Chapter 5 Oxygen and Sulfur Table of Contents
Chapter 5 Oxygen and Sulfur Table of Contents Oxygen Introduction 1. Oxygen 2. Occurrence of Oxygen 3. Chemical Properties of Oxygen 4. Compounds of Oxygen 5. Uses of Oxygen 6. Sulfur 7. Occurrence of Sulfur

Chapter 5 Oxygen and Sulfur Selenium Table of Contents 8. Chemical Properties of Sulfur 9. Compounds of Sulfur 10. Uses of Sulfur and Sulfuric Acid Polonium Tellurium

Chapter 5 Oxygen and Sulfur Warm Up • Why is oxygen is very important element for living organisms? What are the functions of it? • What is the most important compound of oxygen? • What are the products made up of sulfur?

Chapter 5 Introduction

Chapter 5 Introduction • They have 6 electrons in their valance shell. • They tend to have -2 oxidation number. • In the group oxygen (O) and sulfur (S) are nonmetals, selenium (Se), tellurium (Te) are semimetals and polonium (Po) is metal. • They less reactive than halogens in the same periods. • Electro negativity decreases and atomic radius increases from top to bottom. • Selenium and tellurium are semiconductors used in the electronics industry. • Polonium is a radioactive element formed by the decay of radium.

Chapter 5 1. Oxygen Isotopes 16 O , 17 O and 18 O Discovery By Priestley in England in 1774. Earth`s crust abundance 46% Common oxidation num. -2 and -1 Electron configuration [He]2 s 22 p 4 Physical state gas Melting point -218. 30 C Color colorless Boiling point -1830 C Density 1. 43 x 10 -3 g/L

Chapter 5 1. Oxygen • Colorless, odorless, tasteless, heavier gas than air. • Oxygen means acid-producer. • Very important for living organisms, makes up the ¼ of atoms in living things. • The most abundant in the earth crust. It has 21% by volume abundance in the air. • Forms diatomic structure(O 2) and most active nonmetal after fluorine. • Allotropes are O 2 and O 3 (ozone). • Has a parametric property and dissolves in water. • Exist as free and combined states in nature.

Chapter 5 1. Oxygen

Chapter 5 1. Oxygen Ozone, O 3 • Colorless, light blue gas, with a pleasant odor. • Slightly soluble in water. • Exist in the upper part of the atmosphere. • It absorbs ultraviolet light which can cause cancer. It can be prepared by electrical discharge from oxygen gas. 3 O 2 ↔ 2 O 3

Chapter 5 2. Occurrence of Oxygen • Most abundant element on the earth (46% by mass). • Exist in both the free and combined state. • 21% by volume in air. • Water is the most important compound. • Present in many organic compounds. • Living organisms use for respiration. • Makes up ¼ of the atoms in living organisms.

Chapter 5 2. Occurrence of Oxygen Preparation • In laboratory, • Heating of some metal oxides. Ag 2 O(s) → 2 Ag(s) + ½ O 2(g) • Heating of peroxides. 2 H 2 O 2(l) → 2 H 2 O(l) + O 2(g) • Heating of NO 3 - and KCl. O 3 - compounds. 2 KCl. O 3(s) → 2 KCl(s) + 3 O 2(g) • Heating of Mn. O 4 - compounds. 2 KMn. O 4(s) → K 2 Mn. O 4(s) + Mn. O 2 + O 2(g) • Electrolysis of water. 2 H 2 O(l) → O 2(g) + 2 H 2(g)

Chapter 5 2. Occurrence of Oxygen Preparation • In industry, O 2 is prepared from air. There are two steps in the process; • Liquefying of air: After removing of CO 2 pressure is applied about 200 atm. After that at -200 0 C liquid air is formed. • Fractional distillation of liquid air: When we heat the liquid air, after nitrogen oxygen is evaporated at -183 0 C.

Chapter 5 2. Occurrence of Oxygen

Chapter 5 3. Chemical Properties of Oxygen • Reactions with metals, 4 Li(s) + O 2(g) → 2 Li 2 O(s) • Reactions with nonmetals, S(s) + O 2(g) → SO 2(s) • Reactions with compounds, CS 2(l) + 3 O 2(g) → CO 2(g) + 2 SO 2(s) • Combustion reactions. CH 4(g) + 2 O 2(g) → CO 2(g) + 2 H 2 O(g)

Chapter 5 4. Compounds of Oxygen Water, H 2 O • The most common and essetial oxide found in nature. • Only substance naturally present on the earth in all states. • Cover about 75% of the earth surface. Oceans help the balance of the world. • Essential for all living organisms. • Makes up 60% of trees, 70% of the body of an elephant, 95% of a tomato, 70% of the human body and 90% of our blood. • Needed to dissolve chemicals in our cells and to carry chemicals in our body.
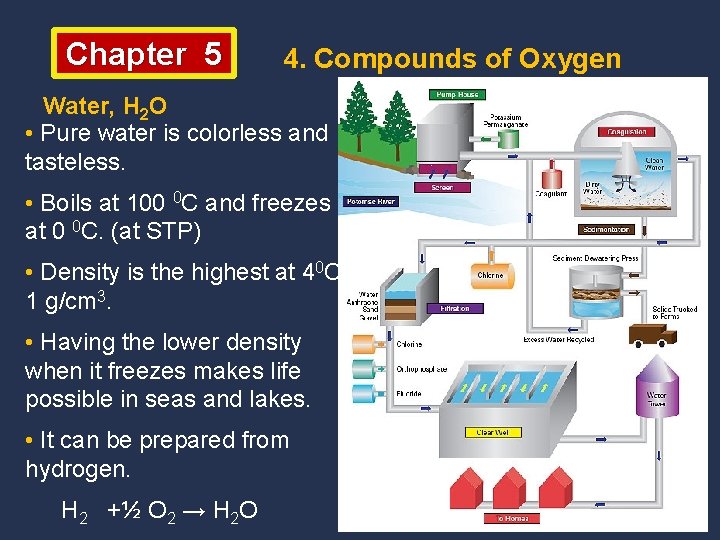
Chapter 5 4. Compounds of Oxygen Water, H 2 O • Pure water is colorless and tasteless. • Boils at 100 0 C and freezes at 0 0 C. (at STP) • Density is the highest at 40 C, 1 g/cm 3. • Having the lower density when it freezes makes life possible in seas and lakes. • It can be prepared from hydrogen. H 2 +½ O 2 → H 2 O

Chapter 5 4. Compounds of Oxygen Water, H 2 O, Uses • In industry as solvent and cooler, • In automobile radiators and nuclear power plants, • Production of steel and paper, • In the home for cooking, drinking and washing, • Water steam is used to generate electricity, • To extract minerals and to manufacture of several chemicals.

Chapter 5 5. Uses of Oxygen • In respiration tubes for divers, patients and astronauts. • Used in oxygen welding to cut very hard metals. • Rocket fuel with hydrogen. • Production of some chemicals…etc.

Chapter 5 Chapter Review Example 1 What are the compositions of gases in air? Example 2 Why oxygen is so important for living beings? Example 3 List down the physical characteristics of oxygen. Example 4 What are the allotropic forms of oxygen?

Chapter 5 Chapter Review Example 5 Match column A with column B. A B Ozone Ca. O, Na 2 O Carbon monoxide CO 2 Steam Fe 2 O 3 Rust O 3 Smoke H 2 O(g) ash CO

Chapter 5 Chapter Review Example 5 A standard 18 -L scuba tank is filled with air under a pressure of 197. 4 atm at 27 o. C. Calculate the mass of oxygen gas in the tank under these conditions. (R: 0. 082, O 2: 32)

Chapter 5 Chapter Review Example 6 What is the percent of oxygen in the rust, Fe 2 O 3? (Fe: 56, O: 16)

Chapter 5 Chapter Review Example 7 Choose the correct usage area for oxygen gas. Breathing Purifying metals Storing food Welding Cooking of cake In ambulances

Chapter 5 Example 7 ……. . Chapter Review

Chapter 5 6. Sulfur Discovered: In 1787 by Lavoisier. 1 st Ionization energy 999. 6 kj/mol Earth`s crust abundance 0. 042% Common oxidation num. -2, +4, +6 Electron configuration [Ne]3 s 23 p 4 Physical state Solid Melting point 115. 20 C Color Lemon yellow Boiling point 4450 C Density 1. 96 g/ml

Chapter 5 6. Sulfur • Sulfur has been known for its medicinal value and its germicidal effect since ancient times. • Nonmetal and poor conductor of heat and electricity. • Insoluble in water. • Exist both in free form and in compounds. • Allotropes are rhombic, monoclinic and plastic sulfur.

Chapter 5 Allotropes of Sulfur Rhombic • A crystal which is stable below 96 0 C. • Bright yellow in color and octahedral (rhombic) in shape. • Odorless and have S 8 molecules. 6. Sulfur Monoclinic • A crystal which is stable between 96 -119 0 C. • Crystals are long, thin and needle shaped. • Reverts to rhombic one at room temperature. Plastic • Obtained by heating sulfur to its boiling point. When it is poured into cold water , it will roll up into yellow ribbons called plastic sulfur.

Chapter 5 7. Occurrence of Sulfur • Frasch Process; In the process sulfur is obtained from underground about 200 m. Super heated water at about 170 0 C pumped at 10 atm. By steel pipes liquid sulfur is extracted. • In this way SO 2 is obtained firstly than S is produced. 4 Fe. S 2 + 11 O 2 → 2 H 2 S + SO 2 → 2 Fe 2 O 3 + 8 SO 2 3 S + 2 H 2 O

8. Chemical Properties of Sulfur Chapter 5 • Reactions with active metals, 2 Li + S → Li 2 S • Reactions with nonmetals, S(s) + O 2(g) → SO 2(s) • Reactions with some compounds, S + 2 H 2 SO 4(conc) → 2 H 2 O + 3 SO 2

Chapter 5 8. Chemical Properties of Sulfur

Chapter 5 8. Chemical Properties of Sulfur

Chapter 5 9. Compounds of Sulfur Oxides, SO 2 • Formed by the combustion reaction of S or Fe. S 2. • Toxic, colorless gas with a sharp, bad odor. • Plays a major role in producing acid rain. Oxides, SO 3 • Formed by the reaction of SO 2 with O 2. • Very active and volatile substance. • Also it plays a major role in producing acid rain. Carbon Disulfide, CS 2 • Flammable and colorless liquid, • Toxic and have a pleasant smell, good solvent for chemicals.

Chapter 5 9. Compounds of Sulfuric Acid, H 2 SO 4 • It is the most widely used chemical in the world, • Non volatile and hydroscopic liquid, • It is prepared by Contact Process. S + O 2 → O 2 SO 2 → H 2 SO 4 SO 3 → H 2 O H 2 S 2 O 7 → Oleum H 2 SO 4

Chapter 5 9. Compounds of Sulfuric Acid, H 2 SO 4, Uses • Charles Goodyear invented the automobile tires and took the “Legion d’honore” medal from Napoleon, but he died like a beggar.

Chapter 5 9. Compounds of Sulfur Hydrogen Sulfide, H 2 S • Highly toxic, colorless gas. • Smells like rotten egg. • A small amount of H 2 S in air causes the formation of a black sulfide layer on silver metal. • The odor of rotten eggs comes from H 2 S, formed by the bacterial decomposition of sulfur compounds.

Chapter 5 10. Uses of Sulfur and Sulfuric Acid • In the production of rubber, dyes, paper, fertilizers, medicines, accumulators, match boxes, soaps and detergents.

Chapter 5 Chapter Review Example 8 Choose the correct statements: A. Sulfur is placed in the third period and in the sixth A group. B. Sulfur atom contains 32 electrons. C. The valance shell of a sulfur atom contains 6 electrons. D. There are two energy levels in the sulfur atoms. E. Sulfur is an oxidizing agent, and accepting two electrons. F. Superior oxidation state of sulfur is +4. H. Sulfur forms acid oxides and acids. I. The oxidation states of sulfur are -2, -4, and -6.

Chapter 5 Chapter Review Example 9 Choose the correct statements: A. Sulfur possesses a high electrical conductivity. B. It melts easily. C. It forms S 8 molecule in free state. D. Plastic sulfur is formed by freezing of molten sulfur. E. It is found in nature only in pure state. F. The most abundant the natural sulfur ions are sulfate and sulfides. G. It can be dissolved in water.

Chapter 5 Chapter Review Example 10 Determine the oxidation states of sulfur in the following compounds: A. Al 2 S 3 B. Li 2 S C. H 2 S D. H 2 SO 3 E. SO 2 F. Na 2 SO 3 G. SO 3 H. K 2 SO 4

Chapter 5 Chapter Review Example 11 Determine the types of chemical bonds in the following compounds of sulfur: A. Hydrogen sulfide, B. Sulfur dioxide, C. Potassium sulfide Draw the structures of molecules.

Chapter 5 Chapter Review Example 12 Write down the equations of sulfur reacting with the following substances: A. Zinc B. Aluminum C. Sodium D. Hydrogen E. Iron F. Oxygen

Chapter 5 Chapter Review Example 13 Write down the equations of the formation of following substances: A. Hydrogen sulfide B. Potassium sulfide C. Mercury (II) sulfide D. Copper (II) sulfide E. Silver sulfide F. Sulfur dioxide G. Sulfur tetrafluoride H. Aluminum sulfide

Chapter 5 Chapter Review Example 14 Calculate the volume of sulfuric dioxide that can be obtained by burning of 320 kg of 4% impure sulfur.

Chapter 5 Chapter Review Example 15 Sulfur is obtained in big quantities from hydrogen sulfide, which is found as an impurity in natural gases, oil and etc. What volume of hydrogen sulfide is necessary to obtain 1 ton of sulfur, which contains 1% impurity?

Chapter 5 Chapter Review Example 16 Hydrogen sulfide burns in excess oxygen, so that sulfur dioxide and water are formed, whereas an insufficient of oxygen yields only sulfur and water. A. Write down the reactions and balance them. B. Calculate the volume of sulfur dioxide that can be obtained by complete burning of 1 m 3 of hydrogen sulfide having 20% impure by volume.

Chapter 5 Chapter Review Example 17 Write down the reactions for the following transformations: A. Fe. S H 2 S K 2 S Pb. S B. S Fe. S H 2 S SO 2 C.

Chapter 5 Example 18 A. H 2 SO 4 + Al B. Na 2 SO 4 + KNO 3 C. SO 3 + KOH D. SO 2 + Na. OH E. Cu. SO 4 + Ba. Cl 2 F. Cu(OH)2 + H 2 SO 4 G. SO 3 + Ba. O H. H 2 SO 4 + Fe 2 O 3 I. Na 2 SO 4 + Ba(NO 3)2 J. H 2 S + Cu K. KCl + H 2 S L. Na 2 SO 4 + Al(OH)3 M. H 2 SO 3 + Ca(OH)2 N. Na 2 CO 3 + H 2 S Chapter Review

Chapter 5 Chapter Review Example 19 Write down equations of reaction for following transformations: A. S SO 2 Na 2 SO 3 Ba. SO 3 B. S H 2 S SO 2 Ca. SO 3 SO 2 C. H 2 S S SO 2 K 2 SO 3 K 2 SO 4 D. Zn. S SO 2 SO 3 H 2 SO 4 Ba. SO 4 E. Fe. S H 2 S S SO 2 SO 3 Na 2 SO 4 Ba. SO 4

Chapter 5 Chapter Review Example 20 A 53. 2 g of sulfur trioxide was obtained from oxidation of 15. 68 L of sulfur dioxide at STP. Calculate the percent yield of the reaction.

Chapter 5 Chapter Review Example 21 Sulfur dioxide, which is obtained by burning of sulfur, is used in order to disinfect mushrooms, mold and etc. What is mass of sulfur with 5% impurity necessary to disinfect a room with a volume of 75 m 3, if the standard usage of sulfur dioxide is 1. 6 g per cubic meter volume?

Chapter 5 Chapter Review Example 22 ? 405 tons of anhydrous sulfuric acid is obtained from 320 tons of pyrite, Fe. S 2, which contains 45% sulfur. Calculate the mass of sulfuric acid yield.

End of the chapter 5
- Slides: 52