Chapter 5 The Gas Laws 1 Pressure Force















































- Slides: 58

Chapter 5 The Gas Laws 1

Pressure Force per unit area. n Gas molecules fill container. n Molecules move around and hit sides. n Collisions are the force. n Container has the area. n Measured with a barometer. n 2

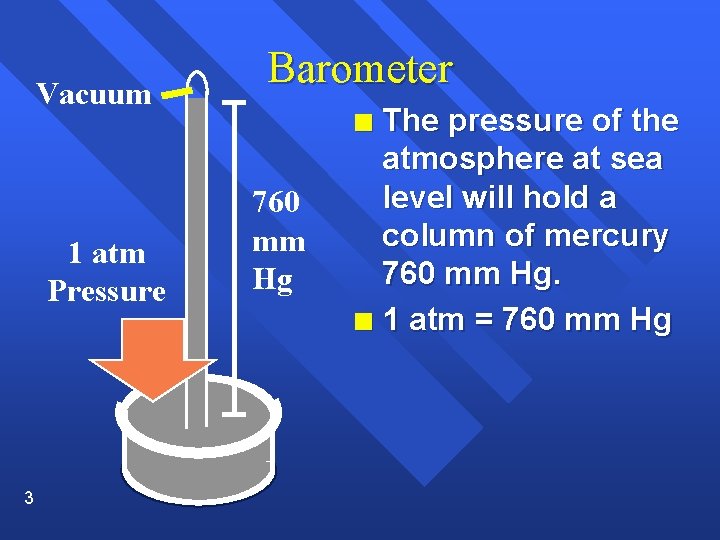
Vacuum Barometer The pressure of the atmosphere at sea level will hold a column of mercury 760 mm Hg. n 1 atm = 760 mm Hg n 1 atm Pressure 3 760 mm Hg

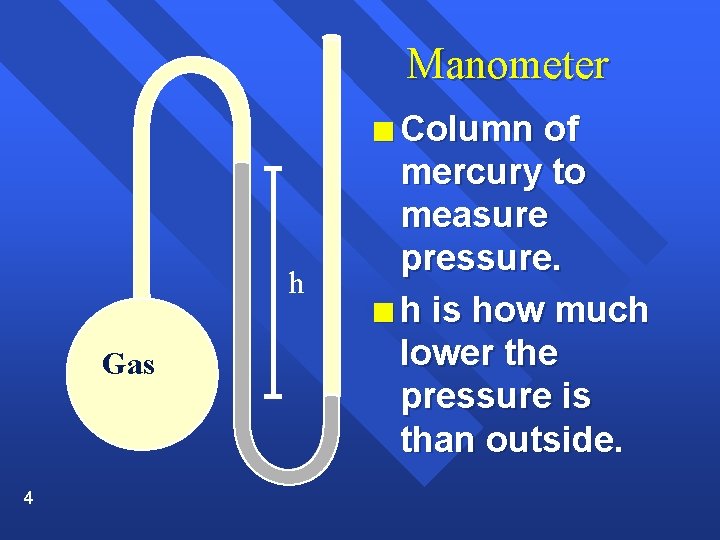
Manometer n Column h Gas 4 of mercury to measure pressure. n h is how much lower the pressure is than outside.

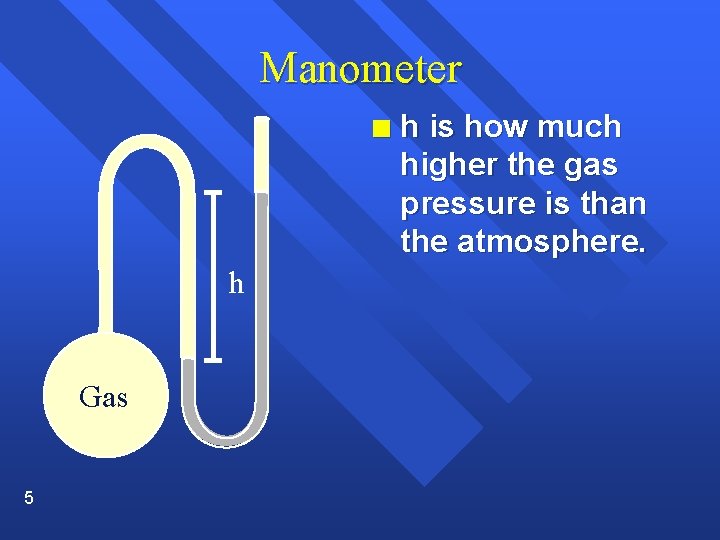
Manometer n h Gas 5 h is how much higher the gas pressure is than the atmosphere.

Units of pressure 1 atmosphere = 760 mm Hg n 1 mm Hg = 1 torr n 1 atm = 101, 325 Pascals = 101. 325 k. Pa n Can make conversion factors from these. n What is 724 mm Hg in k. Pa? n in torr? n in atm? n 6

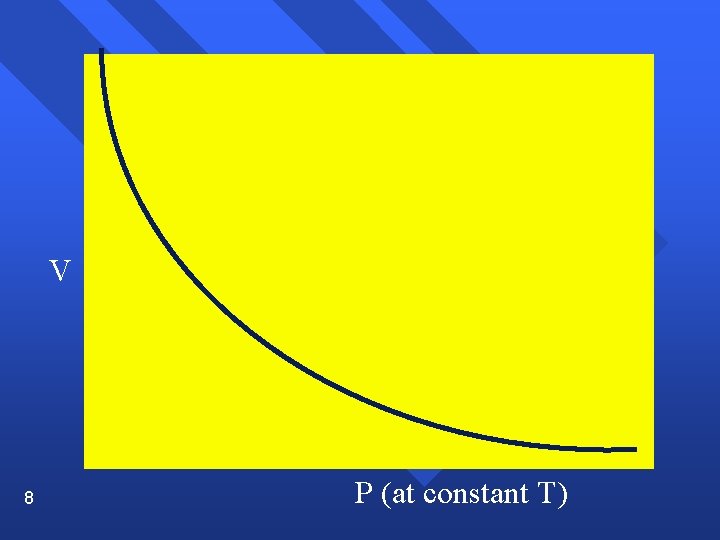
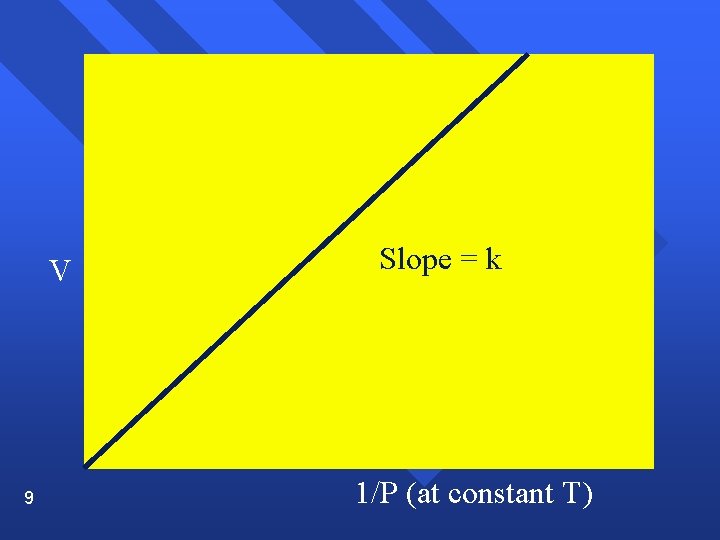
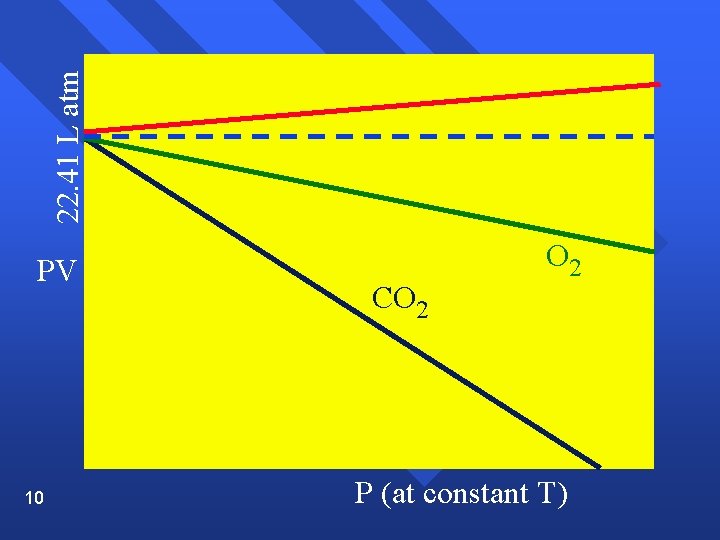
The Gas Laws Boyle’s Law n Pressure and volume are inversely related at constant temperature. n PV= k n As one goes up, the other goes down. n P 1 V 1 = P 2 V 2 n Graphically n 7

V 8 P (at constant T)

V 9 Slope = k 1/P (at constant T)

22. 41 L atm PV 10 CO 2 P (at constant T)

Examples 20. 5 L of nitrogen at 25ºC and 742 torr are compressed to 9. 8 atm at constant T. What is the new volume? n 30. 6 m. L of carbon dioxide at 740 torr is expanded at constant temperature to 750 m. L. What is the final pressure in k. Pa? n 11

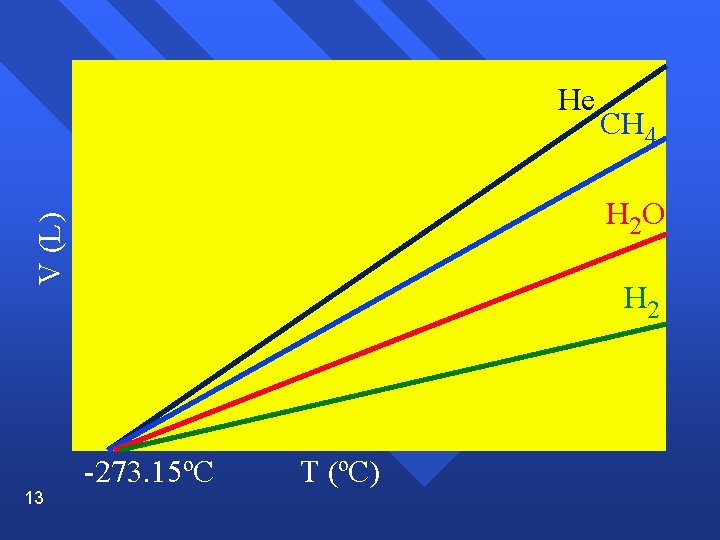
Charle’s Law Volume of a gas varies directly with the absolute temperature at constant pressure. n V = k. T (if T is in Kelvin) n n V 1 = V 2 T 1 = T 2 n Graphically 12

He H 2 O V (L) 13 CH 4 H 2 -273. 15ºC T (ºC)

Examples n 14 What would the final volume be if 247 m. L of gas at 22ºC is heated to 98ºC , if the pressure is held constant?

Examples n 15 At what temperature would 40. 5 L of gas at 23. 4ºC have a volume of 81. 0 L at constant pressure?

Avogadro's Law Avagadro’s n At constant temperature and pressure, the volume of gas is directly related to the number of moles. n V = k n (n is the number of moles) n n V 1 = V 2 n 1 = n 2 16

Gay- Lussac Law At constant volume, pressure and absolute temperature are directly related. n. P=k. T n n P 1 = P 2 T 1 = T 2 17

Combined Gas Law n If the moles of gas remains constant, use this formula and cancel out the other things that don’t change. n P 1 V 1 = P 2 V 2 T 1 T 2. 18

Examples n 19 A deodorant can has a volume of 175 m. L and a pressure of 3. 8 atm at 22ºC. What would the pressure be if the can was heated to 100. ºC?

Ideal Gas Law PV = n. RT n V = 22. 41 L at 1 atm, 0ºC, n = 1 mole, what is R? n R is the ideal gas constant. n R = 0. 08306 L atm/ mol K n Tells you about a gas is NOW. n The other laws tell you about a gas when it changes. n 20

Ideal Gas Law An equation of state. n Independent of how you end up where you are at. Does not depend on the path. n Given 3 you can determine the fourth. n An Empirical Equation - based on experimental evidence. n 21

Ideal Gas Law A hypothetical substance - the ideal gas n Think of it as a limit. n Gases only approach ideal behavior at low pressure (< 1 atm) and high temperature. n Use the laws anyway, unless told to do otherwise. n They give good estimates. n 22

Examples A 47. 3 L container containing 1. 62 mol of He is heated until the pressure reaches 1. 85 atm. What is the temperature? n Kr gas in a 18. 5 L cylinder exerts a pressure of 8. 61 atm at 24. 8ºC What is the mass of Kr? n 23

Gas Density and Molar Mass D = m/V n Let M stand for molar mass n M = m/n n n= PV/RT n. M= m PV/RT n M = m. RT = m RT = DRT PV V P P n 24

Examples n 25 What is the density of ammonia at 23ºC and 735 torr?

Gases and Stoichiometry Reactions happen in moles n At Standard Temperature and Pressure (STP, 0ºC and 1 atm) 1 mole of gas occuppies 22. 42 L. n If not at STP, use the ideal gas law to calculate moles of reactant or volume of product. n 26

Examples n Mercury can be achieved by the following reaction What volume of oxygen gas can be produced from 4. 10 g of mercury (II) oxide at STP? n At 400. ºC and 740 torr? 27

Dalton’s Law The total pressure in a container is the sum of the pressure each gas would exert if it were alone in the container. n The total pressure is the sum of the partial pressures. n PTotal = P 1 + P 2 + P 3 + P 4 + P 5. . . n For each P = n. RT/V n 31

Dalton's Law PTotal = n 1 RT + n 2 RT + n 3 RT +. . . V V V n In the same container R, T and V are the same. n 32 n PTotal = (n 1+ n 2 + n 3+. . . )RT V n PTotal = (n. Total)RT V

The mole fraction n Ratio of moles of the substance to the total moles. n symbol is Greek letter chi n 33 c 1 = n 1 = P 1 n. Total PTotal c

Examples The partial pressure of nitrogen in air is 592 torr. Air pressure is 752 torr, what is the mole fraction of nitrogen? n What is the partial pressure of nitrogen if the container holding the air is compressed to 5. 25 atm? n 34

Vapor Pressure Water evaporates! n When that water evaporates, the vapor has a pressure. n Gases are often collected over water so the vapor. pressure of water must be subtracted from the total pressure. n It must be given. n 36

Example n N 2 O can be produced by the following reaction what volume of N 2 O collected over water at a total pressure of 94 k. Pa and 22ºC can be produced from 2. 6 g of NH 4 NO 3? ( the vapor pressure of water at 22ºC is 21 torr) 37

Kinetic Molecular Theory tells why the things happen. n explains why ideal gases behave the way they do. n Assumptions that simplify theory, but don’t work in real gases. 1 The particles are so small we can ignore their volume. 2 The particles are in constant motion and their collisions cause pressure. n 38

Kinetic Molecular Theory 3 The particles do not affect each other, neither attracting or repelling. 4 The average kinetic energy is proportional to the Kelvin temperature. n Appendix 2 shows the derivation of the ideal gas law and the definition of temperature. n We need the formula KE = 1/2 mv 2 39

What it tells us (KE)avg = 3/2 RT n This the meaning of temperature. n u is the particle velocity. n u is the average particle velocity. n u 2 is the average particle velocity squared. n the root mean square velocity is n Ö u 2 40 =u rms

Combine these two equations (KE)avg = NA(1/2 mu 2 ) n (KE)avg = 3/2 RT n 41

Combine these two equations (KE)avg = NA(1/2 mu 2 ) n (KE)avg = 3/2 RT n Where M is the molar mass in kg/mole, and R has the units 8. 3145 J/Kmol. n The velocity will be in m/s 42

Example Calculate the root mean square velocity of carbon dioxide at 25ºC. n Calculate the root mean square velocity of hydrogen at 25ºC. n Calculate the root mean square velocity of chlorine at 25ºC. n 43

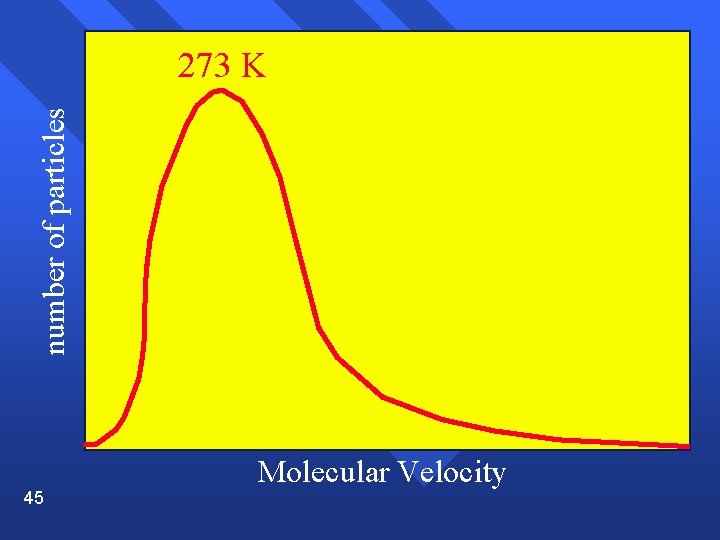
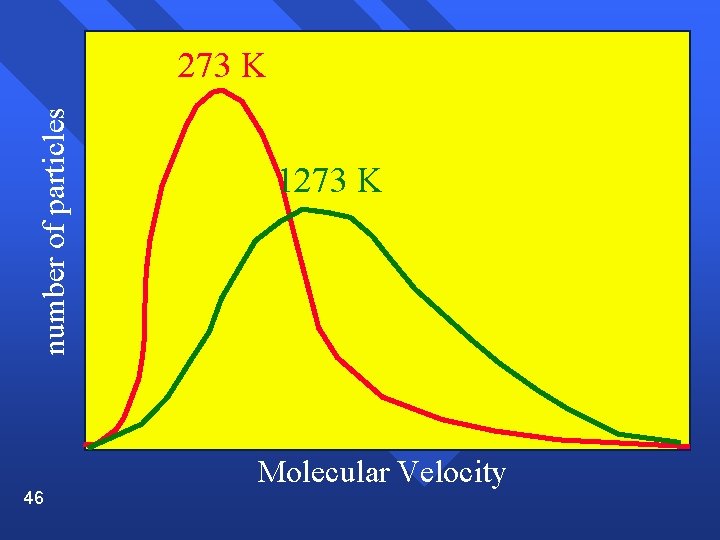
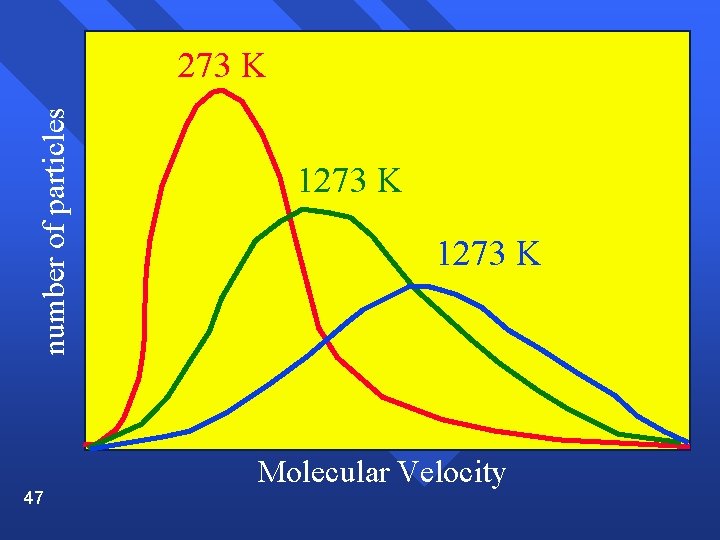
Range of velocities The average distance a molecule travels before colliding with another is called the mean free path and is small (near 10 -7) n Temperature is an average. There are molecules of many speeds in the average. n Shown on a graph called a velocity distribution n 44

number of particles 273 K 45 Molecular Velocity

number of particles 273 K 46 1273 K Molecular Velocity

number of particles 273 K 47 1273 K Molecular Velocity

Velocity Average increases as temperature increases. n Spread increases as temperature increases. n 48

Effusion Passage of gas through a small hole, into a vacuum. n The effusion rate measures how fast this happens. n Graham’s Law the rate of effusion is inversely proportional to the square root of the mass of its particles. n 49

Effusion Passage of gas through a small hole, into a vacuum. n The effusion rate measures how fast this happens. n Graham’s Law the rate of effusion is inversely proportional to the square root of the mass of its particles. n 50

Deriving 51 n The rate of effusion should be proportional to urms n Effusion Rate 1 = urms 1 Effusion Rate 2 = urms 2

Deriving 52 n The rate of effusion should be proportional to urms n Effusion Rate 1 = urms 1 Effusion Rate 2 = urms 2

Diffusion The spreading of a gas through a room. n Slow considering molecules move at 100’s of meters per second. n Collisions with other molecules slow down diffusions. n Best estimate is Graham’s Law. n 53

Examples A compound effuses through a porous cylinder 3. 20 time faster than helium. What is it’s molar mass? n If 0. 00251 mol of NH 3 effuse through a hole in 2. 47 min, how much HCl would effuse in the same time? n A sample of N 2 effuses through a hole in 38 seconds. what must be the molecular weight of gas that effuses in 55 seconds under identical conditions? n 54

Diffusion The spreading of a gas through a room. n Slow considering molecules move at 100’s of meters per second. n Collisions with other molecules slow down diffusions. n Best estimate is Graham’s Law. n 55

Real Gases Real molecules do take up space and they do interact with each other (especially polar molecules). n Need to add correction factors to the ideal gas law to account for these. n 56

Volume Correction n The actual volume free to move in is less because of particle size. More molecules will have more effect. Corrected volume V’ = V - nb b is a constant that differs for each gas. n P’ = n n n 57 n. RT (V-nb)

Pressure correction Because the molecules are attracted to each other, the pressure on the container will be less than ideal n depends on the number of molecules per liter. n since two molecules interact, the effect must be squared. n 58

Pressure correction Because the molecules are attracted to each other, the pressure on the container will be less than ideal n depends on the number of molecules per liter. n since two molecules interact, the effect must be squared. 2 n Pobserved = P’ - a 59 () n V

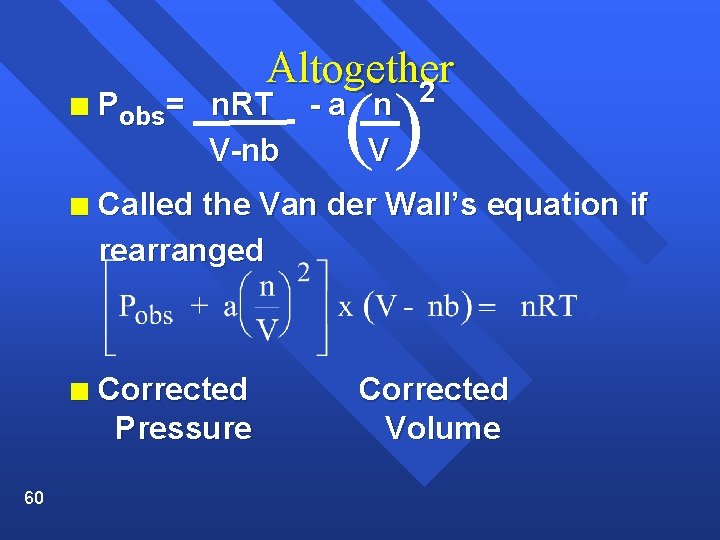
60 Altogether 2 () n Pobs= n. RT - a n V-nb V n Called the Van der Wall’s equation if rearranged n Corrected Pressure Corrected Volume

Where does it come from a and b are determined by experiment. n Different for each gas. n Bigger molecules have larger b. n a depends on both size and polarity. n once given, plug and chug. n 61

Example Calculate the pressure exerted by 0. 5000 mol Cl 2 in a 1. 000 L container at 25. 0ºC n Using the ideal gas law. n Van der Waal’s equation – a = 6. 49 atm L 2 /mol 2 – b = 0. 0562 L/mol n 62