Chapter 5 Nuclear Chemistry Radiation technologists specialize in











































































- Slides: 90

Chapter 5 Nuclear Chemistry Radiation technologists specialize in the use of imaging techniques to diagnose and treat medical problems. Patients may be given radioactive tracers that emit gamma radiation that is then detected by the scanner and used to develop an image of the desired area of the body. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

5. 1 Natural Radioactivity Mass number (protons + neutrons) Atomic number (protons) Symbol of element Learning Goal Describe alpha, beta, positron, and gamma radiation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Isotopes and Radiation Elements with atomic numbers 20 and higher usually have one or more isotopes with unstable nuclei. An unstable nucleus is radioactive, emitting small particles of energy called radiation to become more stable. Radiation may take the form of • alpha (α) particles • beta (β) particles • positrons (β+) • pure energy such as gamma (γ) rays Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Radioactive Isotopes An isotope of an element that emits radiation is called a radioisotope. When radiation is emitted, a radioisotope • may undergo a change in the number of protons • may be converted to an atom of another element Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Radioactive Isotopes Atomic symbols for various isotopes are written with • the mass number in the upper left corner and the atomic number in the lower left corner of the element symbol • the mass number after the element name iodine-131 or I-131 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Stable and Radioactive Isotopes Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Types of Radiation An alpha ( ) particle has two protons and two neutrons, and it is identical to He nucleus (2+ charge is omitted). A beta ( ) particle is a high-energy electron, with a charge of − 1, formed when a neutron is changed to a proton. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Types of Radiation A positron ( +), an example of antimatter, is a positive electron produced when a proton is transformed to a neutron and positron. Gamma rays are the high-energy radiation released from a nucleus when it decays. They have no mass or charge. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Types of Radiation Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Study Check Give the name and symbol for the following types of radiation: A. alpha radiation B. radiation with a mass number of 0 and atomic number of +1 C. radiation that represents high-energy radiation released from the nucleus Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution Give the name and symbol for the following types of radiation: A. alpha radiation B. radiation with a mass number of 0 and atomic number of +1 C. radiation that represents high-energy radiation released from the nucleus Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Biological Effects of Radiation When radiation strikes molecules, it may • knock away electrons, forming unstable ions • interact with water molecules, removing electrons and producing H 2 O+, which can cause undesirable chemical reactions Cells most sensitive to radiation are those that undergo rapid cell division, such as those in bone marrow, skin, reproductive cells, and cancer cells. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Radiation Protection Radiation protection requires • paper and clothing for alpha particles • a lab coat or gloves for beta particles • a lead shield or a thick concrete wall for gamma rays • limiting the amount of time spent near a radioactive source • increasing distance from the source Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Shielding for Radiation Protection If you work in an environment where radioactive materials are present, try to keep the time you spend in a radioactive area to a minimum. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Study Check What type of radiation will be blocked by each of the following A. lead shield B. paper, clothing Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution What type of radiation will be blocked by each of the following A. lead shield B. paper, clothing gamma radiation alpha radiation Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

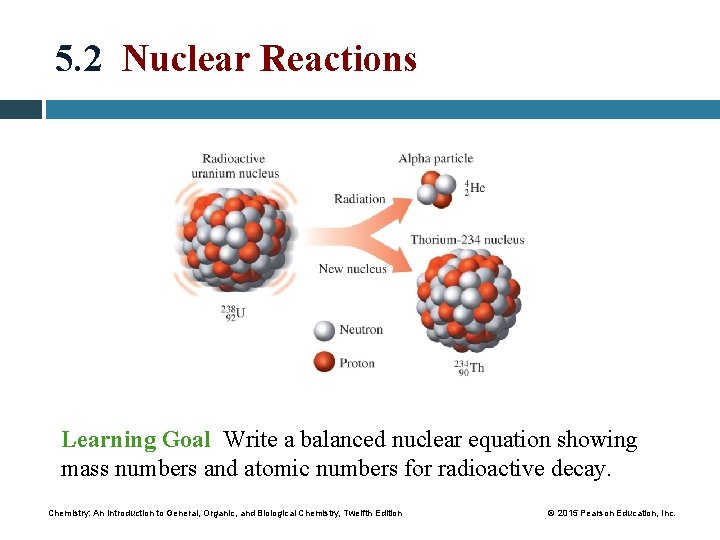
5. 2 Nuclear Reactions Learning Goal Write a balanced nuclear equation showing mass numbers and atomic numbers for radioactive decay. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Balancing Nuclear Equations In a balanced nuclear equation, particles are emitted from the nucleus, and • the mass number and atomic number may change • the sum of the mass numbers and the sum of the atomic numbers are equal for the reactants and the products Mass number sum: 251 = 251 Cf 247 Cm + 4 He 98 96 2 Atomic number sum: 98 = 98 Core Chemistry Skill Writing Nuclear Equations Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Alpha Decay When a radioactive nucleus emits an alpha particle, a new nucleus forms with a mass number that is decreased by 4 and an atomic number that is decreased by 2. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Guide to Completing Nuclear Equations Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Equation for Alpha Emission Complete the following nuclear equation for the decay of americium-241: SOLUTION: ANALYZE THE PROBLEM GIVEN Am-241, alpha decay NEED balanced nuclear equation STEP 1 Write the incomplete nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Equation for Alpha Emission Complete the following nuclear equation for the decay of americium-241: STEP 2 Determine the missing mass number. 241 =? + 4 241 − 4 =? 241 – 4 = 237 (mass number of new nucleus) STEP 3 Determine the missing atomic number. 95 =? + 2 95 – 2 = ? 95 − 2 = 93 (atomic number of new nucleus) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Equation for Alpha Emission Complete the following nuclear equation for the decay of americium-241: STEP 4 Determine the symbol of the new nucleus. The element that has atomic number 93 is neptunium, (Np). STEP 5 Complete the nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

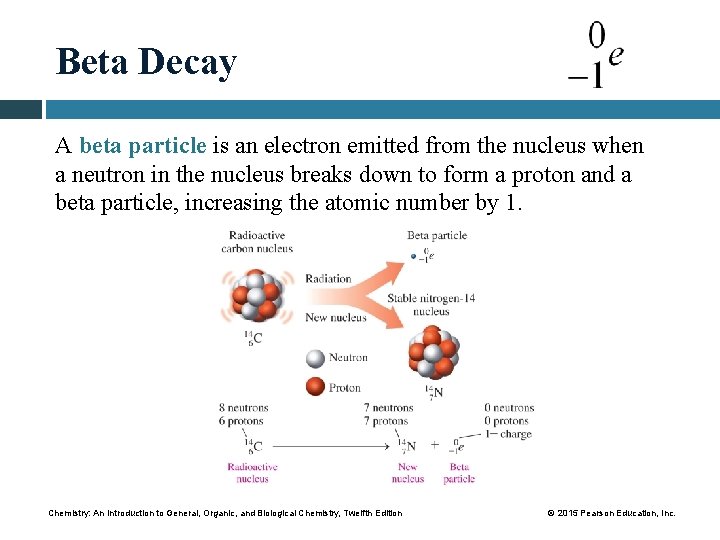
Beta Decay A beta particle is an electron emitted from the nucleus when a neutron in the nucleus breaks down to form a proton and a beta particle, increasing the atomic number by 1. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Equation for Beta Decay Write an equation for the decay of 42 K, a beta emitter. ANALYZE THE PROBLEM GIVEN K-42, beta decay NEED balanced nuclear equation STEP 1 Write the incomplete nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Equation for Beta Decay Write an equation for the decay of 42 K, a beta emitter. STEP 2 Determine the missing mass number. 42 =? +0 42 – 0 = ? 42 − 0 = 42 (mass number of new nucleus) STEP 3 Determine the missing atomic number. 19 =? − 1 19 + 1 = ? 19 + 1 = 20 (atomic number of new nucleus) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Equation for Beta Decay Write an equation for the decay of 42 K, a beta emitter. STEP 4 Determine the symbol of the new nucleus. The element that has atomic number 20 is calcium (Ca). STEP 5 Complete the nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Study Check Write the nuclear equation for the beta decay of 60 Co. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution Write the nuclear equation for the beta decay of 60 Co. ANALYZE THE PROBLEM GIVEN Co-60, beta decay NEED balanced nuclear equation STEP 1 Write the incomplete nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution Write the nuclear equation for the beta decay of 60 Co. STEP 2 Determine the missing mass number. 60 =? +0 60 – 0 = ? 60 − 0 = 60 (mass number of new nucleus) STEP 3 Determine the missing atomic number. 27 =? − 1 27 + 1 = ? 27 + 1 = 28 (atomic number of new nucleus) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution Write the nuclear equation for the beta decay of 60 Co. STEP 4 Determine the symbol of the new nucleus. The element that has atomic number 28 is nickel (Ni). STEP 5 Complete the nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Positron Emission In positron emission, • a proton is converted to a neutron and a positron • the mass number of the new nucleus is the same, but the atomic number decreases by 1 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Gamma Radiation In gamma radiation, • energy is emitted from an unstable nucleus, indicated by m following the mass number • the mass number and the atomic number of the new nucleus are the same Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Summary of Types of Radiation When the nuclei of alpha, beta, positron, and gamma emitters emit radiation, new and more stable nuclei are produced. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Producing Radioactive Isotopes Radioactive isotopes are produced • when a stable nucleus is converted to a radioactive nucleus by bombarding it with a small particle • in a process called transmutation When nonradioactive B-10 is bombarded by an alpha particle, the products are radioactive N-13 and a neutron. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Equation for Producing New Isotopes by Bombardment Write the balanced nuclear equation for the bombardment of nickel-58 by a proton, , which produces a radioactive isotope and an alpha particle. SOLUTION: ANALYZE THE PROBLEM GIVEN proton bombardment, Ni-58 NEED balanced nuclear equation STEP 1 Write the incomplete nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Equation for Producing New Isotopes by Bombardment Write the balanced nuclear equation for the bombardment of nickel-58 by a proton, , which produces a radioactive isotope and an alpha particle. STEP 2 Determine the missing mass number. 1 + 58 =? +4 59 – 4 =? 59 − 4 = 55 (mass number of new nucleus) STEP 3 Determine the missing atomic number. 1 + 28 =? +2 29 − 2 =? 29 − 2 = 27 (atomic number of new nucleus) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Equation for Producing New Isotopes by Bombardment Write the balanced nuclear equation for the bombardment of nickel-58 by a proton, , which produces a radioactive isotope and an alpha particle. STEP 4 Determine the symbol of the new nucleus. The element that has atomic number 27 is cobalt (Co). STEP 5 Complete the nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Study Check What radioactive isotope is produced when a neutron bombards 98 Tc, releasing an alpha particle? Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution What radioactive isotope is produced when a neutron bombards 98 Tc, releasing an alpha particle? ANALYZE THE PROBLEM GIVEN neutron bombardment, Tc-98 NEED balanced nuclear equation STEP 1 Write the incomplete nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution What radioactive isotope is produced when a neutron bombards 98 Tc, releasing an alpha particle? STEP 2 Determine the missing mass number. 1 + 98 = ? + 4 99 – 4 = ? 99 − 4 = 95 (mass number of new nucleus) STEP 3 Determine the missing atomic number. 43 + 0 = ? + 2 43 − 2 = ? 43 − 2 = 41 (atomic number of new nucleus) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution What radioactive isotope is produced when a neutron bombards 98 Tc, releasing an alpha particle? STEP 4 Determine the symbol of the new nucleus. The element that has atomic number 41 is niobium (Nb). STEP 5 Complete the nuclear equation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

5. 3 Radiation Measurement A radiation technician uses a Geiger counter to check radiation levels. Learning Goal Describe the detection and measurement of radiation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Geiger Counter A Geiger counter • detects beta and gamma radiation • uses ions produced by radiation to create an electrical current Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Units for Measuring Radiation Units for measuring radiation activity include the following: • curie (Ci), the number of disintegrations that occurs in 1 s for 1 g of radium, equal to 3. 7 × 1010 disintegrations/s • becquerel (Bq), the SI unit of radiation activity, which is 1 disintegration/s • rad (radiation absorbed dose), which measures the amount of radiation absorbed by a gram of material such as body tissues • gray (Gy), the SI unit for absorbed dose, defined as the joules of energy absorbed by 1 kg of body tissue Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Units for Measuring Radiation The rem (radiation equivalent in humans) measures biological effects of different types of radiation, including • alpha particles, which do not penetrate the skin; however, if they enter the body, extensive damage may occur in tissues • high-energy radiation, such as beta particles, high-energy protons, and neutrons, which travels into tissues and causes more damage than alpha particles • gamma rays, which are damaging because they travel a long way through body tissue Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Measuring Radiation Activity Often the measurement for an equivalent dose is in millirems (mrem). One rem is equal to 1000 mrem. The SI unit is the sievert (Sv). One sievert is equal to 100 rem. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

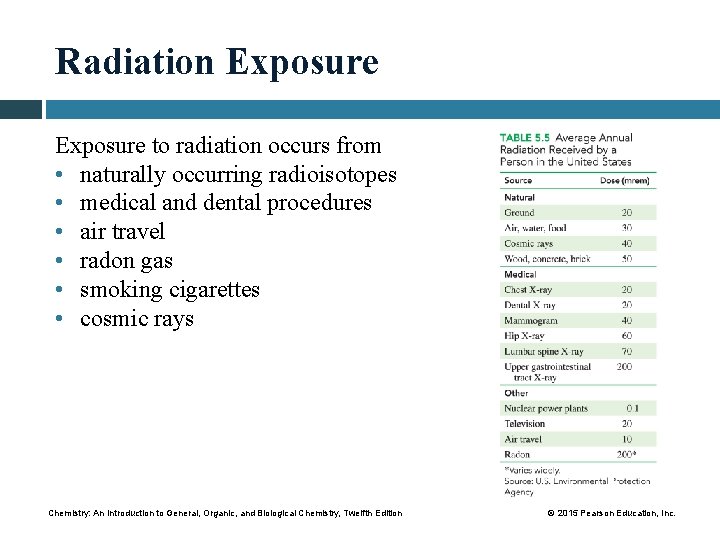
Radiation Exposure to radiation occurs from • naturally occurring radioisotopes • medical and dental procedures • air travel • radon gas • smoking cigarettes • cosmic rays Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Radiation Sickness The larger the dose of radiation received at one time, the greater the effect on the body. LD 50 • is the amount of radiation to the whole body that is the lethal dose for one-half the population • varies for different life forms, as Table 5. 6 shows Whole-body radiation of 600 rem or greater would be fatal to all humans within a few weeks. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Study Check A typical intravenous dose of I-125 for a thyroid diagnostic test is 100 Ci. What is this dosage in megabecquerels (MBq)? (3. 7 1010 Bq = 1 Ci) A. 3. 7 MBq B. 3. 7 106 MBq C. 2. 7 102 MBq Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution A typical intravenous dose of I-125 for a thyroid diagnostic test is 100 Ci. What is this dosage in megabecquerels (MBq)? (3. 7 1010 Bq = 1 Ci) 100 Ci 1 Ci 3. 7 1010 Bq 1 MBq 1 106 Ci 1 106 Bq = 3. 7 MBq The answer is A. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

5. 4 Half-Life of a Radioisotope The age of the Dead Sea Scrolls was determined using carbon-14. Learning Goal Given the half-life of a radioisotope, calculate the amount of radioisotope remaining after one or more half-lives. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

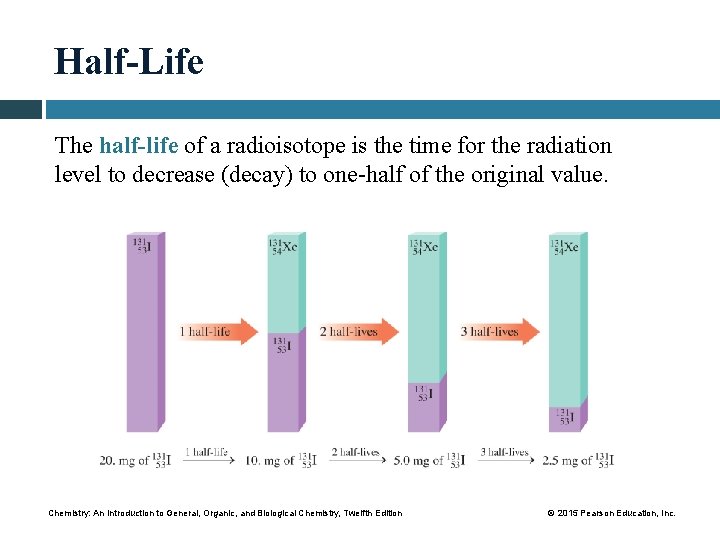
Half-Life The half-life of a radioisotope is the time for the radiation level to decrease (decay) to one-half of the original value. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

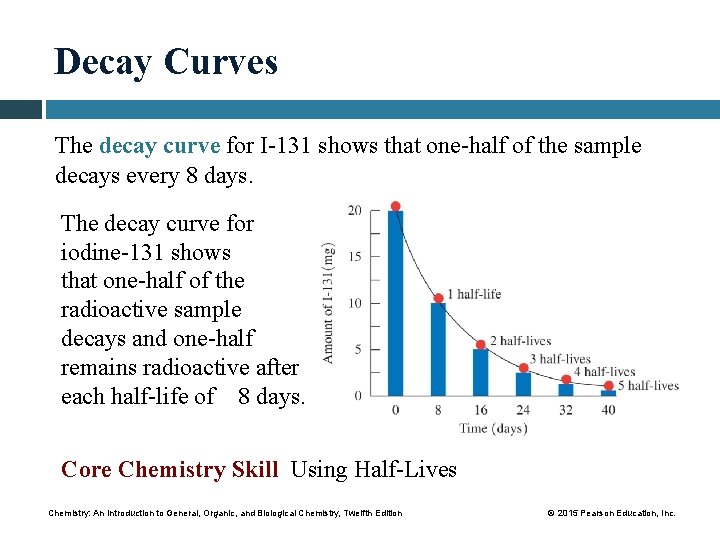
Decay Curves The decay curve for I-131 shows that one-half of the sample decays every 8 days. The decay curve for iodine-131 shows that one-half of the radioactive sample decays and one-half remains radioactive after each half-life of 8 days. Core Chemistry Skill Using Half-Lives Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

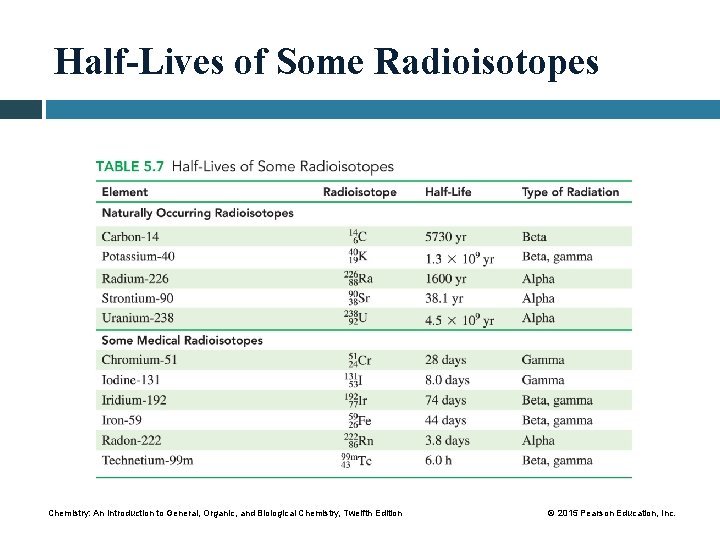
Half-Lives of Some Radioisotopes Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Guide to Using Half-Lives Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Half-Life Calculations The radioisotope strontium-90 has a half-life of 38. 1 years. If a sample contains 36 mg of Sr-90, how many milligrams will remain after 152. 4 years? SOLUTION: STEP 1 State the given and needed quantities. ANALYZE THE PROBLEM GIVEN NEED 36 mg 152. 4 yrs milligrams of Sr-90 half-life = 38. 1 yrs remaining Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Half-Life Calculations The radioisotope strontium-90 has a half-life of 38. 1 years. If a sample contains 36 mg of Sr-90, how many milligrams will remain after 152. 4 years? STEP 2 Write a plan to calculate the unknown quantity. Half-life years number of half-lives mg of Sr-90 Number of half -lives Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition mg of Sr-90 remaining © 2015 Pearson Education, Inc.

Half-Life Calculations The radioisotope strontium-90 has a half-life of 38. 1 years. If a sample contains 36 mg of Sr-90, how many milligrams will remain after 152. 4 years? STEP 3 Write the half-life equality and conversion factors. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Half-Life Calculations The radioisotope strontium-90 has a half-life of 38. 1 years. If a sample contains 36 mg of Sr-90, how many milligrams will remain after 152. 4 years? STEP 4 Set up the problem to calculate the needed quantity. 1 half-life 2 half-lives 36 mg 18 mg 9 mg Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition 4 half-lives 4. 5 mg 2. 2 mg Sr-90 © 2015 Pearson Education, Inc.

Study Check Carbon-14 was used to determine the age of the Dead Sea Scrolls. If the Dead Sea Scrolls were determined to be 2000 years old and the half-life of carbon-14 is 5730 years, what fraction of this half-life has passed? Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution Carbon-14 was used to determine the age of the Dead Sea Scrolls. If the Dead Sea Scrolls were determined to be 2000 years old and the half-life of carbon-14 is 5730 years, what fraction of this half-life has passed? STEP 1 State the given and needed quantities. ANALYZE THE PROBLEM GIVEN 2000 years old C-14 half-life = 5730 yrs Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition NEED fraction of half-life passed © 2015 Pearson Education, Inc.

Solution Carbon-14 was used to determine the age of the Dead Sea Scrolls. If the Dead Sea Scrolls were determined to be 2000 years old and the half-life of carbon-14 is 5730 years, what fraction of this half-life has passed? STEP 2 Write a plan to calculate the unknown quantity. years fraction of half-life Half-life STEP 3 Write the half-life equality and conversion factors. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution Carbon-14 was used to determine the age of the Dead Sea Scrolls. If the Dead Sea Scrolls were determined to be 2000 years old and the half-life of carbon-14 is 5730 years, what fraction of this half-life has passed? STEP 4 Set up the problem to calculate the needed quantity. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Study Check The half-life of I-123 is 13 h. How much of a 64 -mg sample of I-123 is left after 26 hours? A. 32 mg B. 16 mg C. 8 mg Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution The half-life of I-123 is 13 h. How much of a 64 -mg sample of I-123 is left after 26 hours? STEP 1 State the given and needed quantities. ANALYZE THE PROBLEM GIVEN 26 h 64 mg I-123 half-life = 13 h NEED milligrams of I-123 remaining STEP 2 Write a plan to calculate the unknown quantity. hours number of half-lives Half-life mg of I-123 Number of half-lives mg of I-123 remaining Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution The half-life of I-123 is 13 h. How much of a 64 -mg sample of I-123 is left after 26 hours? STEP 3 Write the half-life equality and conversion factors. STEP 4 Set up the problem to calculate the needed quantity. 1 half-life 2 half-lives 64 mg 32 mg 16 mg I-123 The answer is B. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

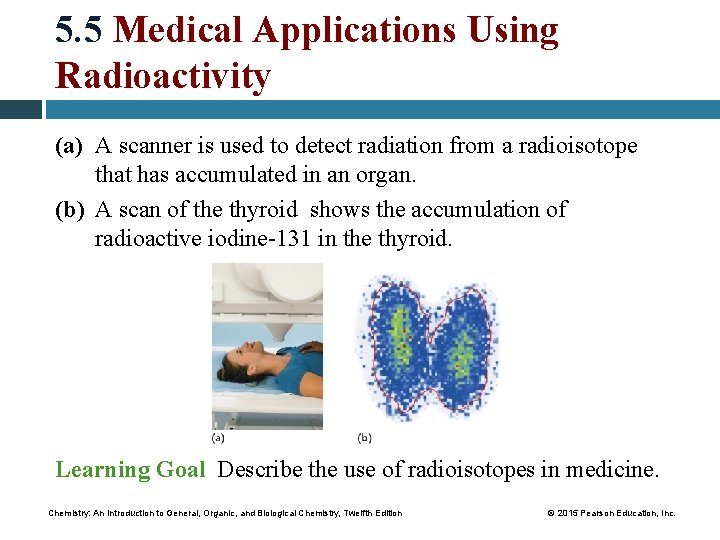
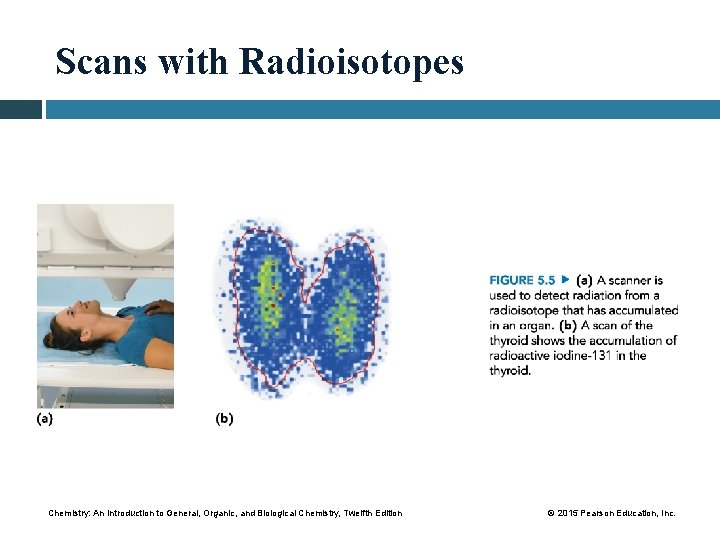
5. 5 Medical Applications Using Radioactivity (a) A scanner is used to detect radiation from a radioisotope that has accumulated in an organ. (b) A scan of the thyroid shows the accumulation of radioactive iodine-131 in the thyroid. Learning Goal Describe the use of radioisotopes in medicine. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Medical Applications of Radioisotopes with short half-lives are used in nuclear medicine because • the cells in the body do not differentiate between nonradioactive atoms and radioactive atoms • once incorporated into cells, the radioactive atoms are detected because they emit radiation giving an image of an organ Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

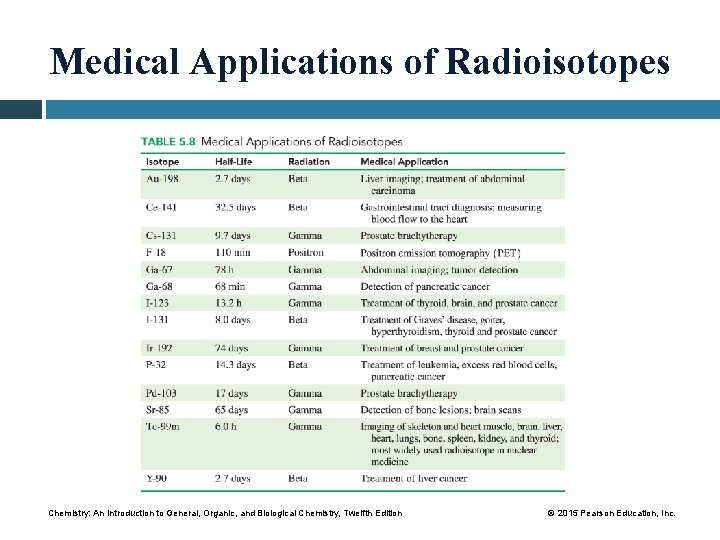
Medical Applications of Radioisotopes Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Scans with Radioisotopes After a radioisotope is ingested by the patient, • the radiologist determines the level and location of the radioactivity emitted by the radioisotope • the scanner moves slowly over the organ in which the radioisotope is absorbed • the gamma rays emitted from the radioisotope can be used to expose a photographic plate, producing a scan of the organ Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Scans with Radioisotopes Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

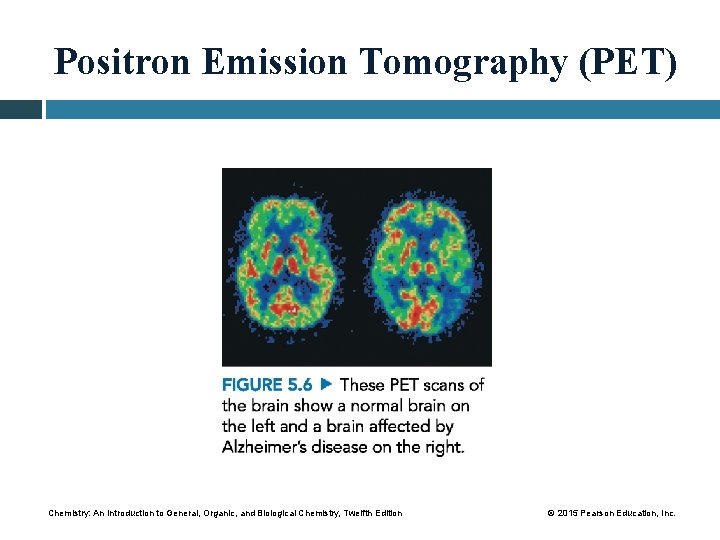
Positron Emission Tomography (PET) Positron emitters with short half-lives • can be used to study brain function, metabolism, and blood flow • include carbon-11, oxygen-15, nitrogen-13, and fluorine-18 • combine with electrons after emission to produce gamma rays, which are then detected by computers, creating a three-dimensional image of the organ Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Positron Emission Tomography (PET) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

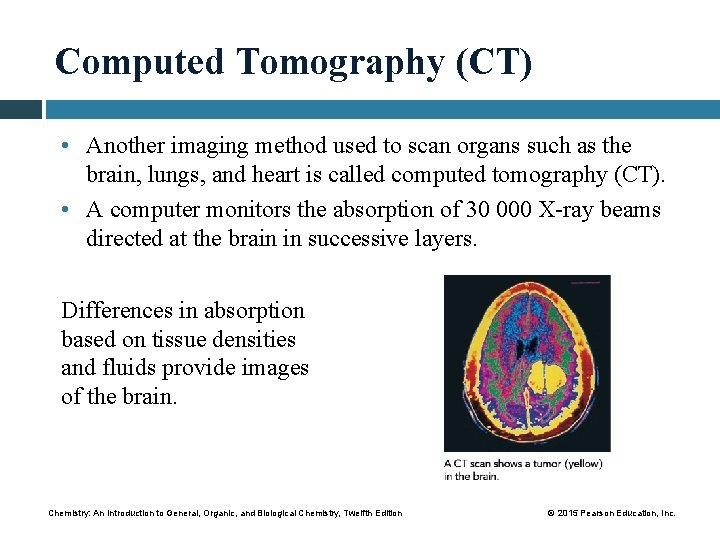
Computed Tomography (CT) • Another imaging method used to scan organs such as the brain, lungs, and heart is called computed tomography (CT). • A computer monitors the absorption of 30 000 X-ray beams directed at the brain in successive layers. Differences in absorption based on tissue densities and fluids provide images of the brain. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

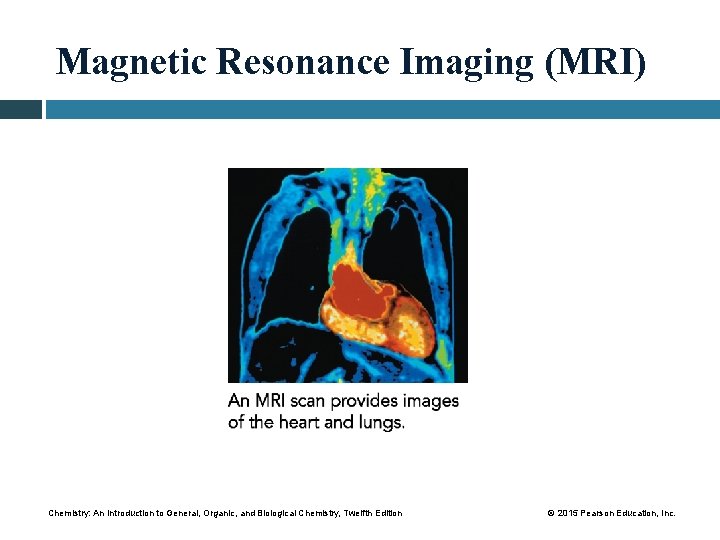
Magnetic Resonance Imaging (MRI) Magnetic resonance imaging • is an imaging technique that does not involve X-ray radiation • is the least invasive imaging method available • is based on absorption of energy when protons in hydrogen atoms are excited by a strong magnetic field • works because the energy absorbed is converted to color images of the body Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Magnetic Resonance Imaging (MRI) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Study Check Which of the following radioisotopes are most likely to be used in nuclear medicine? A. 40 K (half-life 1. 3 109 years) B. 42 K (half-life 12 hours) C. 131 I (half-life 8 days) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution Which of the following radioisotopes are most likely to be used in nuclear medicine? Radioisotopes with short half-lives are used in nuclear medicine. A. 40 K (half-life 1. 3 109 years) Not likely: half-life is too long. B. 42 K (half-life 12 hours) Short half-life, likely used. C. 131 I (half-life 8 days) Short half-life, likely used. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

5. 6 Nuclear Fission and Fusion In the 1930 s, scientists bombarding U-235 with neutrons discovered that the U-235 nucleus splits into two smaller nuclei and produces a large amount of energy. Learning Goal Describe the processes of nuclear fission and fusion. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Nuclear Fission Reaction In nuclear fission, a large nucleus • is bombarded with a small particle • splits into smaller nuclei and several neutrons, releasing large amounts of energy called atomic energy When a neutron bombards 235 U, • an unstable nucleus of 236 U undergoes fission (splits) • smaller nuclei such as Kr-91 and Ba-142 are produced • the neutrons emitted have high energy and bombard more U-235 nuclei Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Nuclear Fission Reaction A typical reaction for nuclear fission is the following: Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

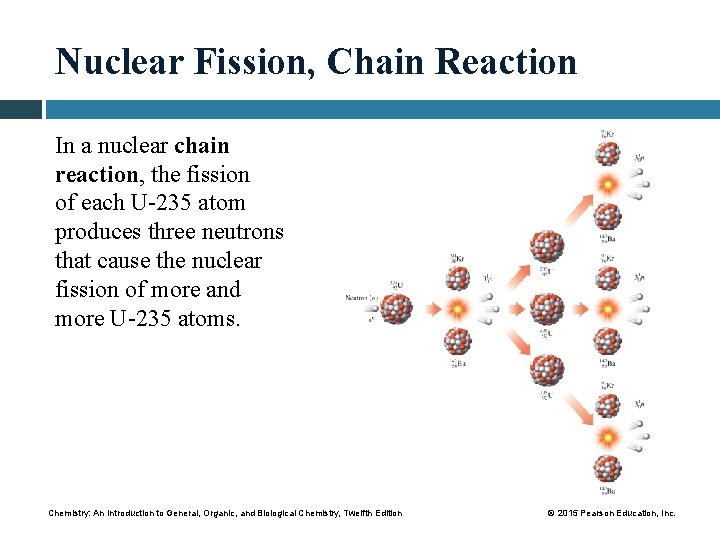
Nuclear Fission, Chain Reaction In a nuclear chain reaction, the fission of each U-235 atom produces three neutrons that cause the nuclear fission of more and more U-235 atoms. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Nuclear Fusion reactions • occur at extremely high temperatures (100 000 C) • combine small nuclei into larger nuclei • release large amounts of energy • occur continuously in the sun and stars, producing large amounts of energy Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

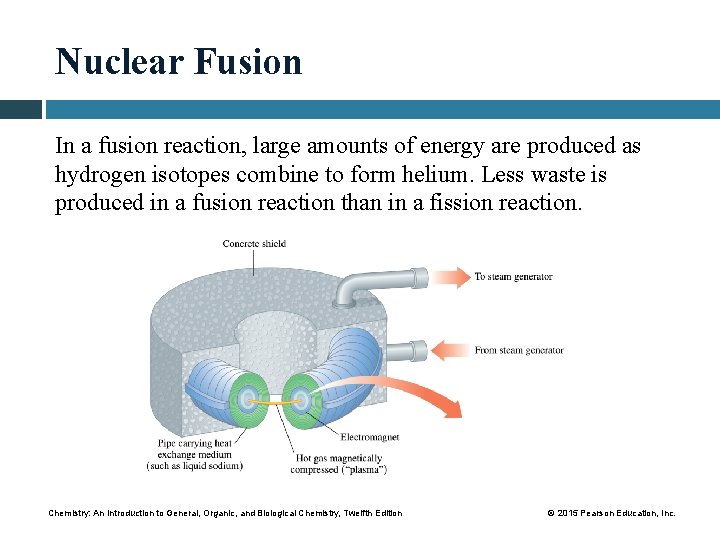
Nuclear Fusion In a fusion reaction, large amounts of energy are produced as hydrogen isotopes combine to form helium. Less waste is produced in a fusion reaction than in a fission reaction. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Study Check Indicate whether each of the following describes nuclear fission or fusion: A. B. C. D. E. A nucleus splits. Large amounts of energy are released. Small nuclei form larger nuclei. Hydrogen nuclei react. Several neutrons are released. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution Indicate whether each of the following describes nuclear fission or fusion: nuclear fission and fusion are released. nuclear fusion nuclear fission A. A nucleus splits. B. Large amounts of energy C. Small nuclei form larger nuclei D. Hydrogen nuclei react. E. Several neutrons are released. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Study Check Supply the missing atomic symbol to complete the equation for the following nuclear fission reaction: Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

Solution Supply the missing atomic symbol to complete the equation for the following nuclear fission reaction: Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.

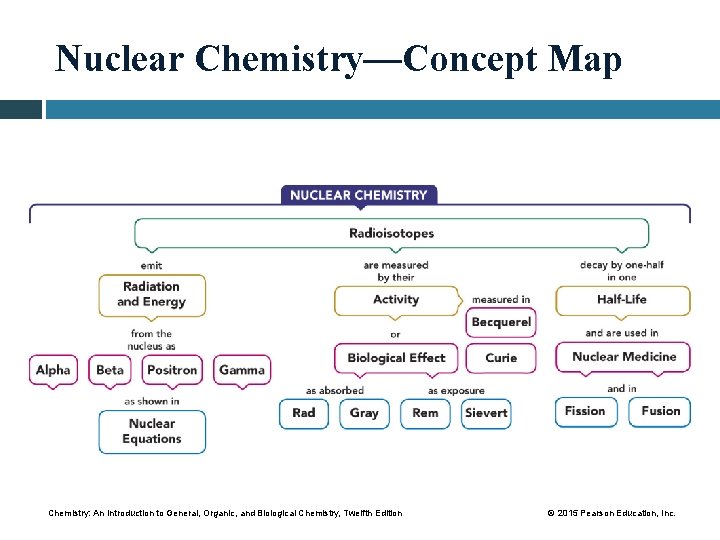
Nuclear Chemistry—Concept Map Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc.