Chapter 5 The Earths Atmosphere Layers of the





















- Slides: 24

Chapter 5 The Earth’s Atmosphere

Layers of the Earth largest of the inner planets l Gravity strong enough to hold gases. l Lots of spheres l Equator divided the Earth into two hemispheres l Lithosphere- solid surface l land areas of the Earth – 30 % as continents – 70% beneath oceans

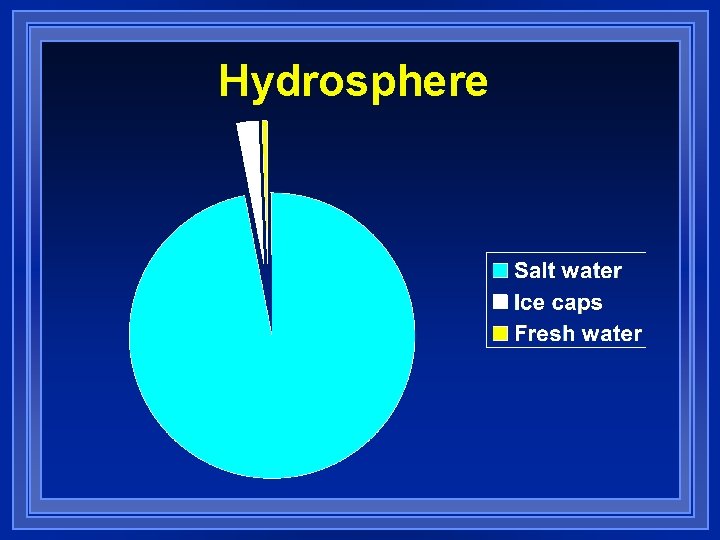
Hydrosphere l Hydro= water l Includes oceans, lakes, rivers, streams, other bodies of water l and ice caps and glaciers l 97 % of water is salt water l 3% fresh water l 85% of fresh water is locked in ice caps. l Not evenly distrubted

Hydrosphere

Atmosphere l The envelope of gases that surround the Earth l Protects the Earth l Provides material necessary for life. l Has changed greatly over time

Past Atmosphere l When Earth formed 4 billion years ago– mainly two deadly gases- methane and ammonia l Methane – CH 4 is natural gas l Ammonia- NH 3 l Also contained water vapor. l 3. 8 billion years ago sunlight powered chemical reactions l Ammonia broke down into nitrogen and hydrogen

Past Atmosphere l Methane broke down and made carbon dioxide and hydrogen l Lightweight hydrogen escaped into space l Leaving lots of nitrogen, some carbon dioxide and water. l First life- organisms deep in the ocean protected from UV l Blue-green bacteria began photosynthesis l Turn carbon dioxide to oxygen

Atmosphere changes l Oxygen rose to upper atmosphere l Chnged to ozone (O 3) l Ozone layer 30 km up l Absorbs UV, protecting the Earth, making life possible l Green plants greatly increased the amount of oxygen l About 600 million years ago amounts of carbon dioxide and oxygen leveled off

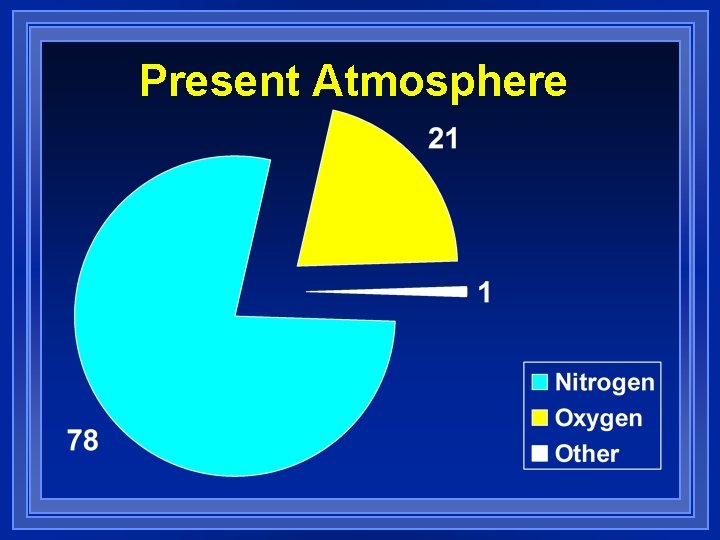
Present Atmosphere l 78 % Nitrogen l 21 % hydrogen l 1 % other – Carbon dioxide – Water vapor – Argon – Trace gases • Kryrpton • Neon • Xenon • Helium

Present Atmosphere

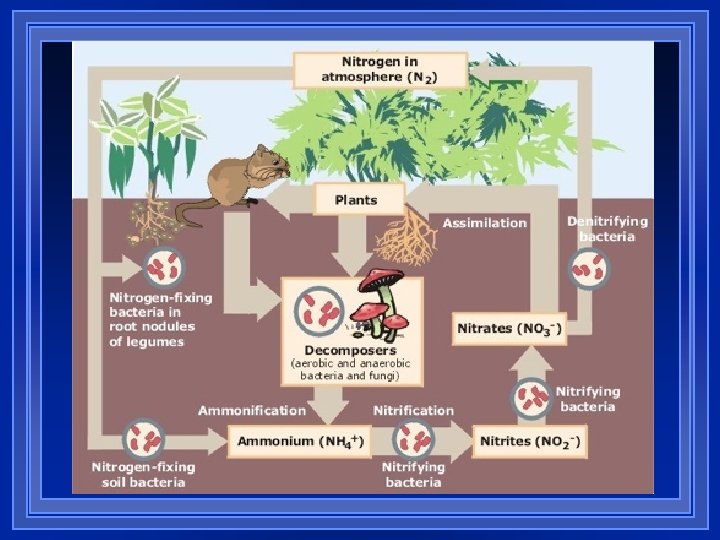
Nitrogen l Most abundant l Needed for proteins l Needed for plan and animal growth l Unreactive, only certain bacteria can change it to nitrates l Some plants can turn nitrates to proteins l Animals eat the plants l Dead things decay back to nitrogen l Nitrogen cycle


Oxygen l Used directly by most plants and animals l Essential for respiration- combine with oxygen to produce energy l Necessary for combustion

Carbon dioxide l Used by plants to make food during photosynthesis l Produced during respiration, decay, and combustion l Burning fossil fuels releases so much it behaves like a greenhouse gas, raising global temperature

Water Vapor l Plays an important role in weather l Heating and cooling of the atmosphere l Energy used to evaporate water keeps ocean air cooler l Solid particles – Salt from ocean water – Dust from volcanoes – From burning fuels

Layers of the Atmosphere l Atmosphere varies as you increase the height – In composition – In Temperature – In Pressure l Separated into layers by temperature l Push of gravity on air causes air pressure

Troposphere l Layer closest to Earth – At equator- 17 km thick – At poles 6 - 8 km thick l Sunlight heats the ground l Then the ground heats the air. l Warm air rises and cold air sinks l Form convection currents which carry heat up into atmosphere

Troposphere l Temperature decreases 6. 5°C for every kilometer l Until you get to 12 km, then temperature is constant l Layer is called the tropopause l Separates from next layer

Stratosphere l From the Troposphere to 50 km l Lower stratosphere very cold near -60°C l In lower stratosphere strong (320 km/hr) eastward blows winds called the jet stream l A special layer of ozone forms in the stratosphere l Protects Earth from ultraviolet which harms living things and causes skin cancer and sunburns

Stratosphere l Upper atmosphere warms as it absorbs ultraviolet l Warms to 18 °C l Highest temperature region is called the stratopause l Separates form next layer

Mesosphere l Above stratosphere temperature drops to about -100 °C l From 50 -80 km above Earth l Coldest region of the atmosphere l Protects from meteoroids. l This is where most burn up

Thermosphere l From 80 km up l Very thin atmosphere l High temperatures up to 2000 °C l From absorbing Ultraviolet light l The high temperature would not be felt because particles are so far apart. l Thermosphere broken into layers

Ionosphere l Lowest layer of thermosphere l 80 -550 km l Gas particles absorb x-rays and ultraviolet and become electrically charged ions l AM radio signals bounce off this layer, so they can travel a long distance l Solar flare disturb this layer and cause static in radio signals

The Exosphere l Upper Thermosphere – 550 km up fro thousands of km l Air is very thin l Where artificial satellites orbit