Chapter 5 Thermochemistry 2015 Pearson Education Inc Energy























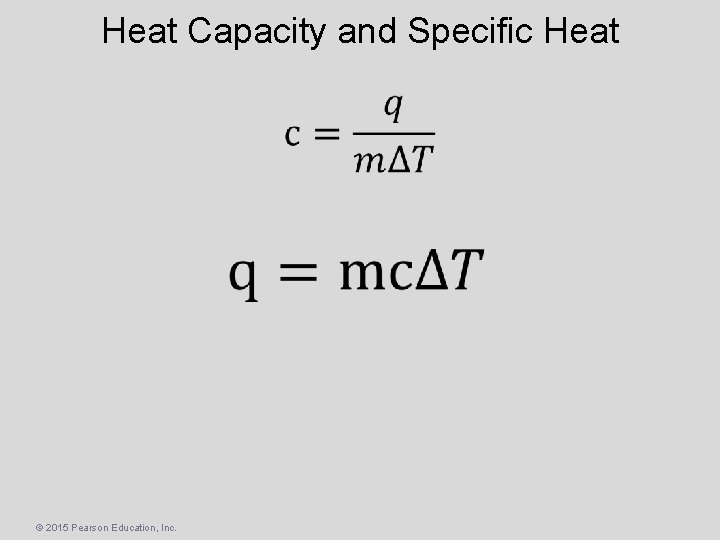
















- Slides: 45

Chapter 5 Thermochemistry © 2015 Pearson Education, Inc.

Energy • Energy is the ability to do work or transfer heat. – Energy used to cause an object that has mass to move is called work (W). – Energy used to cause the temperature of an object to rise is called heat (Q). • Thermodynamics – The study of energy transformations, and thermochemistry, which applies the field to chemical reactions, specifically. © 2015 Pearson Education, Inc.

Energy • Kinetic energy – Energy an object possesses by virtue of its motion • Potential energy – Energy an object possesses by virtue of its potential to do work. • (Gravitational potential energy) © 2015 Pearson Education, Inc.

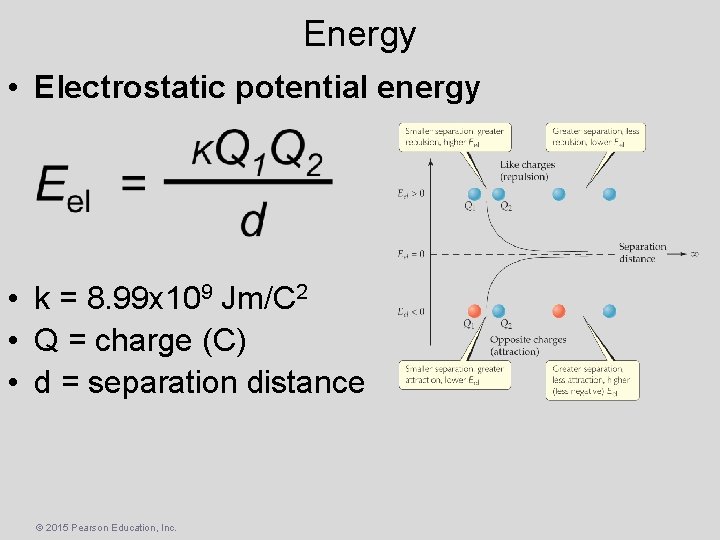
Energy • Electrostatic potential energy • k = 8. 99 x 109 Jm/C 2 • Q = charge (C) • d = separation distance © 2015 Pearson Education, Inc.

Units of Energy • The SI unit of energy is the joule (J): • An older, non-SI unit is still in widespread use, the calorie (cal): 1 cal = 4. 184 J 1 kcal = 4184 J © 2015 Pearson Education, Inc.

System and Surroundings • The system – The molecules we want to study • The surroundings – Everything else © 2015 Pearson Education, Inc.

Transferring Energy: Work and Heat • Heat flows from warmer objects to cooler objects. © 2015 Pearson Education, Inc.

Calculating Energy Changes • What is the kinetic energy, in J, of a mole of Ar atoms moving at 650 m ⁄ s? (Hint: 1 amu = 1. 66 × 10− 27 kg. ) 1. Find the mass in kg 2. Solve for kinetic energy © 2015 Pearson Education, Inc.

First Law of Thermodynamics • “Energy is neither created nor destroyed, it is only transferred from one form to another. ” – The total energy of the universe is a constant; if the system loses energy, it must be gained by the surroundings, and vice versa. Thermodynamics © 2015 Pearson Education, Inc.

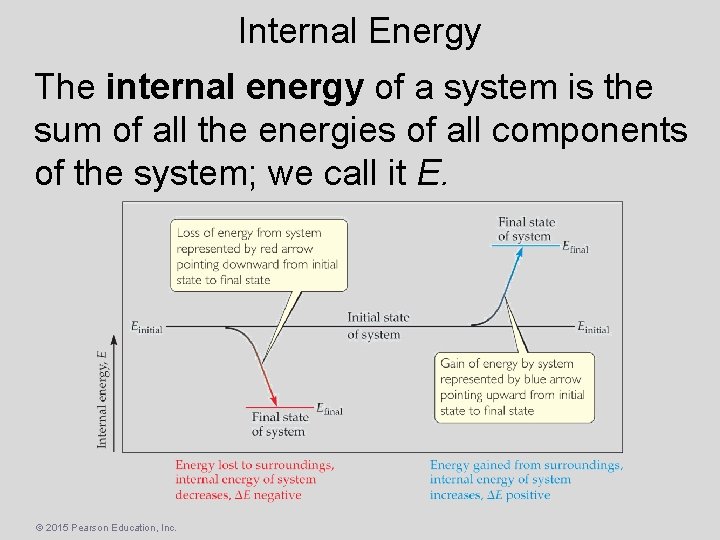
Internal Energy The internal energy of a system is the sum of all the energies of all components of the system; we call it E. © 2015 Pearson Education, Inc.

Internal Energy The change in internal energy is the final energy of the system minus the initial energy of the system: E = Efinal − Einitial © 2015 Pearson Education, Inc.

Internal Energy • Endergonic Reactions • Exergonic Reactions © 2015 Pearson Education, Inc.

Relating E to Heat and Work • When energy is exchanged between the system and the surroundings, it is exchanged as either heat (q) or work (w). © 2015 Pearson Education, Inc.

Endothermic and Exothermic Processes • Endothermic Reactions » System Gains Heat (From Where? ) • Exothermic Reactions » System Loses Heat © 2015 Pearson Education, Inc.

Calculating Heat and Work What is the change in the internal energy for a process in which a system absorbs 140 J of of heat from the surroundings and does 85 J of work on the surroundings? © 2015 Pearson Education, Inc.

State Functions • Internal energy is a state function. – It depends only on the present state of the system, not on the path by which the system arrived at that state. © 2015 Pearson Education, Inc.

State Functions • Q and W are not state functions. © 2015 Pearson Education, Inc.

Enthalpy • Enthalpy (H) – The internal energy plus the product of pressure and volume: © 2015 Pearson Education, Inc.

Pressure-Volume Work © 2015 Pearson Education, Inc.

Calculating Work What is the work done if the volume of a system contracts from 1. 55 L to 0. 85 L at a constant pressure of 0. 985 atm? © 2015 Pearson Education, Inc.

Calculating Work What is the work done if the volume of a system contracts from 1. 55 L to 0. 85 L at a constant pressure of 0. 985 atm? © 2015 Pearson Education, Inc.

Enthalpy Change • Endothermic = + H • Exothermic = - H © 2015 Pearson Education, Inc.

Enthalpy Change Usually in an open container the only work done is by a gas pushing on the surroundings (or by the surroundings pushing on the gas). © 2015 Pearson Education, Inc.

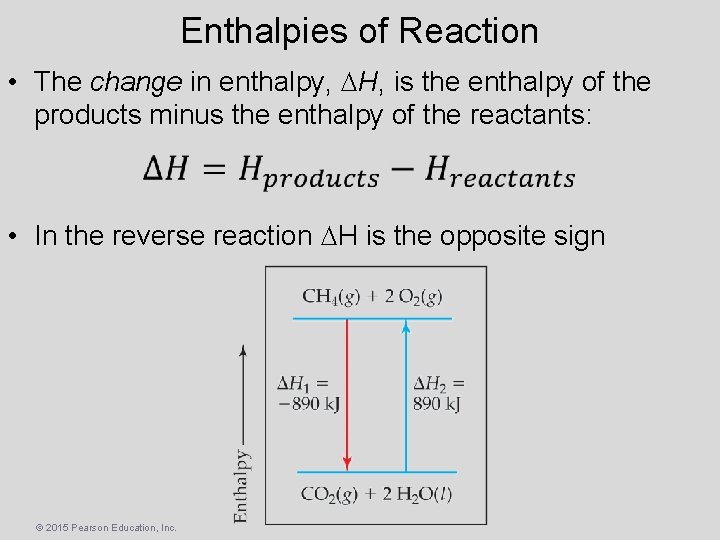
Enthalpies of Reaction • The change in enthalpy, H, is the enthalpy of the products minus the enthalpy of the reactants: • In the reverse reaction H is the opposite sign © 2015 Pearson Education, Inc.

Calculating Enthalpies of Reaction • Hydrogen peroxide decomposes to water and oxygen. • Calculate the quantity of heat released when 5. 00 g of H 2 O 2 decomposes at constant pressure. © 2015 Pearson Education, Inc.

Calorimetry • Since we cannot know the exact enthalpy of the reactants and products, we measure H through calorimetry, the measurement of heat flow. © 2015 Pearson Education, Inc.

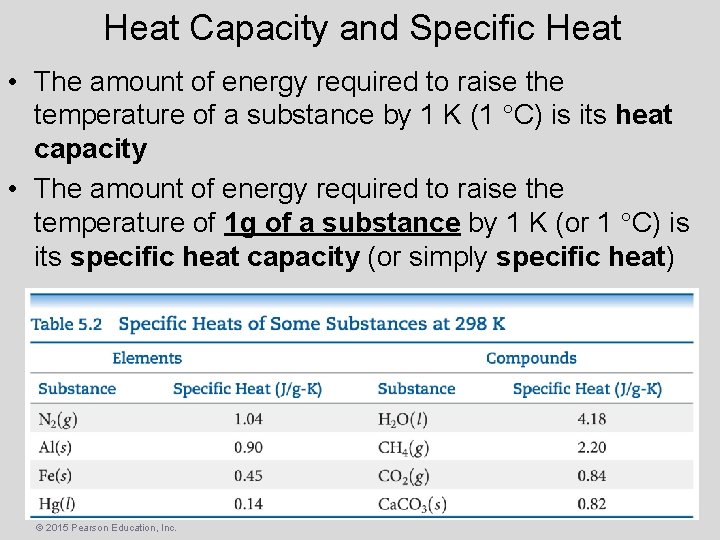
Heat Capacity and Specific Heat • The amount of energy required to raise the temperature of a substance by 1 K (1 C) is its heat capacity • The amount of energy required to raise the temperature of 1 g of a substance by 1 K (or 1 C) is its specific heat capacity (or simply specific heat) © 2015 Pearson Education, Inc.
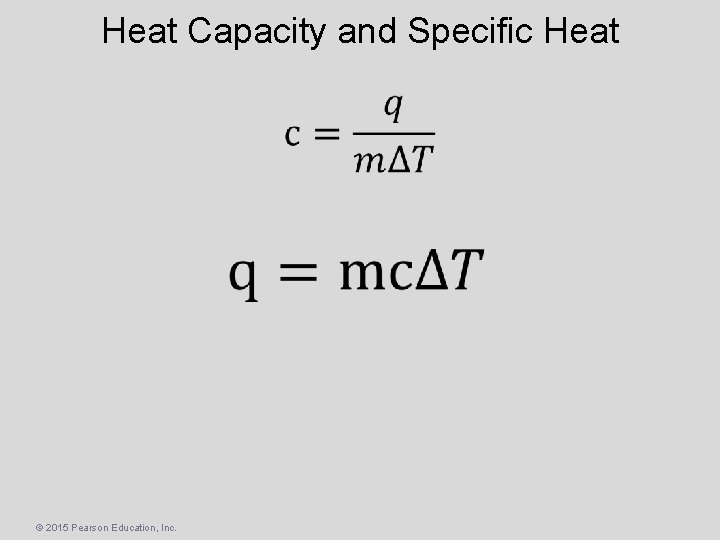
Heat Capacity and Specific Heat © 2015 Pearson Education, Inc.

Calculating Specific Heat • Large beds of rocks are used in some solar-heated homes to store heat. The specific heat of the rocks is 0. 82 J/g*K. What is the heat absorbed by 50. 0 kg of the rocks if their temperature increases by 12. 0 o. C? © 2015 Pearson Education, Inc.

Calculating Specific Heat • Large beds of rocks are used in some solar-heated homes to store heat. The specific heat of the rocks is 0. 82 J/g*K. What temperature change would these rocks undergo if they emitted 450 k. J of heat? © 2015 Pearson Education, Inc.

Constant Pressure Calorimetry • By carrying out a reaction in aqueous solution in a simple calorimeter, the heat change for the system can be found by measuring the heat change for the water in the calorimeter. • The specific heat for water is well known (4. 184 J/g∙K). • We can calculate H for the reaction with this equation: q = m Cs T © 2015 Pearson Education, Inc.

Calculating Specific Heat • When 50. 0 m. L of 0. 100 M Ag. NO 3 and 50 m. L of 0. 100 M HCl are mixed in a calorimeter the temperature of the mixture increases from 22. 30 to 23. 11 o. C. • Calculate H for this reaction in k. J/mol Ag. NO 3 assuming the combined solution has a mass of 100. 0 g. © 2015 Pearson Education, Inc.

Calculating Specific Heat • When 50. 0 m. L of 0. 100 M Ag. NO 3 and 50 m. L of 0. 100 M HCl are mixed in a calorimeter the temperature of the mixture increases from 22. 30 to 23. 11 o. C. • Calculate H for this reaction in k. J/mol Ag. NO 3 assuming the combined solution has a mass of 100. 0 g. © 2015 Pearson Education, Inc.

Constant Volume Calorimetry • Reactions can be carried out in a sealed “bomb” such as this one. • The heat absorbed (or released) by the water is a very good approximation of the enthalpy change for the reaction. © 2015 Pearson Education, Inc.

Hess’s Law • If a reaction is carried out in a series of steps, H for the overall reaction will be equal to the sum of the enthalpy changes for the individual steps. • Because H is a state function, the total enthalpy change depends only on the initial state (reactants) and the final state (products) of the reaction. Hess's Law © 2015 Pearson Education, Inc.

Hess’s Law • The enthalpy of reaction for the combustion of C to CO 2 is − 393. 5 k. J ⁄ mol C, and the enthalpy for the combustion of CO to CO 2 is − 283. 0 k. J ⁄ mol CO: C(s) + O 2(g) → CO 2(g) ΔH = − 393. 5 k. J CO(g) + O 2(g) → CO 2(g) ΔH = − 283. 0 k. J Using these data, calculate the enthalpy for the combustion of C to CO: C(s) + O 2(g) → CO(g) ΔH = ? © 2015 Pearson Education, Inc.

Hess’s Law C(s) + O 2(g) → CO 2(g) ΔH = − 393. 5 k. J CO(g) + ½ O 2(g) CO 2(g) ΔH = +283. 0 k. J (-393. 5 k. J) + (+283. 0 k. J) = -110. 5 k. J C(s) + ½ O 2(g) → CO(g) ΔH = -110. 5 k. J © 2015 Pearson Education, Inc.

Hess’s Law Calculate ΔH for: 2 NO(g) + O 2(g) → N 2 O 4(g) → 2 NO 2(g) 2 NO(g) + O 2(g) → 2 NO 2(g) © 2015 Pearson Education, Inc. ΔH = +57. 9 k. J ΔH = − 113. 1 k. J

Hess’s Law Calculate ΔH for: 2 NO(g) + O 2(g) → N 2 O 4(g) 2 NO(g) + O 2(g) → 2 NO 2(g) N 2 O 4(g) 2 NO 2(g) ΔH = − 113. 1 k. J ΔH = -57. 9 k. J ΔH = − 171. 0 k. J © 2015 Pearson Education, Inc.

Hess’s Law • Carbon occurs in two forms, graphite and diamond. The enthalpy of the combustion of graphite is − 393. 5 k. J ⁄ mol, and that of diamond is − 395. 4 k. J ⁄ mol: C(graphite) + O 2(g) → CO 2(g) ΔH = − 393. 5 k. J C(diamond) + O 2(g) → CO 2(g) ΔH = − 395. 4 k. J • Calculate ΔH for the conversion of graphite to diamond: C(graphite) → C(diamond) © 2015 Pearson Education, Inc.

Hess’s Law C(graphite) + O 2(g) → CO 2(g) ΔH = − 393. 5 k. J C(diamond) + O 2(g) → CO 2(g) ΔH = − 395. 4 k. J • Calculate ΔH for the conversion of graphite to diamond: C(graphite) + O 2(g) → CO 2(g) C(diamond) + O 2(g) CO 2(g) © 2015 Pearson Education, Inc. ΔH = − 393. 5 k. J ΔH = +395. 4 k. J ΔH = +1. 9 k. J

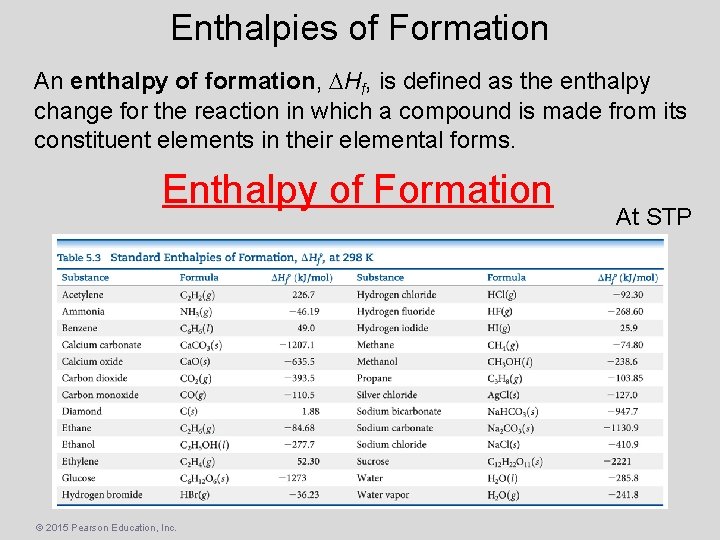
Enthalpies of Formation An enthalpy of formation, Hf, is defined as the enthalpy change for the reaction in which a compound is made from its constituent elements in their elemental forms. Enthalpy of Formation © 2015 Pearson Education, Inc. At STP

Calculation of H using Values from the Standard Enthalpy Table C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) C 3 H 8= -103. 85 k. J/mol CO 2 = -393. 5 k. J/mol H 2 O = -285. 8 k. J/mol O 2 = 0 final initial H = [3(CO 2) + 4(H 2 O)] – [1(C 3 H 8) + 5(O 2)] H = [3(− 393. 5 k. J) + 4(− 285. 8 k. J)] – [1(− 103. 85 k. J) + 5(0 k. J)] = [(− 1180. 5 k. J) + (− 1143. 2 k. J)] – [(− 103. 85 k. J) + (0 k. J)] = (− 2323. 7 k. J) – (− 103. 85 k. J) = − 2219. 9 k. J © 2015 Pearson Education, Inc.

Energy in Foods and Fuels Most of the fuel in the food we eat comes from carbohydrates and fats. © 2015 Pearson Education, Inc.

Energy in Foods and Fuels Fuel Value from Food Dry red beans contain 62% carbohydrate, 22% protein, and 1. 5% fat. Estimate the fuel value of these beans. Carbs = 17 k. J/g x 0. 62 = 10. 54 k. J/g Protein = 17 k. J/g x 0. 22 = 3. 74 k. J/g Fat = 38 k. J/g x 0. 015 = 0. 57 k. J/g © 2015 Pearson Education, Inc.