Chapter 5 Thermochemistry Energy Changes in Reactions Outline





























- Slides: 57

Chapter 5 Thermochemistry: Energy Changes in Reactions Outline I. A. B. III. A. B. IV. V. VI. Energy Types Law of Conservation of Energy First Law of Thermodynamics State Functions Work Enthalpy Heating Curve of Water Calorimetry Hess’ Law

Kinetic & Potential Energy

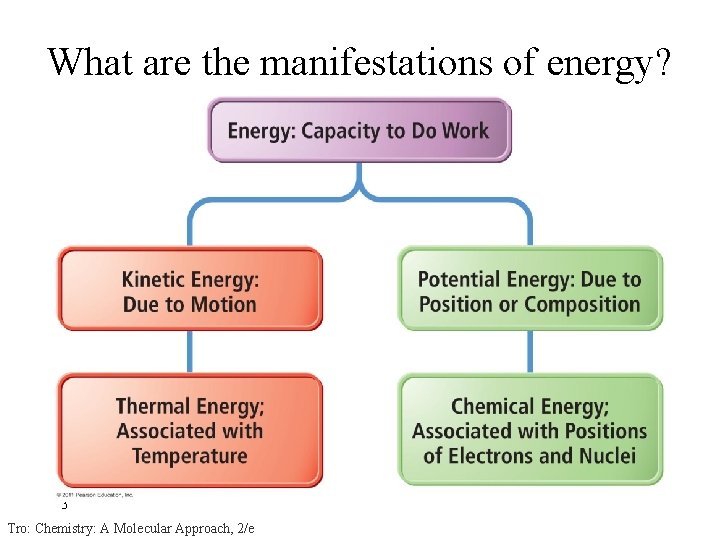
What are the manifestations of energy? 3 Tro: Chemistry: A Molecular Approach, 2/e

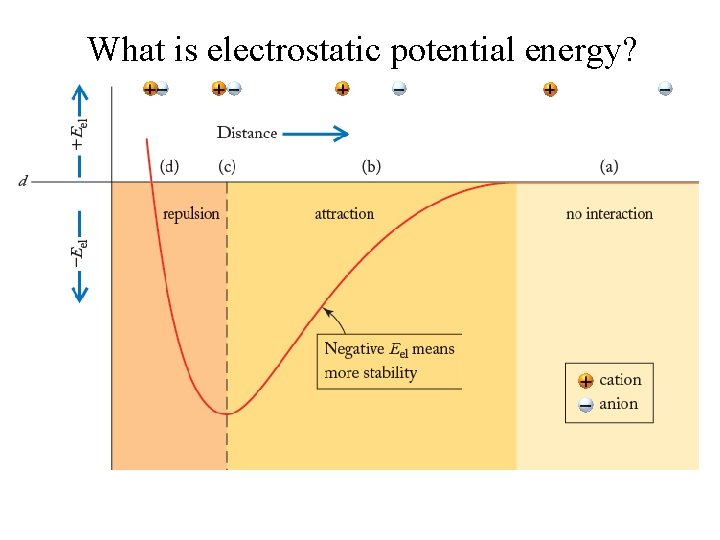
What is electrostatic potential energy?

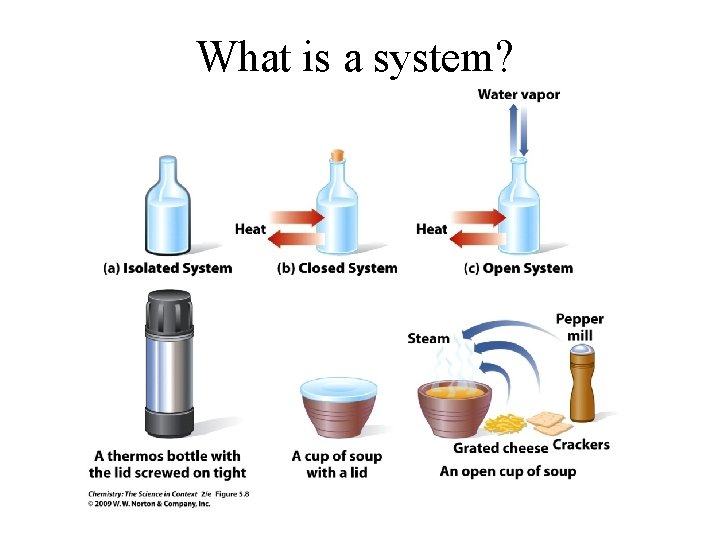
What is a system?

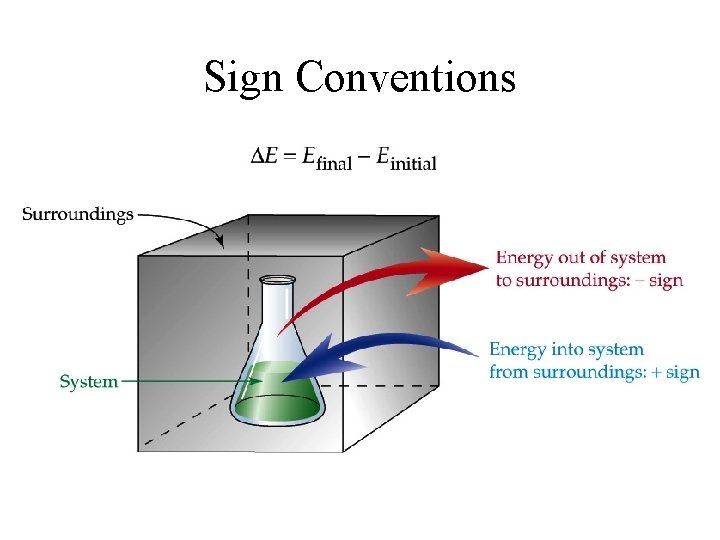
Sign Conventions

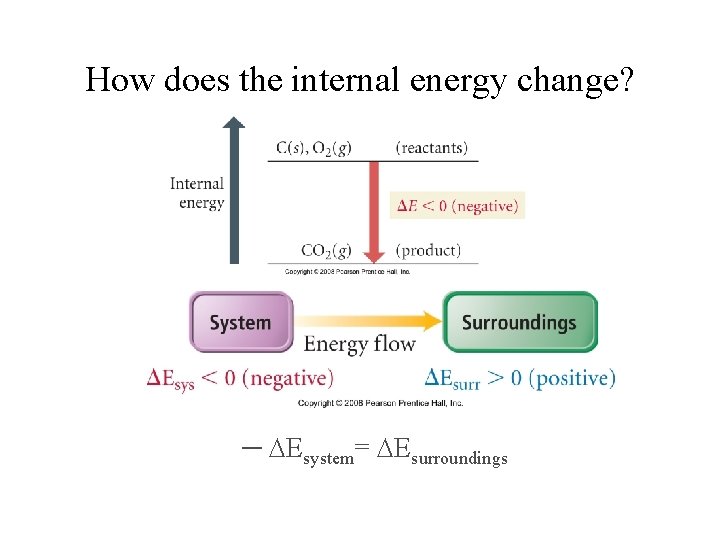
How does the internal energy change? ─ Esystem= Esurroundings

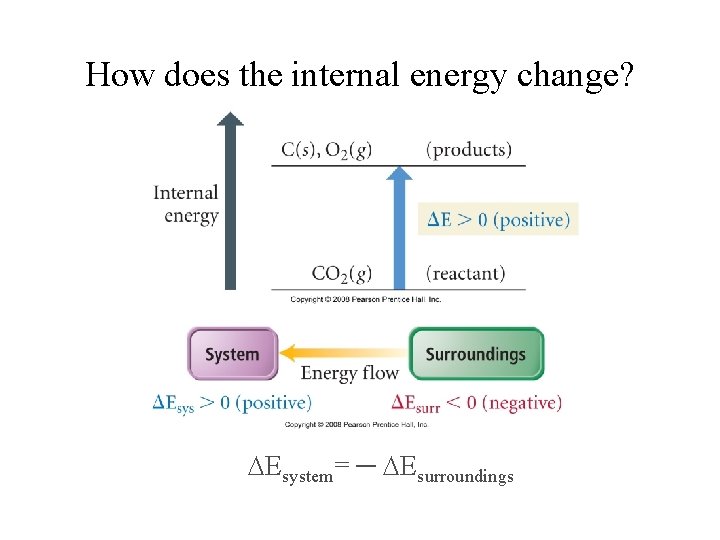
How does the internal energy change? Esystem= ─ Esurroundings

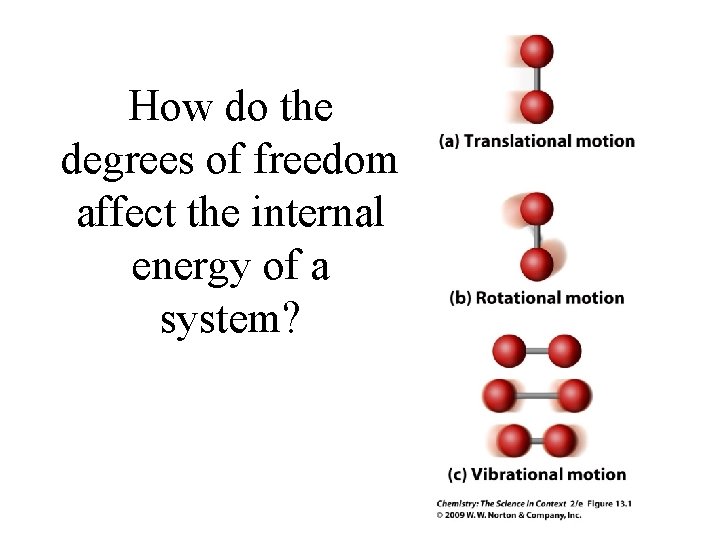
How do the degrees of freedom affect the internal energy of a system?

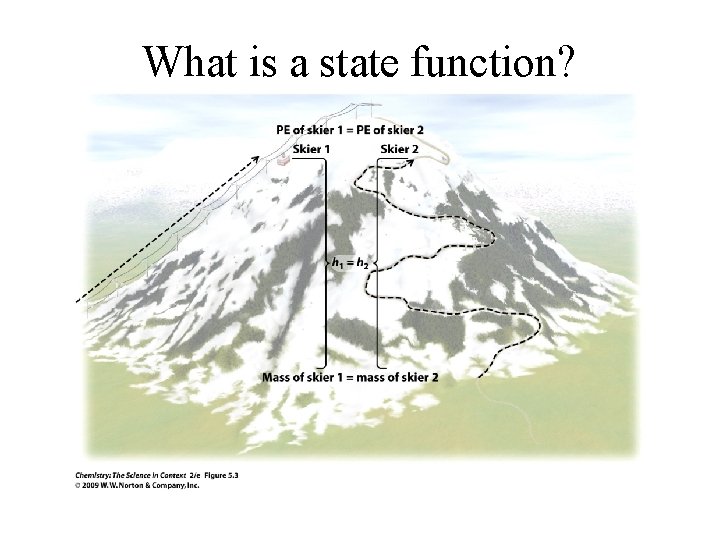
What is a state function?

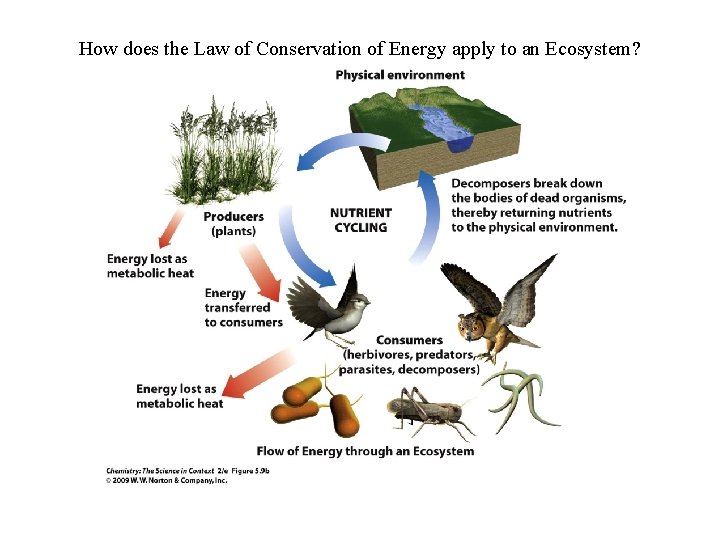
How does the Law of Conservation of Energy apply to an Ecosystem?

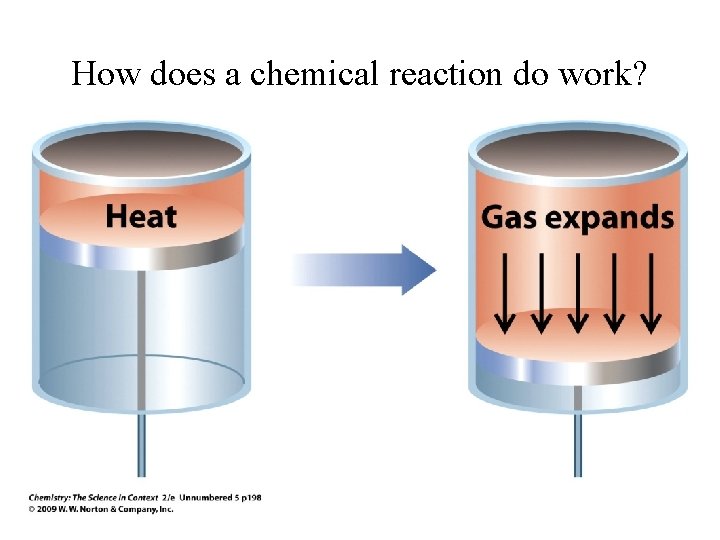
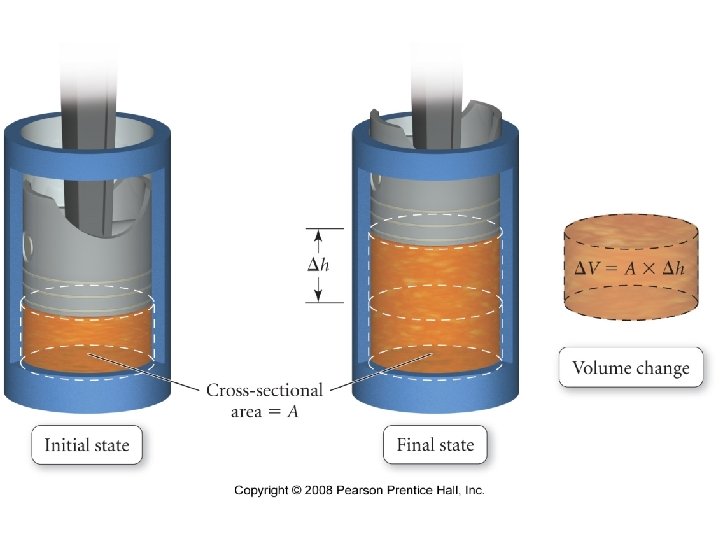
How does a chemical reaction do work?

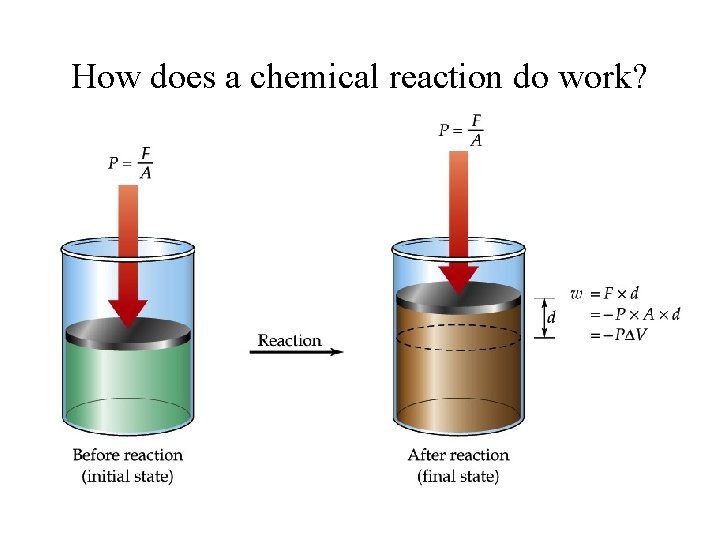
How does a chemical reaction do work?


Example – Work Calculate the work associated with the expansion of a gas from 46 L to 64 L at a constant external pressure of 15 atm.


Examples – Energy A 100 W electric heater (1 W = 1 J/s) operates for 20 minutes to heat a gas cylinder. The gas expands from 2. 04 L to 2. 54 L against an atmospheric pressure of 1. 0 atm. What is the change in internal energy of the gas?

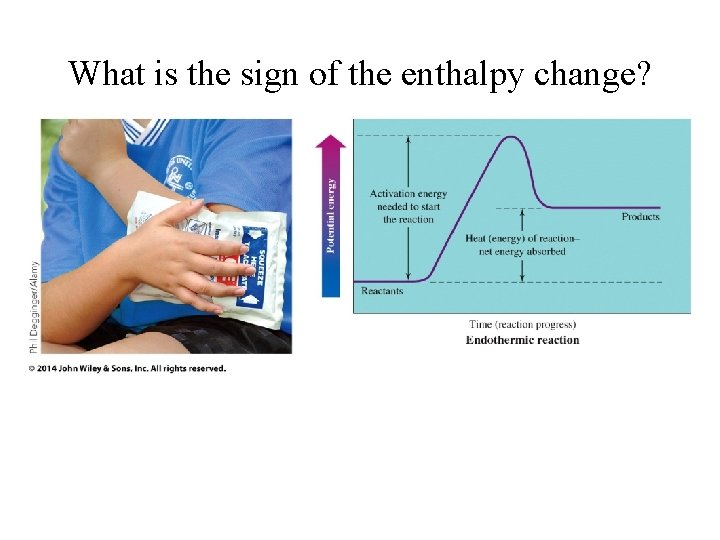
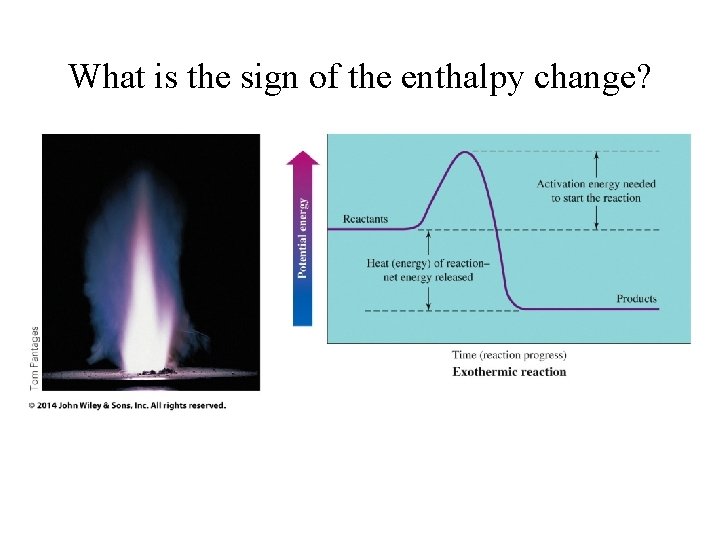
What is the sign of the enthalpy change?


What is the sign of the enthalpy change?

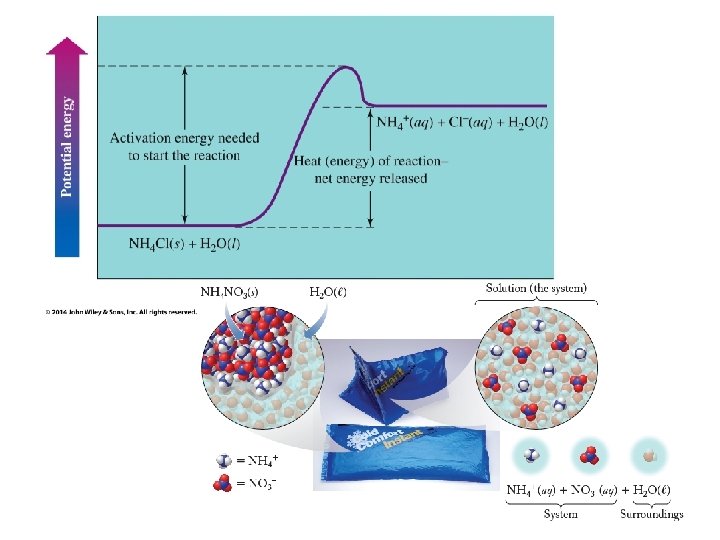
Where does the heat flow? • Enthalpy transferred into products • endothermic • H = + • Enthalpy transferred out of reactants • exothermic • H =

How are the enthalpy of a forward and a reverse reaction related? H 2 O(g) H 2(g) + 1/2 O 2(g) H = +241. 8 k. J H 2(g) + 1/2 O 2(g) H 2 O(g) H = 241. 8 k. J Hforward = Hreverse (For reversible reactions)

How does the amount of substance undergoing change affect the enthalpy? H 2 O(g) H 2(g) + 1/2 O 2(g) H = +241. 8 k. J 2 H 2 O(g) 2 H 2(g) + 1 O 2(g) H = +483. 6 k. J

Does the physical state of reactions and products affect the enthalpy? H 2 O(g) H 2(g) + 1/2 O 2(g) H = +241. 8 k. J H 2 O(l) H 2(g) + 1/2 O 2(g) H = +285. 8 k. J

Examples – Energy Stoichiometry If 5, 449 k. J of energy is absorbed when gaseous water is decomposed into its elements, how many grams of water was used? H 2 O (g) H 2 (g) + ½ O 2 (g) ∆H = 241. 8 k. J

Example – Energy Stoichiometry When 2. 00 moles of sulfur dioxide gas reacts completely with 1. 00 moles of oxygen gas to form 2. 00 moles of sulfur trioxide gas at 25 °C and a constant pressure of 1. 00 atm, 198 k. J of energy is released as heat. Calculate ΔH and ΔE for this process.

Example – Energy Stoichiometry Solid sulfur, S, reacts with carbon dioxide gas to produce sulfur dioxide gas and carbon solid, ΔH = - 75. 8 k. J. If 12. 9 g of sulfur react with 9. 70 g of carbon dioxide, how many k. J of energy are released or absorbed?

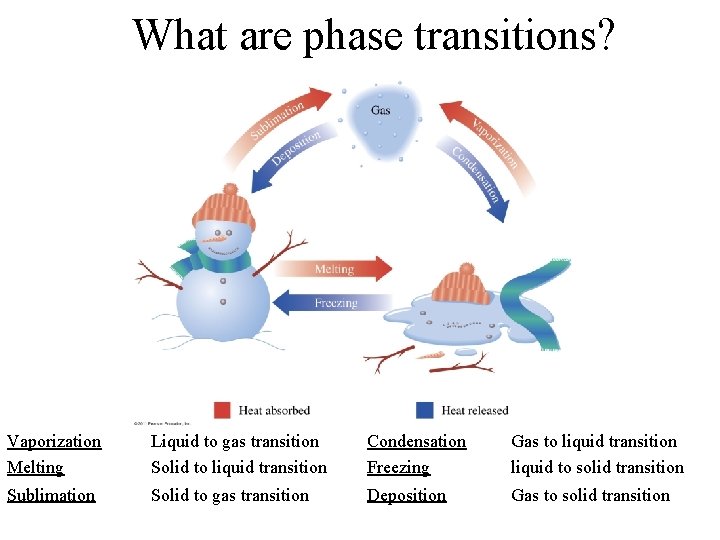
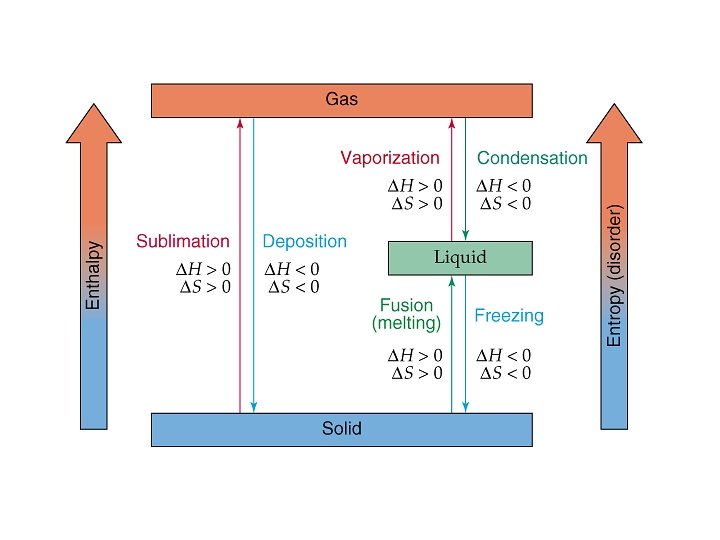
What are phase transitions? Vaporization Melting Liquid to gas transition Solid to liquid transition Condensation Freezing Gas to liquid transition liquid to solid transition Sublimation Solid to gas transition Deposition Gas to solid transition


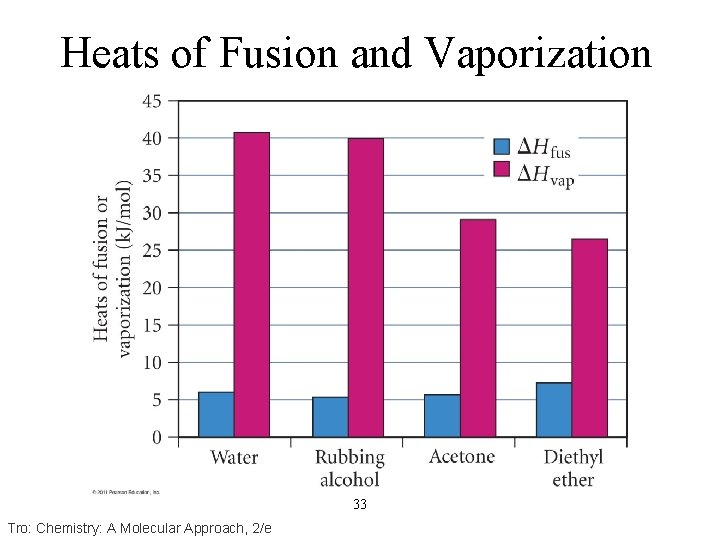
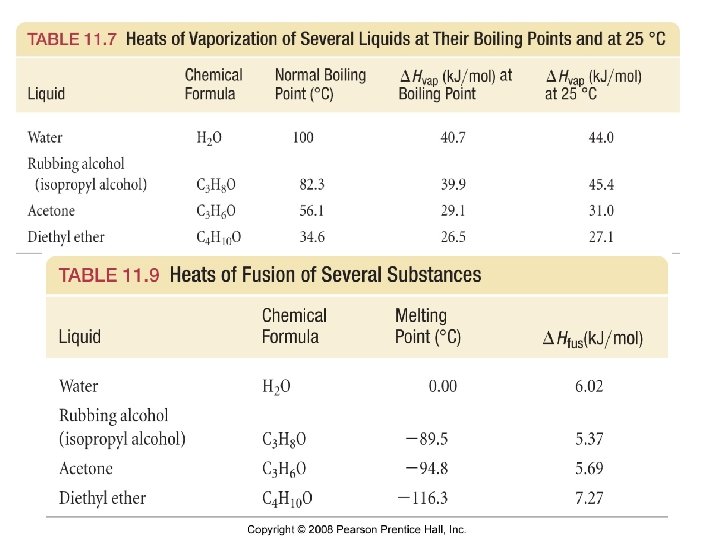
Heats of Fusion and Vaporization 33 Tro: Chemistry: A Molecular Approach, 2/e

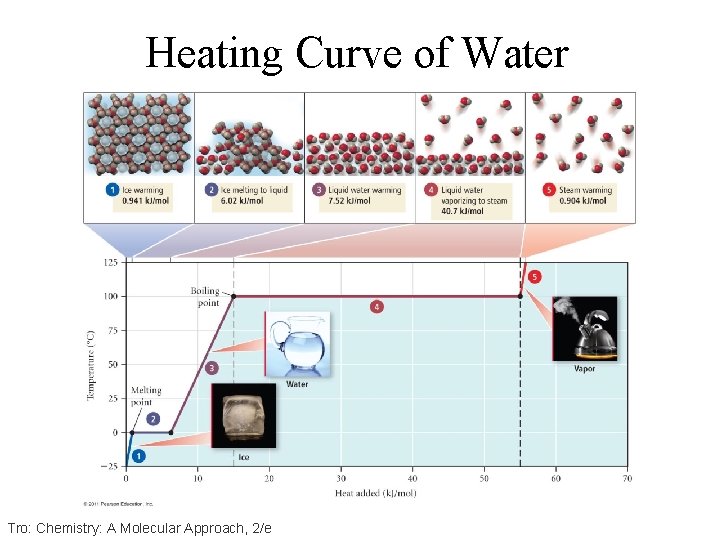
Heating Curve of Water 34 Tro: Chemistry: A Molecular Approach, 2/e


Example – Heating Curve of Water A quantity of ice at 0 ˚C is added to 90. 0 g of water at 80 ˚C. After the ice is melted the temperature of the water is 25 ˚C. How many grams of ice was added? Given that cice is 2. 06 J/g · ˚C, cwater is 4. 184 J/g · ˚C , the enthalpy of fusion of ice is 333 J/g.

Example – Heating Curve of Water If 50. 0 g of ice at -20. 0 ˚C is added to 342. 0 g of water at 86. 0 ˚C, what will the final temperature of the sample be? Given that cice is 2. 06 J/g · ˚C, cwater is 4. 184 J/g · ˚C , the enthalpy of fusion of ice is 333 J/g.


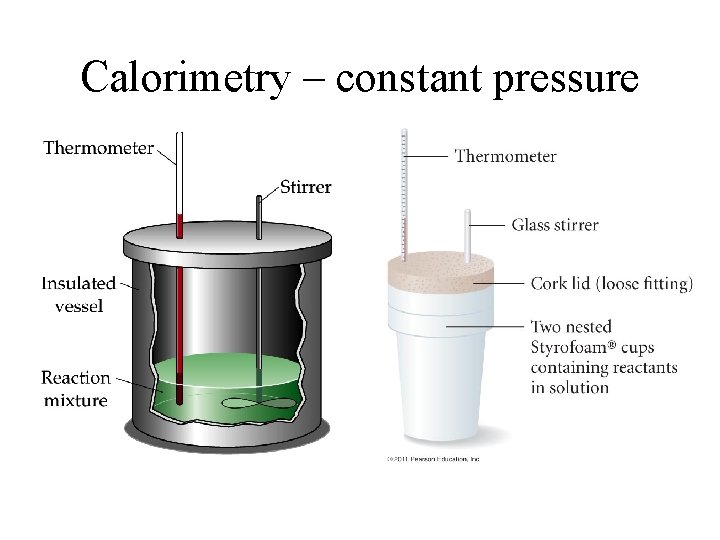
Calorimetry – constant pressure

How can the enthalpy of a reaction be found?

Example – Calorimetry a) A reaction known to release 1. 78 k. J of heat takes place in a calorimeter containing 0. 100 L of solution. The temperature rose by 3. 65 ˚C. b) Next, 50 m. L of hydrochloric acid and 50 m. L of aqueous sodium hydroxide were mixed in the same calorimeter and the temperature rose by 1. 26 ˚C. What is the heat output of the neutralization reaction?

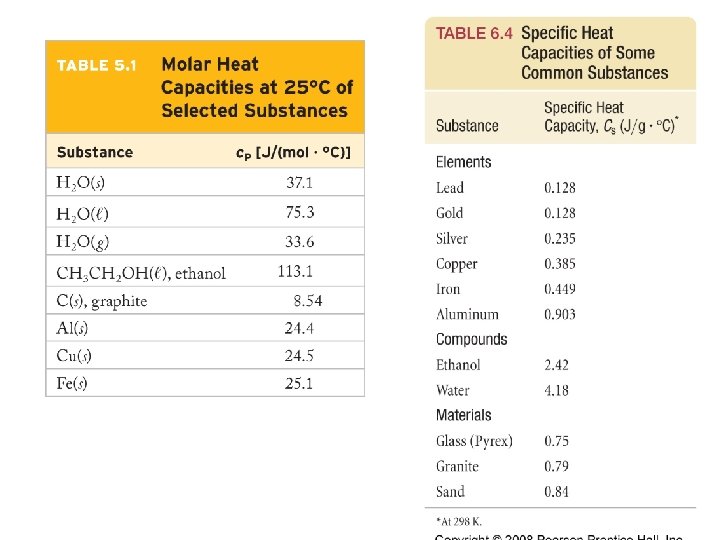
Example – Calorimetry A 55. 0 g piece of metal was heated in boiling water to 99. 8 ˚C and dropped into an insulated beaker with 225 m. L of water (ρ = 0. 997992 g/m. L) at 21. 0 ˚C. The final temperature of the metal and the water is 23. 1 ˚C and cwater is 4. 184 J/g · ˚C. Calculate the specific heat of the metal assuming that no heat was lost to the surroundings.

Example – Calorimetry A 33. 14 g sample of copper (c. Cu = 0. 385 J/g · ˚C) and aluminum (c. Al = 0. 902 J/g · ˚C) was heated to 119. 25 ˚C and dropped into a calorimeter containing 250. 0 g of water at 21. 00 ˚C. The temperature rose to 23. 05 ˚C. Assuming that no heat was lost to the surroundings, what is the mass of copper in the sample?

Bomb Calorimeter – constant volume

Example – Calorimetry Octane, C 8 H 18, a primary constituent of gasoline burns in air. Suppose that a 1. 00 g sample of octane is burned in a calorimeter that contains 1. 20 kg of water. The temperature of the water and the bomb rises from 25. 00 ˚C to 33. 20 ˚C. If the heat capacity of the bomb is 837 J/˚C, cwater is 4. 184 J/g · ˚C, calculate the molar heat of reaction of octane.

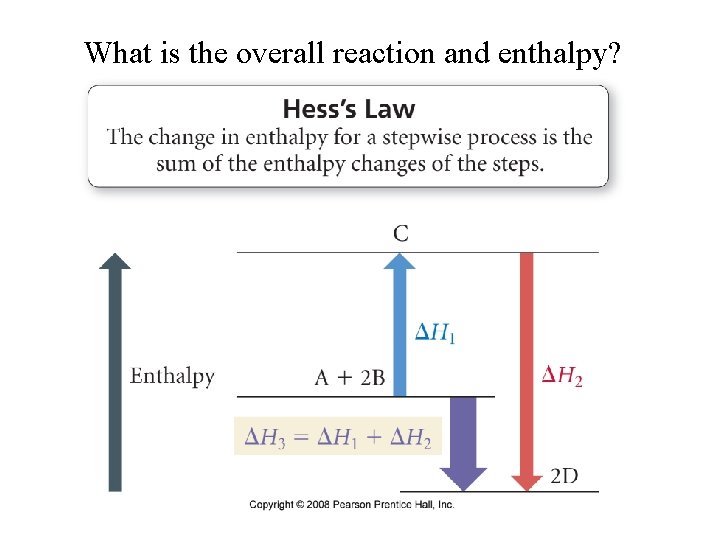
What is the overall reaction and enthalpy?

How can the enthalpy of sublimation be determined?

Example – Hess’ Law Determine the ΔHsublimation of ice to water vapor: H 2 O (s)→ H 2 O (g) Given: H 2 O (s)→ H 2 O (l) ΔHfusion = 6. 02 k. J H 2 O (g)→ H 2 O (l) ΔHvaporization= -40. 7 k. J

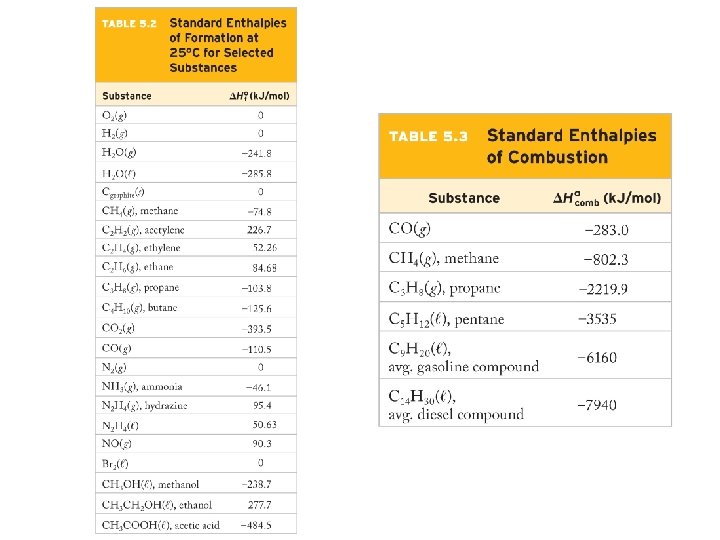
Example – Hess’ Law Two forms of carbon are graphite, the soft, black, slippery material used in “lead” pencils, and as a lubricant for locks and diamond, the beautiful, hard gemstone. Using the enthalpies of combustion for graphite (-394 k. J/mol) and diamond (-396 k. J/mol), calculate ΔH for the conversion of graphite to diamond: C graphite (s)→ C diamond (s) Given: C graphite (s) + O 2 (g) → CO 2 (g) ΔH = -394 k. J C diamond (s) + O 2 (g) → CO 2 (g) ΔH = -396 k. J



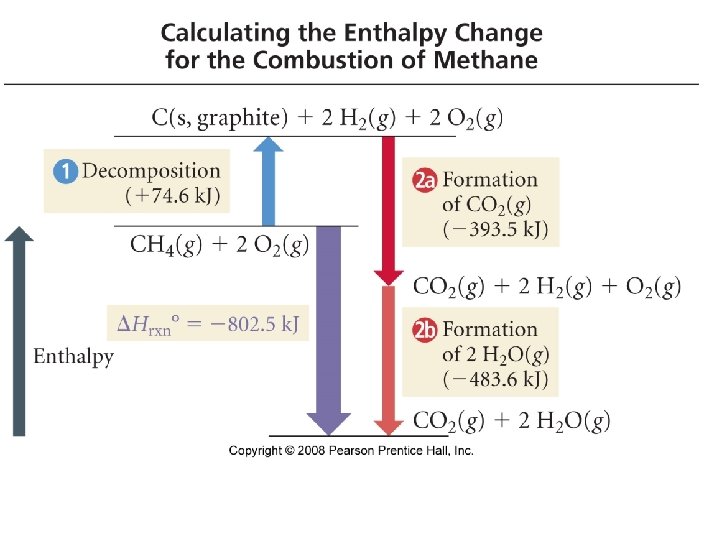
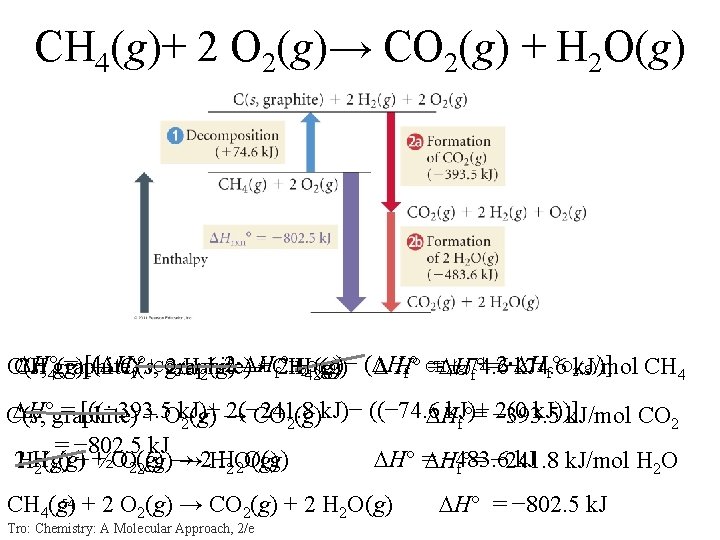
CH 4(g)+ 2 O 2(g)→ CO 2(g) + H 2 O(g) H° 4 graphite) = [( H °HH 24 O(g) )− ( H (g)74. 6 + 2∙ H (g))] CH (g) → C(s, (g) H° = H C(s, 2 graphite) H 2+(g)2∙ H →+ CH 74. 6 CH 4 2(g) 4+ f°+CO f 2 f° CH f°O 2 k. J/mol 2(g) f°= −k. J H°graphite) = [((− 393. 5 2(− 241. 8 k. J)− ((− 74. 6 k. J)+ 2(0 k. J))] C(s, + Ok. J)+ H 2(g) → CO 2(g) f°= − 393. 5 k. J/mol CO 2 = − 802. 5 k. J 2 HH H° = H − 483. 6 k. J/mol H 2 O + +½OO 2(g) → 2 HH 2 O(g) 2(g)→ f°= − 241. 8 54 + 2 O (g) → CO (g) + 2 H O(g) CH 4(g) 2 2 2 Tro: Chemistry: A Molecular Approach, 2/e H° = − 802. 5 k. J

Example – Hess’ Law Calculate the enthalpy change of the formation of methane, CH 4, from solid carbon as graphite and hydrogen gas: C(s) + 2 H 2 (g) → CH 4 (g) Given: C(s) + O 2 (g) → CO 2 (g) ΔH = -393. 5 k. J H 2 (g) + ½ O 2 (g) → H 2 O (l) ΔH = -285. 8 k. J CH 4 (g) + 2 O 2 (g) → CO 2 (g) + 2 H 2 O (l) ΔH = -890. 3 k. J

Example – Hess’ Law Calculate the enthalpy change for S (s) + O 2 (g) → SO 2 (g) Given: 2 SO 2 (g) + O 2 (g) → 2 SO 3 (g) ΔH = -196 k. J 2 S (s) + 3 O 2 (g) → 2 SO 3 (g) ΔH = -790 k. J

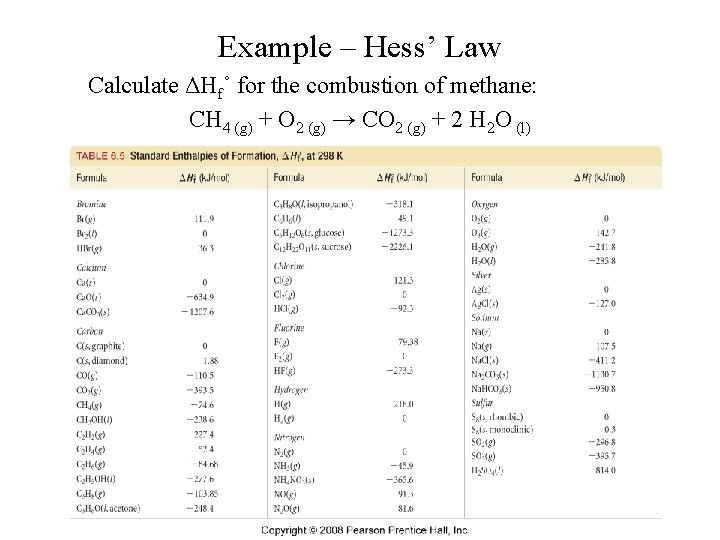
Example – Hess’ Law Calculate ΔHf˚ for the combustion of methane: CH 4 (g) + O 2 (g) → CO 2 (g) + 2 H 2 O (l)

Example – Hess’ Law Benzene, C 6 H 6, is an important hydrocarbon. Calculate its enthalpy of combustion (ΔH˚): C 6 H 6 (l) + O 2 (g) → 6 CO 2 (g) + 3 H 2 O (l) Given: ΔHf˚ [C 6 H 6 (l)] = +49. 0 k. J/mol ΔHf˚ [CO 2 (g)] = -393. 5 k. J/mol ΔHf˚ [H 2 O (l)] = -285. 8 k. J/mol ΔHf˚ [O 2 (g)] = 0 k. J/mol

Example – Hess’ Law Benzene, C 6 H 6, is an important hydrocarbon. Calculate its enthalpy of combustion (ΔH˚): C 6 H 6 (l) + O 2 (g) → 6 CO 2 (g) + 3 H 2 O (l) Given: 6 C (s) + 3 H 2 (g) C 6 H 6 (l) ΔH = 49. 0 k. J C (s) + O 2 (g) CO 2 (g) ΔH = -393. 5 k. J H 2 (g) + ½ O 2 (g) H 2 O (l) ΔH = -285. 8 k. J

Example – Hess’ Law Calculate the enthalpy of formation of the fermentation of glucose to ethanol: C 6 H 12 O 6 (s) → 2 C 2 H 5 OH (l) + 2 CO 2 (g) Given: ΔHf˚ [C 6 H 12 O 6 (s)] = -1, 260 k. J/mol ΔHf˚ [CO 2 (g)] = -393. 5 k. J/mol ΔHf˚ [C 2 H 5 OH (l)] = -277. 7 k. J/mol

Example – Hess’ Law Calculate ΔHf˚ (in kilojoules) for the synthesis of lime (calcium oxide) from limestone (calcium carbonate), an important step in the manufacture of cement. Ca. CO 3 (s) → Ca. O (s) + CO 2 (g) Given: ΔHf˚ [Ca. CO 3 (s)] = -1, 206. 9 k. J/mol ΔHf˚ [Ca. O (s)] = -635. 1 k. J/mol ΔHf˚ [CO 2 (g)] = -393. 5 k. J/mol

Example – Hess’ Law Nitroglycerin is a powerful explosive, giving four different gases when detonated: 2 C 3 H 5(NO 3)3 (l) Given: 3 N 2 (g) + ½ O 2 (g) + 6 CO 2 (g) + 5 H 2 O (g) ΔHf˚ [C 3 H 5(NO 3)3 (l)] = -364 k. J/mol ΔHf˚ [CO 2 (g)] = -393. 5 k. J/mol ΔHf˚ [H 2 O (g)] = -241. 8 k. J/mol ΔHf˚ [O 2 (g)] = 0 k. J/mol ΔHf˚ [N 2 (g)] = 0 k. J/mol Calculate the energy liberated when 7. 00 g is detonated.