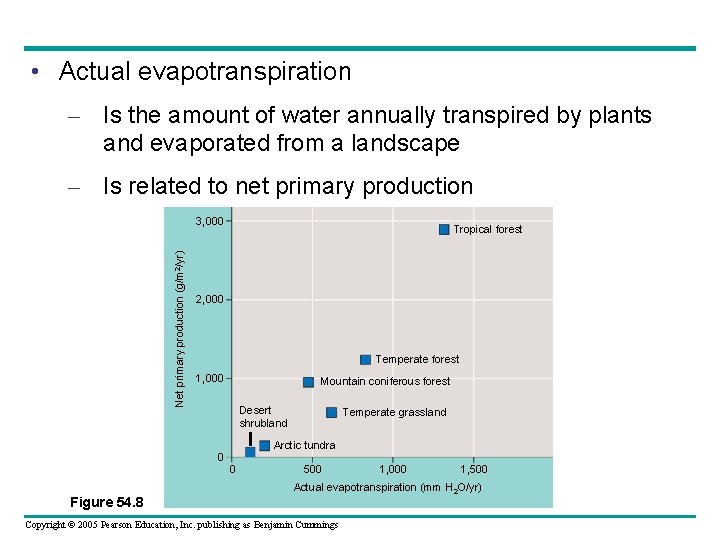
• Actual evapotranspiration – Is the amount of water annually transpired by plants and evaporated from a landscape – Is related to net primary production Net primary production (g/m 2/yr) 3, 000 Tropical forest 2, 000 Temperate forest 1, 000 Mountain coniferous forest Desert shrubland Arctic tundra 0 0 Figure 54. 8 Temperate grassland 500 1, 000 1, 500 Actual evapotranspiration (mm H 2 O/yr) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• On a more local scale – A soil nutrient is often the limiting factor in primary production EXPERIMENT Live, above-ground biomass (g dry wt/m 2) Over the summer of 1980, researchers added phosphorus to some experimental plots in the salt marsh, nitrogen to other plots, and both phosphorus and nitrogen to others. Some plots were left unfertilized as controls. Adding nitrogen (N) boosts net primary RESULTS production. 300 N P 250 200 150 N only 100 Control 50 P only 0 July June August 1980 Experimental plots receiving just phosphorus (P) do not outproduce the unfertilized control plots. CONCLUSION Figure 54. 9 These nutrient enrichment experiments confirmed that nitrogen was the nutrient limiting plant growth in this salt marsh. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Concept 54. 3: Energy transfer between trophic levels is usually less than 20% efficient • The secondary production of an ecosystem – Is the amount of chemical energy in consumers’ food that is converted to their own new biomass during a given period of time Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
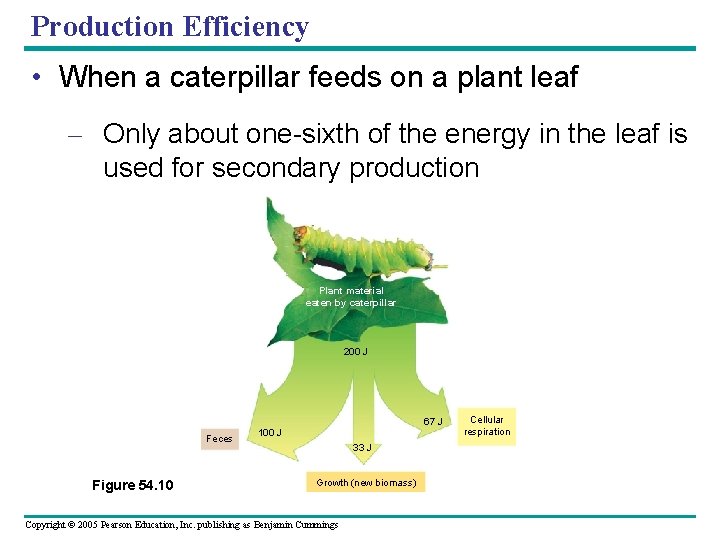
Production Efficiency • When a caterpillar feeds on a plant leaf – Only about one-sixth of the energy in the leaf is used for secondary production Plant material eaten by caterpillar 200 J 67 J Feces Figure 54. 10 100 J 33 J Growth (new biomass) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Cellular respiration

• The production efficiency of an organism – Is the fraction of energy stored in food that is not used for respiration Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Trophic Efficiency and Ecological Pyramids • Trophic efficiency – Is the percentage of production transferred from one trophic level to the next – Usually ranges from 5% to 20% Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
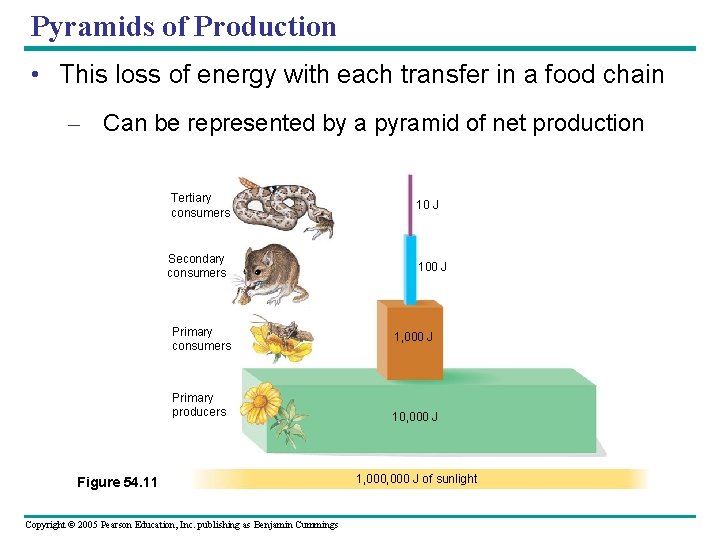
Pyramids of Production • This loss of energy with each transfer in a food chain – Can be represented by a pyramid of net production Tertiary consumers Secondary consumers Primary producers Figure 54. 11 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 10 J 100 J 1, 000 J 10, 000 J 1, 000 J of sunlight

Pyramids of Biomass • One important ecological consequence of low trophic efficiencies – Can be represented in a biomass pyramid Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
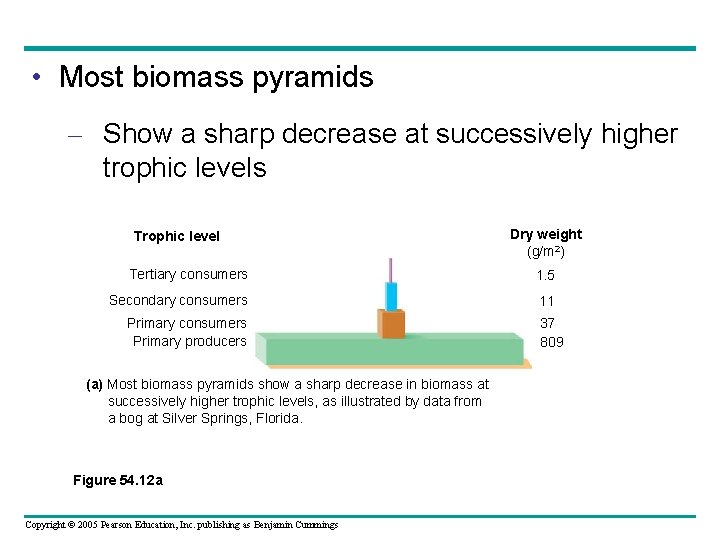
• Most biomass pyramids – Show a sharp decrease at successively higher trophic levels Trophic level Dry weight (g/m 2) Tertiary consumers 1. 5 Secondary consumers 11 Primary consumers Primary producers (a) Most biomass pyramids show a sharp decrease in biomass at successively higher trophic levels, as illustrated by data from a bog at Silver Springs, Florida. Figure 54. 12 a Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 37 809
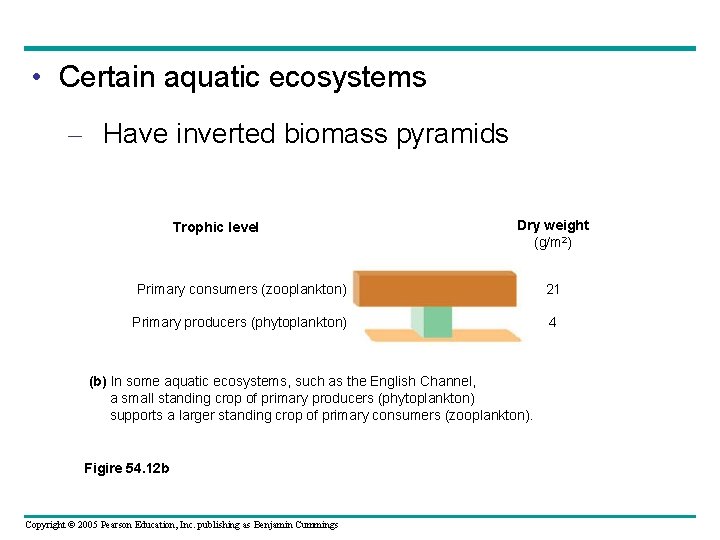
• Certain aquatic ecosystems – Have inverted biomass pyramids Trophic level Dry weight (g/m 2) Primary consumers (zooplankton) 21 Primary producers (phytoplankton) 4 (b) In some aquatic ecosystems, such as the English Channel, a small standing crop of primary producers (phytoplankton) supports a larger standing crop of primary consumers (zooplankton). Figire 54. 12 b Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
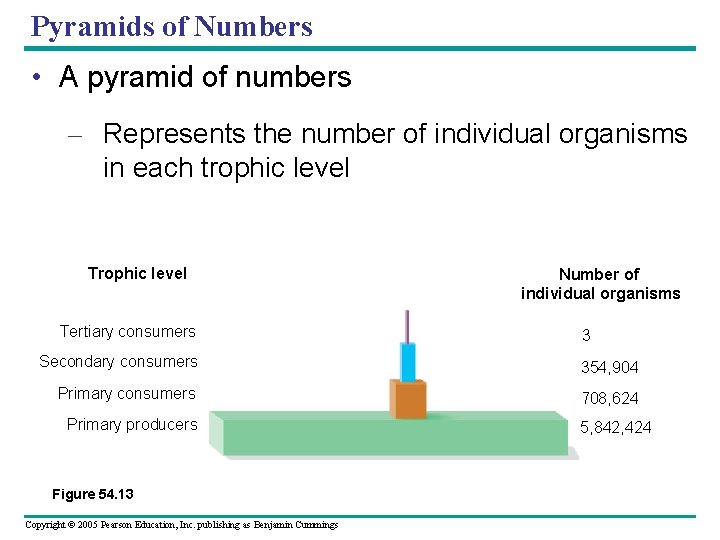
Pyramids of Numbers • A pyramid of numbers – Represents the number of individual organisms in each trophic level Tertiary consumers Number of individual organisms 3 Secondary consumers 354, 904 Primary consumers 708, 624 Primary producers Figure 54. 13 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 5, 842, 424

• The dynamics of energy flow through ecosystems – Have important implications for the human population • Eating meat – Is a relatively inefficient way of tapping photosynthetic production Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Worldwide agriculture could successfully feed many more people – If humans all fed more efficiently, eating only plant material Trophic level Secondary consumers Primary producers Figure 54. 14 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

The Green World Hypothesis • According to the green world hypothesis – Terrestrial herbivores consume relatively little plant biomass because they are held in check by a variety of factors Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Most terrestrial ecosystems – Have large standing crops despite the large numbers of herbivores Figure 54. 15 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• The green world hypothesis proposes several factors that keep herbivores in check – Plants have defenses against herbivores – Nutrients, not energy supply, usually limit herbivores – Abiotic factors limit herbivores – Intraspecific competition can limit herbivore numbers – Interspecific interactions check herbivore densities Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Concept 54. 4: Biological and geochemical processes move nutrients between organic and inorganic parts of the ecosystem • Life on Earth – Depends on the recycling of essential chemical elements • Nutrient circuits that cycle matter through an ecosystem – Involve both biotic and abiotic components and are often called biogeochemical cycles Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

A General Model of Chemical Cycling • Gaseous forms of carbon, oxygen, sulfur, and nitrogen – Occur in the atmosphere and cycle globally • Less mobile elements, including phosphorous, potassium, and calcium – Cycle on a more local level Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
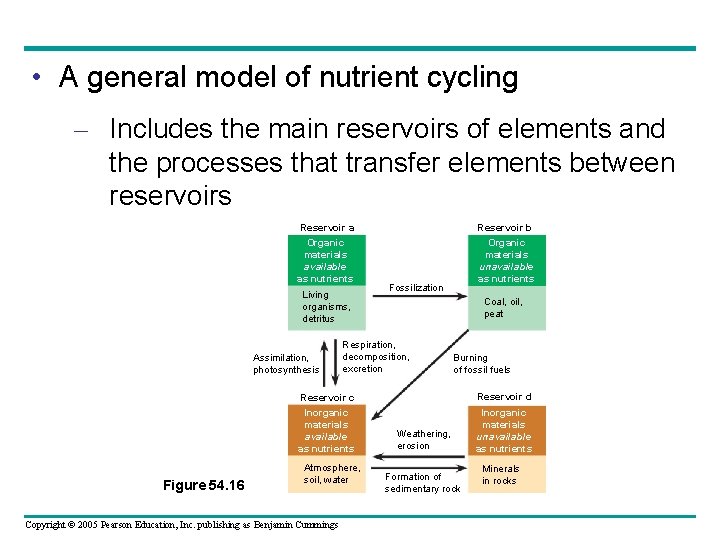
• A general model of nutrient cycling – Includes the main reservoirs of elements and the processes that transfer elements between reservoirs Reservoir a Organic materials available as nutrients Living organisms, detritus Assimilation, photosynthesis Figure 54. 16 Reservoir b Organic materials unavailable as nutrients Fossilization Coal, oil, peat Respiration, decomposition, excretion Burning of fossil fuels Reservoir c Reservoir d Inorganic materials available as nutrients Inorganic materials unavailable as nutrients Atmosphere, soil, water Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Weathering, erosion Formation of sedimentary rock Minerals in rocks

• All elements – Cycle between organic and inorganic reservoirs Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
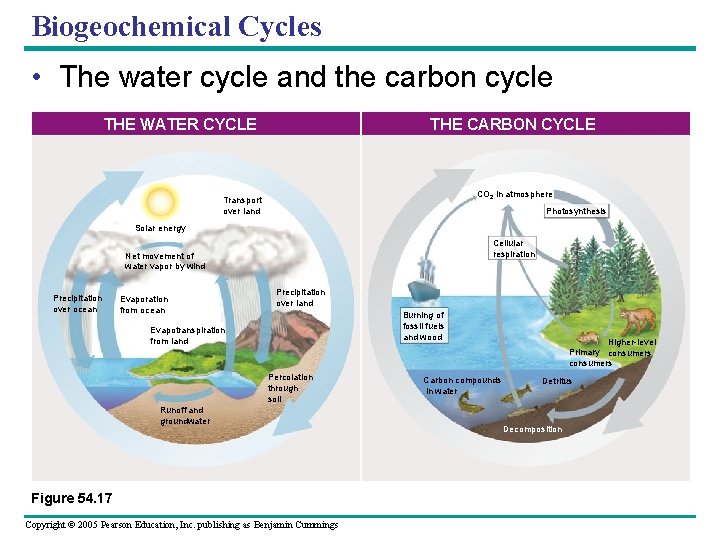
Biogeochemical Cycles • The water cycle and the carbon cycle THE CARBON CYCLE THE WATER CYCLE CO 2 in atmosphere Transport over land Photosynthesis Solar energy Cellular respiration Net movement of water vapor by wind Precipitation over ocean Evaporation from ocean Precipitation over land Burning of fossil fuels and wood Evapotranspiration from land Percolation through soil Runoff and groundwater Figure 54. 17 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Carbon compounds in water Higher-level Primary consumers Detritus Decomposition

• Water moves in a global cycle – Driven by solar energy • The carbon cycle – Reflects the reciprocal processes of photosynthesis and cellular respiration Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
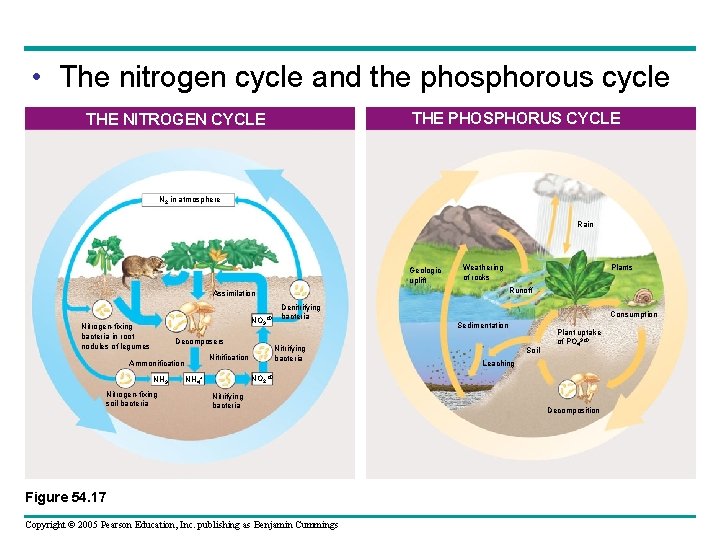
• The nitrogen cycle and the phosphorous cycle THE PHOSPHORUS CYCLE THE NITROGEN CYCLE N 2 in atmosphere Rain Geologic uplift Runoff Assimilation NO 3 Nitrogen-fixing bacteria in root nodules of legumes Decomposers Ammonification NH 3 Nitrogen-fixing soil bacteria Denitrifying bacteria Nitrification Plants Weathering of rocks Consumption Sedimentation Soil Plant uptake of PO 43 Leaching NO 2 NH 4+ Nitrifying bacteria Figure 54. 17 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Decomposition

• Most of the nitrogen cycling in natural ecosystems – Involves local cycles between organisms and soil or water • The phosphorus cycle – Is relatively localized Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
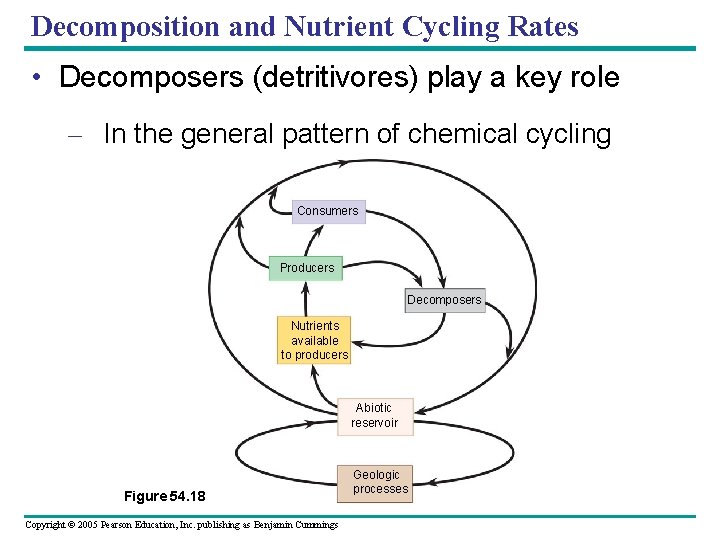
Decomposition and Nutrient Cycling Rates • Decomposers (detritivores) play a key role – In the general pattern of chemical cycling Consumers Producers Decomposers Nutrients available to producers Abiotic reservoir Figure 54. 18 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Geologic processes

• The rates at which nutrients cycle in different ecosystems – Are extremely variable, mostly as a result of differences in rates of decomposition Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Vegetation and Nutrient Cycling: The Hubbard Brook Experimental Forest • Nutrient cycling – Is strongly regulated by vegetation Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Long-term ecological research projects – Monitor ecosystem dynamics over relatively long periods of time • The Hubbard Brook Experimental Forest – Has been used to study nutrient cycling in a forest ecosystem since 1963 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• The research team constructed a dam on the site – To monitor water and mineral loss Figure 54. 19 a (a) Concrete dams and weirs built across streams at the bottom of watersheds enabled researchers to monitor the outflow of water and nutrients from the ecosystem. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• In one experiment, the trees in one valley were cut down – And the valley was sprayed with herbicides Figure 54. 19 b (b) One watershed was clear cut to study the effects of the loss of vegetation on drainage and nutrient cycling. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
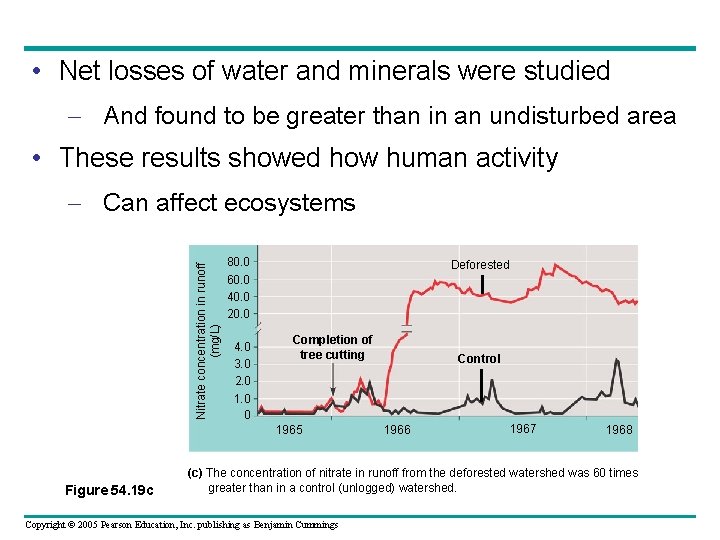
• Net losses of water and minerals were studied – And found to be greater than in an undisturbed area • These results showed how human activity Nitrate concentration in runoff (mg/L) – Can affect ecosystems 80. 0 60. 0 40. 0 20. 0 4. 0 3. 0 2. 0 1. 0 0 Deforested Completion of tree cutting 1965 Figure 54. 19 c Control 1966 1967 1968 (c) The concentration of nitrate in runoff from the deforested watershed was 60 times greater than in a control (unlogged) watershed. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Concept 54. 5: The human population is disrupting chemical cycles throughout the biosphere • As the human population has grown in size – Our activities have disrupted the trophic structure, energy flow, and chemical cycling of ecosystems in most parts of the world Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Nutrient Enrichment • In addition to transporting nutrients from one location to another – Humans have added entirely new materials, some of them toxins, to ecosystems Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Agriculture and Nitrogen Cycling • Agriculture constantly removes nutrients from ecosystems – That would ordinarily be cycled back into the soil Figure 54. 20 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Nitrogen is the main nutrient lost through agriculture – Thus, agriculture has a great impact on the nitrogen cycle • Industrially produced fertilizer is typically used to replace lost nitrogen – But the effects on an ecosystem can be harmful Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Contamination of Aquatic Ecosystems • The critical load for a nutrient – Is the amount of that nutrient that can be absorbed by plants in an ecosystem without damaging it Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• When excess nutrients are added to an ecosystem, the critical load is exceeded – And the remaining nutrients can contaminate groundwater and freshwater and marine ecosystems Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Sewage runoff contaminates freshwater ecosystems – Causing cultural eutrophication, excessive algal growth, which can cause significant harm to these ecosystems Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Acid Precipitation • Combustion of fossil fuels – Is the main cause of acid precipitation Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
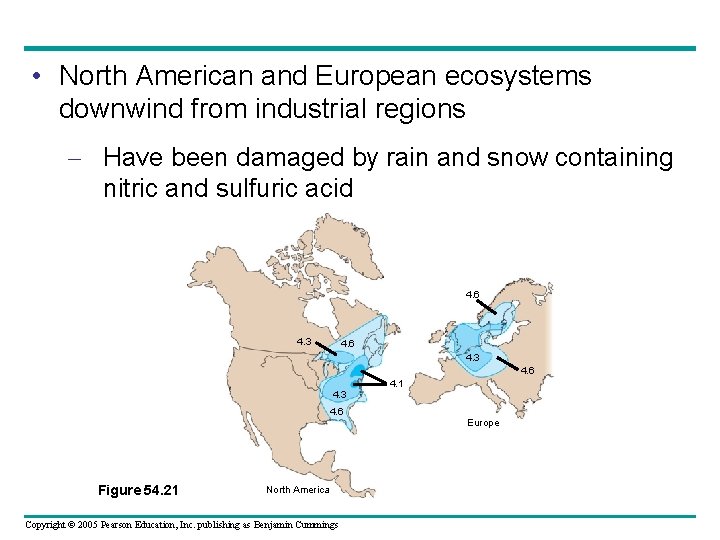
• North American and European ecosystems downwind from industrial regions – Have been damaged by rain and snow containing nitric and sulfuric acid 4. 6 4. 3 4. 1 4. 6 Europe Figure 54. 21 North America Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
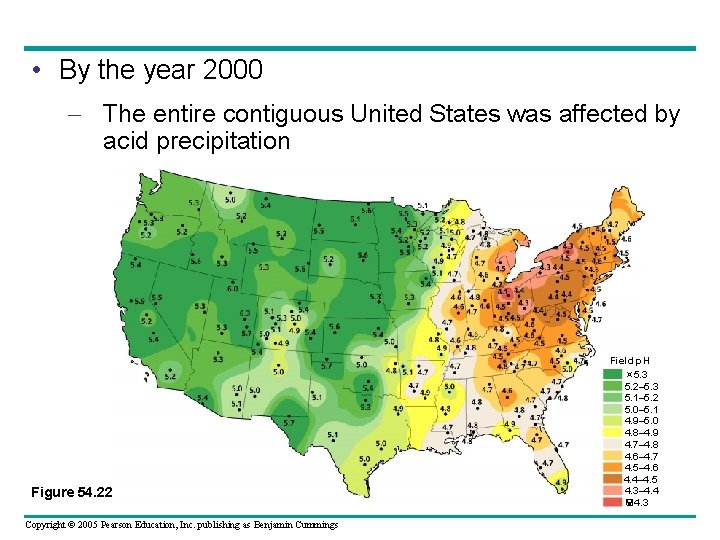
• By the year 2000 – The entire contiguous United States was affected by acid precipitation Figure 54. 22 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Field p. H 5. 3 5. 2– 5. 3 5. 1– 5. 2 5. 0– 5. 1 4. 9– 5. 0 4. 8– 4. 9 4. 7– 4. 8 4. 6– 4. 7 4. 5– 4. 6 4. 4– 4. 5 4. 3– 4. 4 4. 3

• Environmental regulations and new industrial technologies – Have allowed many developed countries to reduce sulfur dioxide emissions in the past 30 years Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Toxins in the Environment • Humans release an immense variety of toxic chemicals – Including thousands of synthetics previously unknown to nature • One of the reasons such toxins are so harmful – Is that they become more concentrated in successive trophic levels of a food web Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
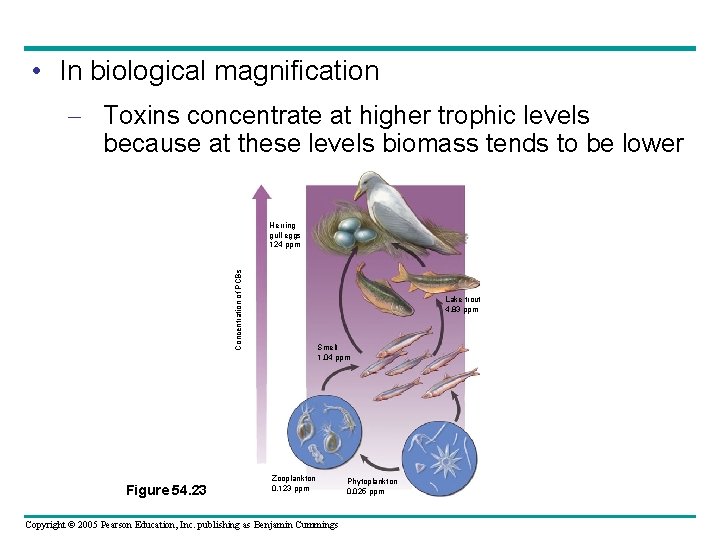
• In biological magnification – Toxins concentrate at higher trophic levels because at these levels biomass tends to be lower Concentration of PCBs Herring gull eggs 124 ppm Figure 54. 23 Lake trout 4. 83 ppm Smelt 1. 04 ppm Zooplankton 0. 123 ppm Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Phytoplankton 0. 025 ppm

• In some cases, harmful substances – Persist for long periods of time in an ecosystem and continue to cause harm Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Atmospheric Carbon Dioxide • One pressing problem caused by human activities – Is the rising level of atmospheric carbon dioxide Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
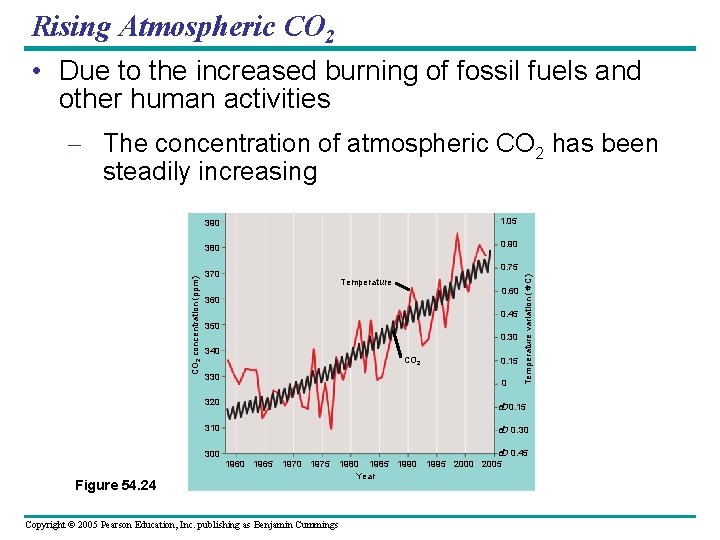
Rising Atmospheric CO 2 • Due to the increased burning of fossil fuels and other human activities 390 1. 05 380 0. 90 0. 75 370 Temperature 0. 60 360 0. 45 350 0. 30 340 CO 2 330 0 320 0. 15 310 300 0. 15 Temperature variation ( C) CO 2 concentration (ppm) – The concentration of atmospheric CO 2 has been steadily increasing 0. 30 1965 1970 1975 Figure 54. 24 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 0. 45 1980 1985 1990 1995 2000 2005 Year

How Elevated CO 2 Affects Forest Ecology: The FACTS-I Experiment • The FACTS-I experiment is testing how elevated CO 2 – Influences tree growth, carbon concentration in soils, and other factors over a ten-year period Figure 54. 25 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

The Greenhouse Effect and Global Warming • The greenhouse effect is caused by atmospheric CO 2 – But is necessary to keep the surface of the Earth at a habitable temperature Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Increased levels of atmospheric CO 2 are magnifying the greenhouse effect – Which could cause global warming and significant climatic change Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Depletion of Atmospheric Ozone • Life on Earth is protected from the damaging effects of UV radiation – By a protective layer or ozone molecules present in the atmosphere Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
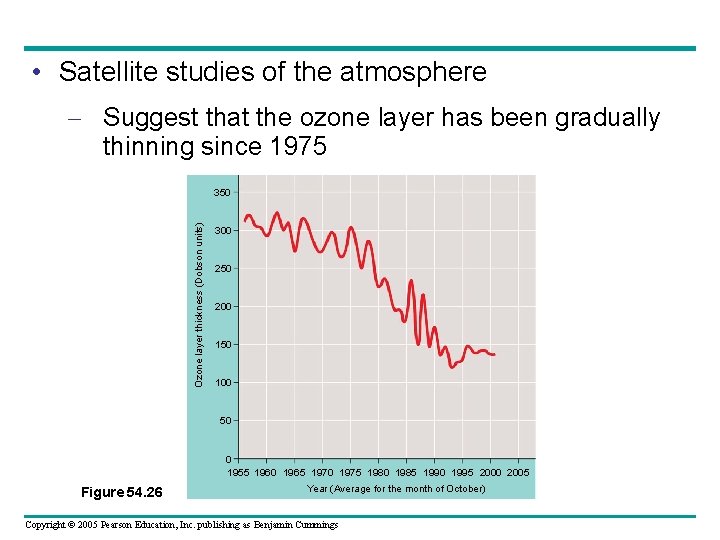
• Satellite studies of the atmosphere – Suggest that the ozone layer has been gradually thinning since 1975 Ozone layer thickness (Dobson units) 350 300 250 200 150 100 50 0 1955 1960 1965 1970 1975 1980 1985 1990 1995 2000 2005 Figure 54. 26 Year (Average for the month of October) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
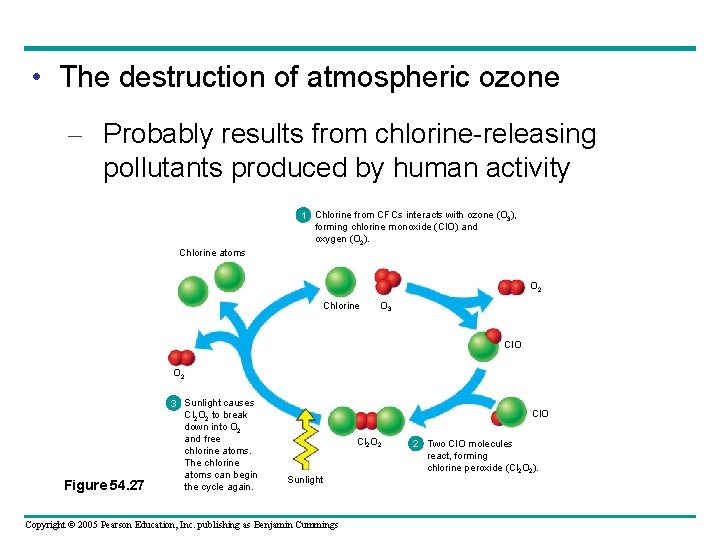
• The destruction of atmospheric ozone – Probably results from chlorine-releasing pollutants produced by human activity 1 Chlorine from CFCs interacts with ozone (O 3), forming chlorine monoxide (Cl. O) and oxygen (O 2). Chlorine atoms O 2 Chlorine O 3 Cl. O O 2 Figure 54. 27 3 Sunlight causes Cl 2 O 2 to break down into O 2 and free chlorine atoms. The chlorine atoms can begin the cycle again. Cl. O Cl 2 O 2 Sunlight Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 2 Two Cl. O molecules react, forming chlorine peroxide (Cl 2 O 2).
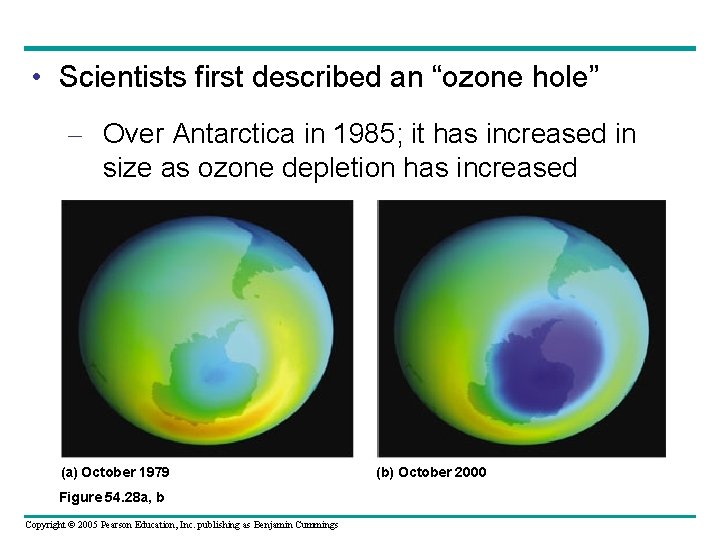
• Scientists first described an “ozone hole” – Over Antarctica in 1985; it has increased in size as ozone depletion has increased (a) October 1979 Figure 54. 28 a, b Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings (b) October 2000
- Slides: 83