Chapter 5 Molecules and Compounds Section 3 Naming





















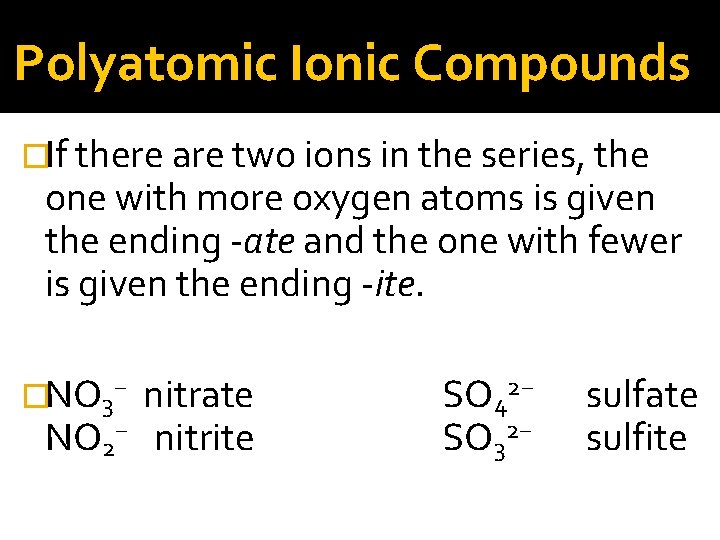



- Slides: 27

Chapter 5: Molecules and Compounds Section 3: Naming Ionic Compounds

Learning Goals �Distinguish between common and systematic names for compounds. �Name binary ionic compounds containing a metal that forms only one type of ion. �Name binary ionic compounds containing a metal that forms more than one type of ion. �Name ionic compounds containing a polyatomic ion.

Naming Compounds �Chemists have developed systematic ways to name compounds. If you learn the naming rules, you can examine a compound’s formula and determine its name, and vice versa. �Many compounds also have a common name. Na. Cl has the common name salt and the systematic name sodium chloride.

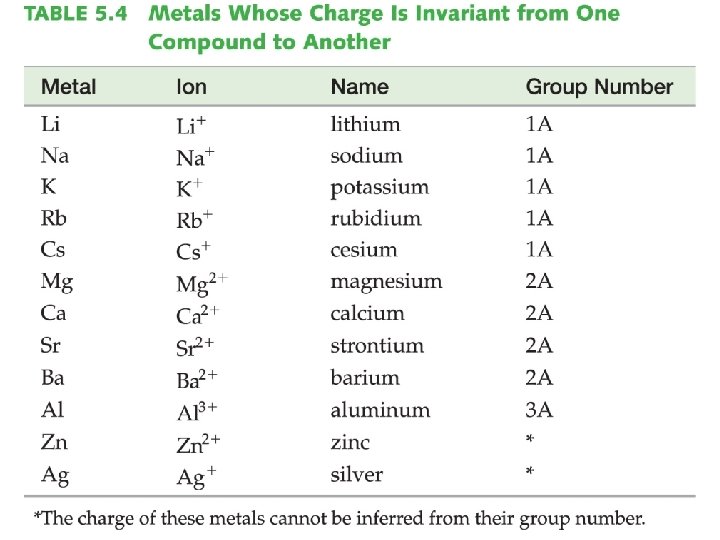
Naming Ionic Compounds �Identify if the metal cation has more than one potential charge. Such metals are usually (but not always) found in the transition metals section of the periodic table. Notable Exceptions: ▪ Always Zn 2+ and Ag+ ▪ Sn and Pb can have multiple charges



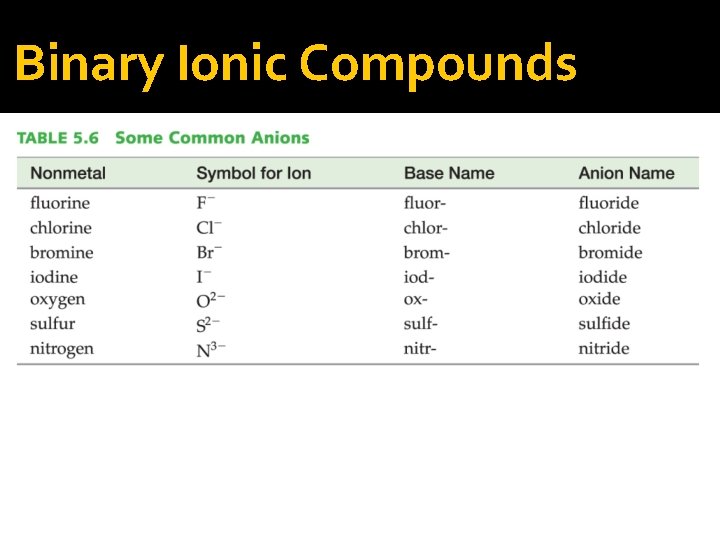
Binary Ionic Compounds �Binary compounds are those that contain only two different elements. The names for binary ionic compounds containing a metal that forms only one type of ion have the following form: Name of Cation + Base Name of Anion + ide

Binary Ionic Compounds �Since the charge of the metal is always the same for these types of compounds, it need not be specified in the compound’s name. Example: Mg. F 2

Binary Ionic Compounds

Practice �Name these compounds: Ca. O Al 2 O 3 KF Mg. Cl 2 Li 3 N

Practice �Write the formula for these ionic compounds: Potassium bromide Sodium oxide Zinc nitride

Binary Ionic Compounds �If the cation could have more than one charge, then the charge must be specified in the metal’s name. We specify the charge with a Roman numeral (in parentheses) following the name of the metal.

Binary Ionic Compounds �For example, we distinguish between Cu+ and Cu 2+ by writing a (I) to indicate the 1+ ion or a (II) to indicate the 2+ ion: Cu+ Copper(I) Cu 2+ Copper(II)

Binary Ionic Compounds �These compounds are named using the following form: Name of (charge of metal in Base Name of + + Cation roman numerals) Anion + ide

Binary Ionic Compounds �We can determine the charge of the metal from the chemical formula of the compound. The sum of all the charges must be zero.

Binary Ionic Compounds �What is the charge of iron in Fe. Cl 3? +3 �What is the name for Fe. Cl 3? Iron (III) Chloride

Practice �Name the following compounds: Pb. Cl 4 Pb. O �Explain why Ca. O is not named calcium (II) oxide.

Practice �Write the formula for the following compounds: Chromium (II) chloride Tin (IV) oxide Iron (III) oxide

Polyatomic Ionic Compounds �Ionic compounds containing polyatomic ions are named using the same procedure we apply to other ionic compounds, except that we use the name of the polyatomic ion whenever it occurs.

Polyatomic Ionic Compounds �For example, we name KNO 3 using its cation, K+, potassium, and its polyatomic anion, NO 3 -, nitrate. KNO 3 potassium nitrate

Practice �Fe(OH)2 �NH 4 NO 3

Practice �Write the formula for the following compounds: barium nitrate ammonium iodide iron (III) hydroxide

Polyatomic Ionic Compounds �Many polyatomic ions are oxyanions, anions containing oxygen. �When a series of oxyanions contain different numbers of oxygen atoms, they are named systematically according to the number of oxygen atoms in the ion.
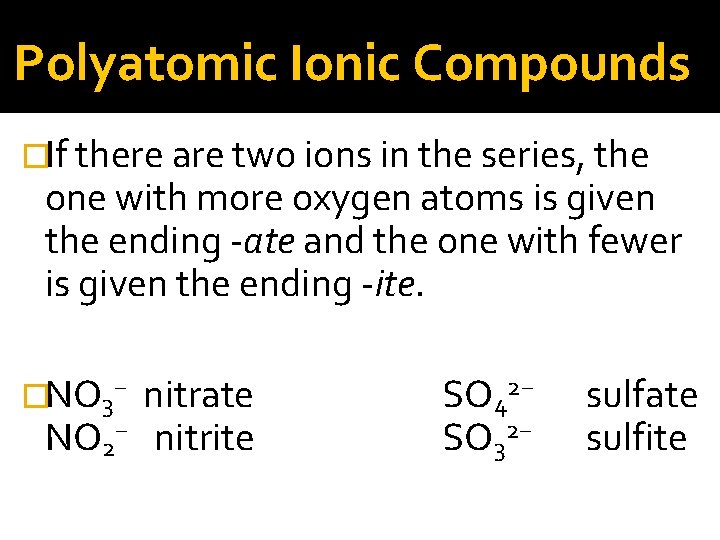
Polyatomic Ionic Compounds �If there are two ions in the series, the one with more oxygen atoms is given the ending -ate and the one with fewer is given the ending -ite. �NO 3− NO 2− nitrate nitrite SO 42− SO 32− sulfate sulfite

Polyatomic Ionic Compounds �If there are more than two ions in the series, then the prefixes hypo-, meaning “less than, ” and per-, meaning “more than, ” are used. � Cl. O − hypochlorite Br. O − hypobromite Cl. O 2− chlorite Br. O 2− bromite Cl. O 3− chlorate Br. O 3− bromate Cl. O 4− perchlorate Br. O 4−perbromate

Practice �K 2 Cr. O 4 �Mn(NO 3)2

Practice �Write the formula for the following compounds: sodium perchlorate sodium chlorite sodium nitrate sodium nitrite