Chapter 5 SECTIONS 3 5 TYPES OF REACTIONS

















- Slides: 18

Chapter 5 SECTIONS 3 -5 TYPES OF REACTIONS

Warm-Up for Thursday, Sept. 18 �Draw a diagram of an endothermic reaction showing how energy in the surrounding environment is different before and after the reaction �Objective: to learn about different types of chemical reactions

Warm-Up for Wednesday, Jan. 7 �What is the difference between an exothermic and endothermic reaction? �Objective: to observe endothermic and exothermic reactions

Warm-Up for Monday, Sept. 22 �Balance the following equation: �HCl + Na. OH ---> Na. Cl + H 2 O �Objective: to learn about the different types of reactions

Warm-Up for Thursday, March 26 Balance the following: Mg. Cl 2(aq) + Ag. NO 3(aq) ----> Mg(NO 3)2(aq) +Ag. Cl(s) Objective: To learn about and be able to identify different types of chemical reactions.

Warm-Up for Wednesday, Sept. 30 Identify the reaction: Mg. Cl 2(aq) + Ag. NO 3(aq) ----> Mg(NO 3)2(aq) +Ag. Cl(s) Objective: To learn about and be able to identify different types of chemical reactions.

Warm-Up for Wed. March 26 Balance the following: Mg. Cl 2(aq) + Ag. NO 3(aq) ----> Mg(NO 3)2(aq) +Ag. Cl(s) Objective: To learn about and be able to identify different types of chemical reactions.

Warm-Up for Thursday, March 31 What are your plans for spring break?

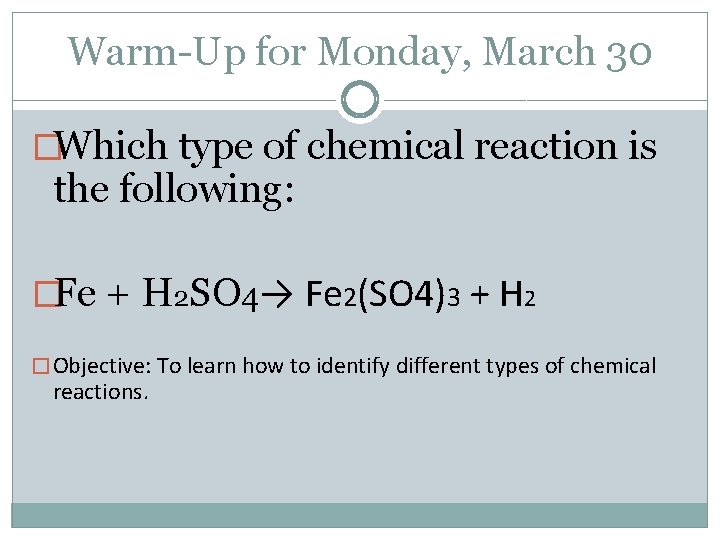
Warm-Up for Monday, March 30 �Which type of chemical reaction is the following: �Fe + H 2 SO 4→ Fe 2(SO 4)3 + H 2 � Objective: To learn how to identify different types of chemical reactions.

Warm-Up for Monday, Sept. 28 �Balance the following equation: C 6 H 14 + O 2 → CO 2 + H 2 O Objective: To learn about different types of chemical reactions.

Combination Reactions �Synthesis Reaction: chemical reactions in which two elements or compounds form one product. 4 Al(s) + 3 O 2 (g) → 2 Al 2 O 3 (s)

Decomposition Reactions �Decomposition Reaction: chemical reaction in which one compound breaks down into two or more smaller products. 2 Al 2 O 3(s)→ 4 Al(s) + 3 O 2(g)

Combustion Reaction �Combustion Reaction: a chemical reaction in which a compound or element burns in the presence of oxygen. Oxygen is always a reactant. At least one product contains oxygen. 4 K+O 2→ 2 K 2 O

Combustion Reaction when an element rapidly combines with oxygen to form an oxide C 3 H 8+5 O 2→ 3 CO 2+4 H 2 O

Single Replacement Reaction �A single replacement reaction: chemical reaction in which an element takes the place of another element in a compound. 3 Na + Al. Cl 3 → Al + 3 Na. Cl

Double Replacement Reaction �Double replacement reaction: Reaction in which the positive ions of two ionic compounds trade places. The result is two new compounds Ba. Cl 2(aq) + Na 2 SO 4(aq)→Ba. SO 4(s) + 2 Na. Cl(aq)

Acid Base Reactions �Acid Base Reaction: Chemical reaction in which an acid reacts with a base to produce water and a salt Vinegar and baking soda

Acid Base Reactions �Salt: ionic compound that has a base for a cation and an acid for an anion �Acid: produces H+ ions when dissolved in water �Base: produces OH- ions when dissolved in water �H 2 SO 4 (aq) + Ca(OH)2(aq) → 2 H 2 O (l) + Ca. SO 4 (s) �Acid Base Water Salt