Chapter 5 Molecules and Compounds Molecules and Compounds



























































- Slides: 65

Chapter 5 Molecules and Compounds

Molecules and Compounds • Salt ü Sodium – shiny, reactive, poisonous ü Chlorine – pale yellow gas, reactive, poisonous ü Sodium chloride – table salt • Sugar ü Carbon – pencil or diamonds ü Hydrogen – flammable gas ü Oxygen – a gas in air ü Combine to form white crystalline sugar 2

Law of Constant Composition • all pure substances have constant composition ü all samples of a pure substance contain the same elements in the same percentages (ratios) ü mixtures have variable composition • If we decompose water by electrolysis, we find 16. 0 grams of oxygen to every 2. 00 grams of hydrogen. Water has a constant Mass Ratio of Oxygen to Hydrogen of 8. 0. 3

Why do Compounds Show Constant Composition • smallest piece of a compound is called a molecule • every molecule of a compound has the same number and type of atoms as determined by the electronic structures of the atoms (more on that later in the year) • since all the molecules of a compound are identical… ü every sample will have the same ratio of the elements ü every sample of the compound will have the same properties 4

Example; Constant Composition EXAMPLE 5. 1 Constant Composition of Compounds Two samples of carbon dioxide, obtained from different sources, were decomposed into their constituent elements. One sample produced 4. 8 g of oxygen and 1. 8 g of carbon, and the other sample produced 17. 1 g of oxygen and 6. 4 g of carbon. Show that these results are consistent with the law of constant composition. To show this, compute the mass ratio of one element to the other by dividing the larger mass by the smaller one. For the first sample: Solution: For the second sample: Since the ratios are the same for the two samples, these results are consistent with the law of constant composition. 5

5. 3 A What is the carbon-hydrogen mass ratio for methane (CH 4)? 1. 2. 3. 4. 5. 1 B 1. 2. 3. 4. 5. Examples; Using Mass Ratios 1 0. 33 3 4 0. 25 If the mass ratio of lead(II) sulfide is 270. 0 g lead and 41. 8 g sulfur, how much lead is required to completely react with 85. 6 g of sulfur? 13. 3 185 228 312 553 6

Formulas Describe Compounds water = H 2 O two atoms of hydrogen and 1 atom of oxygen table sugar = C 12 H 22 O 11 12 atoms of C, 22 atoms of H and 11 atoms O 7

Order of Elements in a Formula • metals written first üNa. Cl • nonmetals written in order from Table 5. 1 üCO 2 üare occasional exceptions for historical or informational reasons ØH 2 O, but Na. OH Table 5. 1 Order of Listing Nonmetals in Chemical Formulas C P N H S I Br Cl O F 8

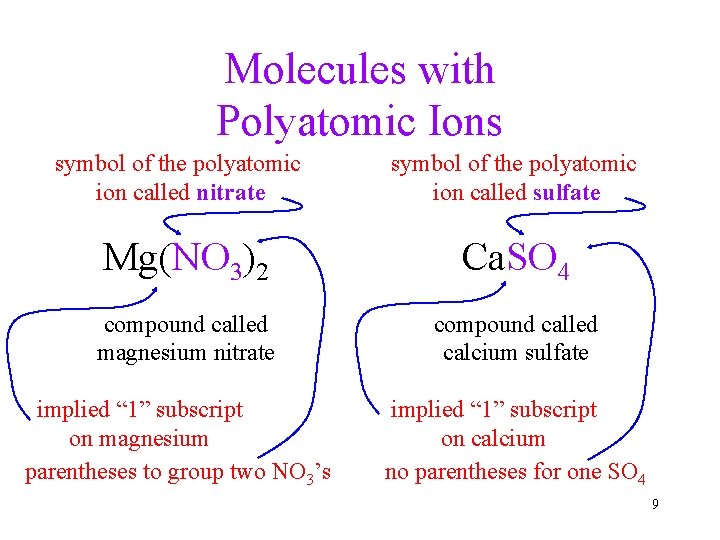
Molecules with Polyatomic Ions symbol of the polyatomic ion called nitrate symbol of the polyatomic ion called sulfate Mg(NO 3)2 Ca. SO 4 compound called magnesium nitrate compound called calcium sulfate implied “ 1” subscript on magnesium parentheses to group two NO 3’s implied “ 1” subscript on calcium no parentheses for one SO 4 9

Molecules with Polyatomic Ions subscript indicating two NO 3 groups no subscript indicating one SO 4 group Mg(NO 3)2 Ca. SO 4 compound called magnesium nitrate compound called calcium sulfate implied “ 1” subscript on nitrogen, total 2 N stated “ 3” subscript on oxygen, total 6 O implied “ 1” subscript on sulfur, total 1 S stated “ 4” subscript on oxygen, total 4 O 10

Classifying Materials • atomic elements = elements whose particles are single atoms • molecular elements = elements whose particles are multi-atom molecules • molecular compounds = compounds whose particles are molecules made of only nonmetals • ionic compounds = compounds whose particles are cations and anions 11

Molecular Elements • Certain elements occur as 2 atom molecules • Rule of 7’s ü there are 7 common diatomic elements ü find the element with atomic number 7, N ü make a figure 7 by going over to Group 7 A, then down ü don’t forget to include H 2 VIIA H 2 N 2 7 P 4 O 2 F 2 S 8 Cl 2 Br 2 I 2 12

Molecular Compounds • two or more nonmetals • smallest unit is a molecule 13

Ionic Compounds • metals + nonmetals • no individual molecule units, instead have a 3 dimensional array of cations and anions made of formula units 14

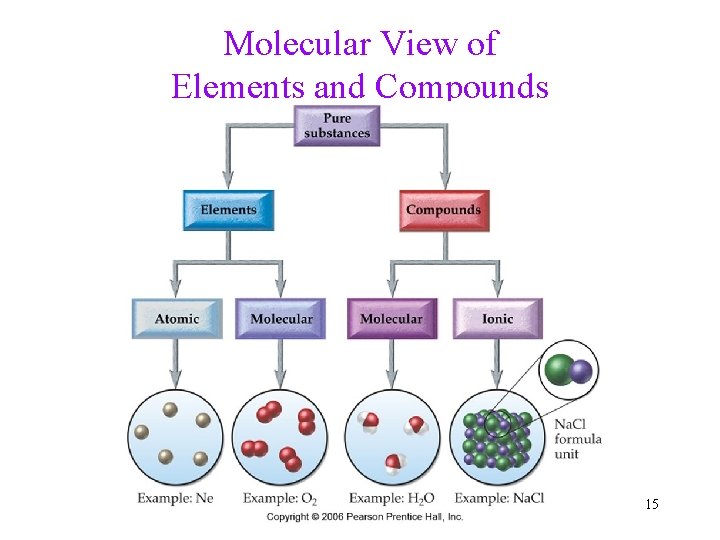
Molecular View of Elements and Compounds 15

Classify each of the following as either an atomic element, molecular compound or ionic compound • aluminum, Al • aluminum chloride, Al. Cl 3 • chlorine, Cl 2 • acetone, C 3 H 6 O • carbon monoxide, CO • cobalt, Co 16

Classify each of the following as either an atomic element, molecular compound or ionic compound • aluminum, Al = atomic element • aluminum chloride, Al. Cl 3 = ionic compound • chlorine, Cl 2 = molecular element • acetone, C 3 H 6 O = molecular compound • carbon monoxide, CO = molecular compound • cobalt, Co = atomic element 17

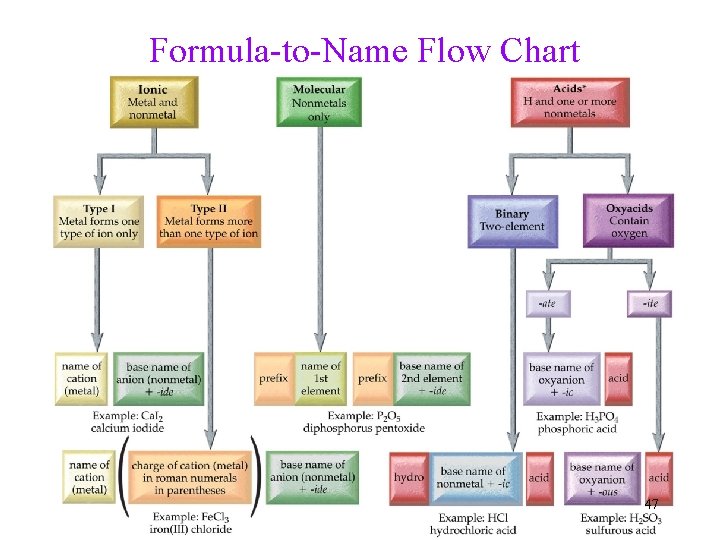
Formula-to-Name Step 1 Is the compound one of the exceptions to the rules? • H 2 O = water, steam, ice • NH 3 = ammonia

Formula-to-Name Step 2 What major class of compound is it? Ionic or Molecular

Major Classes • Ionic ü metal + nonmetal Ø metal first in formula Ø Binary Ionic ü compounds with polyatomic ions • Molecular ü 2 nonmetals Ø Binary Molecular (or Binary Covalent) ü Acids – formula starts with H Ø though acids are molecular, they behave as ionic when dissolved in water Ø may be binary or oxyacid 20

Formula-to-Name Step 3 What major subclass of compound is it? Binary Ionic, Ionic with Polyatomic Ions, Binary Molecular, Binary Acid, Oxyacid

Classifying Compounds • Compounds containing a metal and a nonmetal = binary ionic ü Type I and II • Compounds containing a polyatomic ion = ionic with polyatomic ion • Compounds containing two nonmetals = binary molecular compounds • Compounds containing H and a nonmetal = binary acids • Compounds containing H and a polyatomic ion = oxyacids 22

Formula-to-Name Step 4 Apply Rules for the Class and Subclass

Formula-to-Name Rules for Ionic • Made of cation and anion • Name by simply naming the ions üIf cation is: ØType I metal = metal name ØType II metal = metal name(charge) ØPolyatomic ion = name of polyatomic ion üIf anion is: ØNonmetal = stem of nonmetal name + ide ØPolyatomic ion = name of polyatomic ion 24

Monatomic Nonmetal Anion • determine the charge from position on the Periodic Table • to name anion, change ending on the element name to –ide 4 A = -4 5 A = -3 6 A = -2 7 A = -1 C = carbide N = nitride O = oxide F = fluoride S = sulfide Cl = chloride Si = silicide P = phosphide 25

Metal Cations • Type I ü metals whose ions can only have one possible charge Ø IA, IIA, (Al, Ga, In) ü determine charge by position on the Periodic Table Ø IA = +1, IIA = +2, (Al, Ga, In = +3) • Type II ü metals whose ions can have more than How do you know a one possible charge metal cation is Type II? ü determine charge by charge on anion its not Type I !!! 26

Determine if the following metals are Type I or Type II. If Type I, determine the charge on the cation it forms. • • • lithium, Li copper, Cu gallium, Ga tin, Sn strontium, Sr 27

Determine if the following metals are Type I or Type II. If Type I, determine the charge on the cation it forms. • • • lithium, Li copper, Cu gallium, Ga tin, Sn strontium, Sr Type II Type I +1 +3 +2 28

Type I Binary Ionic Compounds • Contain Metal Cation + Nonmetal Anion • Metal listed first in formula & name 1. name metal cation first, name nonmetal anion second 2. cation name is the metal name 3. nonmetal anion named by changing the ending on the nonmetal name to -ide 29

Type II Binary Ionic Compounds • • Contain Metal Cation + Nonmetal Anion Metal listed first in formula & name 1. name metal cation first, name nonmetal anion second 2. metal cation name is the metal name followed by a Roman Numeral in parentheses to indicate its charge ü determine charge from anion charge ü Common Type II cations in Table 5. 5 3. nonmetal anion named by changing the ending on the nonmetal name to -ide 30

Examples • • Li. Cl = lithium chloride Al. Cl 3 = aluminum chloride Pb. O = lead(II) oxide Pb. O 2 = lead(IV) oxide Mn 2 O 3 = manganese(III) oxide Zn. Cl 2 = zinc(II) chloride or zinc chloride Ag. Cl = silver(I) chloride or silver chloride 31

Compounds Containing Polyatomic Ions • Polyatomic ions are single ions that contain more than one atom • Name any ionic compound by naming cation first and then anion üNon-polyatomic cations named like Type I and II üNon-polyatomic anions named with -ide 32

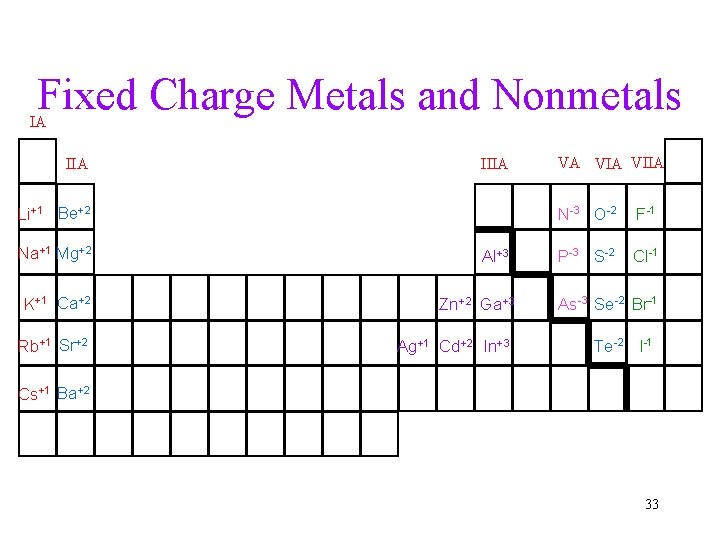
Fixed Charge Metals and Nonmetals IA IIIA Li+1 Be+2 Na+1 Mg+2 K+1 Ca+2 Rb+1 Sr+2 Al+3 Zn+2 Ga+3 Ag+1 Cd+2 In+3 VA VIIA N-3 O-2 F-1 P-3 S-2 Cl-1 As-3 Se-2 Br-1 Te-2 I-1 Cs+1 Ba+2 33

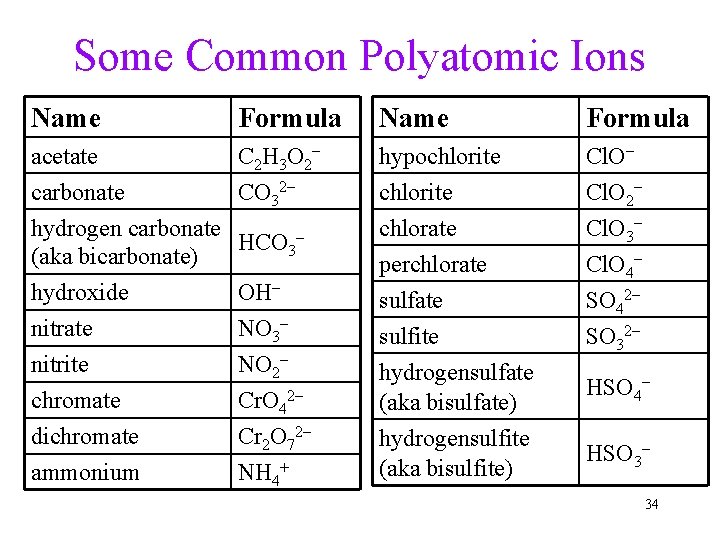
Some Common Polyatomic Ions Name Formula acetate C 2 H 3 O 2– carbonate CO 32– hydrogen carbonate HCO 3– (aka bicarbonate) hydroxide nitrate nitrite chromate dichromate ammonium OH– NO 3– NO 2– Cr. O 42– Cr 2 O 72– NH 4+ Name Formula hypochlorite chlorate Cl. O– Cl. O 2– Cl. O 3– perchlorate sulfite hydrogensulfate (aka bisulfate) hydrogensulfite (aka bisulfite) Cl. O 4– SO 42– SO 32– HSO 4– HSO 3– 34

Patterns for Polyatomic Ions 1. elements in the same column form similar polyatomic ions ü same number of O’s and same charge Cl. O 3 - = chlorate Br. O 3 - = bromate 2. if the polyatomic ion starts with H, the name adds hydrogen- prefix before name and add 1 to the charge CO 32 - = carbonate HCO 3 -1 = hydrogencarbonate 35

Periodic Pattern of Polyatomic Ions -ate groups IIIA -3 BO 3 IVA VA VIIA -2 CO 3 -1 NO 3 -2 Si. O 3 -3 PO 4 -2 SO 4 -1 Cl. O 3 -3 As. O 4 -2 Se. O 4 -1 Br. O 3 -2 Te. O 4 -1 IO 3 36

Binary Molecular Compounds of 2 Nonmetals 1. Name first element in formula first ü use the full name of the element 2. Name the second element in the formula with an -ide ü as if it were an anion, however, remember these compounds do not contain ions! 3. Use a prefix in front of each name to indicate the number of atoms a) Never use the prefix mono- on the first element 37

Subscript - Prefixes • • • 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca- not used on first nonmetal drop last “a” if name begins with vowel 38

Acids • Contain H+1 cation and anion üin aqueous solution • Binary acids have H+1 cation and nonmetal anion • Oxyacids have H+1 cation and polyatomic anion 39

Formula-to-Name Acids • acids are molecular compounds that often behave like they are made of ions • All names have acid at end • Binary Acids = hydro prefix + stem of the name of the nonmetal + ic suffix • Oxyacids ü if polyatomic ion ends in –ate = name of polyatomic ion with –ic suffix ü if polyatomic ion ends in –ite = name of polyatomic ion with –ous suffix 40

Example – Naming Binary Acids HCl 1. Is it one of the common exceptions? H 2 O, NH 3, CH 4, Na. Cl, C 12 H 22 O 11 = No! 2. Identify Major Class first element listed is H, Acid 3. Identify the Subclass 2 elements, Binary Acid 41

Sample - Naming Binary Acids – HCl 4. Identify the anion Cl = Cl-, chloride because Group 7 A 5. Name the anion with an –ic suffix Cl- = chloride chloric 6. Add a hydro- prefix to the anion name hydrochloric 7. Add the word acid to the end hydrochloric acid 42

Example – Naming Oxyacids H 2 SO 4 1. Is it one of the common exceptions? H 2 O, NH 3, CH 4, Na. Cl, C 12 H 22 O 11 = No! 2. Identify Major Class first element listed is H, Acid 3. Identify the Subclass 3 elements in the formula, Oxyacid 43

Example – Naming Oxyacids H 2 SO 4 4. Identify the anion SO 4 = SO 42 - = sulfate 5. If the anion has –ate suffix, change it to –ic. If the anion has –ite suffix, change it to -ous SO 42 - = sulfate sulfuric 6. Write the name of the anion followed by the word acid sulfuric acid (kind of an exception, to make it sound nicer!) 44

Example – Naming Oxyacids H 2 SO 3 1. Is it one of the common exceptions? H 2 O, NH 3, CH 4, Na. Cl, C 12 H 22 O 11 = No! 2. Identify Major Class first element listed is H, Acid 3. Identify the Subclass 3 elements in the formula, Oxyacid 45

Example – Naming Oxyacids H 2 SO 3 4. Identify the anion SO 3 = SO 32 - = sulfite 5. If the anion has –ate suffix, change it to –ic. If the anion has –ite suffix, change it to -ous SO 32 - = sulfite sulfurous 6. Write the name of the anion followed by the word acid sulfurous acid 46

Formula-to-Name Flow Chart 47

Name – to – Formula

Writing the Formulas from the Names • • For binary molecular compounds, use the prefixes to determine the subscripts For Type I, Type II, Ternary Compounds and Acids 1. Determine the ions present 2. Determine the charges on the cation and anion 3. Balance the charges to get the subscripts 49

Example – Binary Molecular dinitrogen pentoxide • Identify the symbols of the elements nitrogen = N oxide = oxygen = O • Write the formula using prefix number for subscript di = 2, penta = 5 N 2 O 5 50

Compounds that Contain Ions • compounds of metals with nonmetals are made of ions ümetal atoms form cations, nonmetal atoms for anions • compound must have no total charge, therefore we must balance the numbers of cations and anions in a compound to get 0 charge • if Na+ is combined with S 2 -, you will need 2 Na+ ions for every S 2 - ion to balance the charges, therefore the formula must be Na 2 S 51

Writing Formulas for Ionic Compounds 1. 2. 3. 4. 5. Write the symbol for the metal cation and its charge Write the symbol for the nonmetal anion and its charge Charge (without sign) becomes subscript for other ion Reduce subscripts to smallest whole number ratio Check that the sum of the charges of the cation cancels the sum of the anions 52

Write the formula of a compound made from aluminum ions and oxide ions 1. 2. 3. 4. 5. Write the symbol for the metal cation and its charge Write the symbol for the nonmetal anion and its charge Charge (without sign) becomes subscript for other ion Reduce subscripts to smallest whole number ratio Check that the total charge of the cations cancels the total charge of the anions Al+3 column IIIA O 2 - column VIA Al+3 O 2 Al 2 O 3 Al = (2)∙(+3) = +6 O = (3)∙(-2) = -6 53

Practice - What are the formulas for compounds made from the following ions? • potassium ion with a nitride ion • calcium ion with a bromide ion • aluminum ion with a sulfide ion 54

Practice - What are the formulas for compounds made from the following ions? • K+ with N 3 - K 3 N • Ca+2 with Br- Ca. Br 2 • Al+3 with S 2 - Al 2 S 3 55

Example – Ionic Compounds manganese(IV) sulfide 1. 2. 3. 4. 5. Write the symbol for the cation and its charge Write the symbol for the anion and its charge Charge (without sign) becomes subscript for other ion Reduce subscripts to smallest whole number ratio Check that the total charge of the cations cancels the total charge of the anions Mn+4 S 2 - Mn 2 S 4 Mn. S 2 Mn = (1)∙(+4) = +4 S = (2)∙(-2) = -4 56

Example – Ionic Compounds Iron(III) phosphate 1. 2. 3. 4. 5. Write the symbol for the cation and its charge Write the symbol for the anion and its charge Charge (without sign) becomes subscript for other ion Reduce subscripts to smallest whole number ratio Check that the total charge of the cations cancels the total charge of the anions Fe+3 PO 43 - Fe 3(PO 4)3 Fe. PO 4 Fe = (1)∙(+3) = +3 PO 4 = (1)∙(-3) = -3 57

Example – Ionic Compounds ammonium carbonate 1. 2. 3. 4. 5. Write the symbol for the cation and its charge Write the symbol for the anion and its charge Charge (without sign) becomes subscript for other ion Reduce subscripts to smallest whole number ratio Check that the total charge of the cations cancels the total charge of the anions NH 4+ CO 32 - (NH 4)2 CO 3 NH 4 = (2)∙(+1) = +2 CO 3 = (1)∙(-2) = -2 58

Practice - What are the formulas for compounds made from the following ions? • copper(II) ion with a nitride ion • iron(III) ion with a bromide ion • aluminum ion with a sulfate ion 59

Practice - What are the formulas for compounds made from the following ions? • Cu 2+ with N 3 - Cu 3 N 2 • Fe+3 with Br- Fe. Br 3 • Al+3 with SO 42 - Al 2(SO 4)3 60

Example – Binary Acids hydrosulfuric acid 1. 2. 3. 4. 5. Write the symbol for the cation and its charge Write the symbol for the anion and its charge Charge (without sign) becomes subscript for other ion Reduce subscripts to smallest whole number ratio Check that the total charge of the cations cancels the total charge of the anions H+ S 2 - in all acids the cation is H+ hydro means binary H 2 S H = (2)∙(+1) = +2 S = (1)∙(-2) = -2 61

Example – Oxyacids carbonic acid 1. 2. 3. 4. 5. Write the symbol for the cation and its charge Write the symbol for the anion and its charge Charge (without sign) becomes subscript for other ion Reduce subscripts to smallest whole number ratio Check that the total charge of the cations cancels the total charge of the anions H+ CO 32 - in all acids the cation is H+ no hydro means polyatomic ion -ic means -ate ion H+ CO 32 - H 2 CO 3 H = (2)∙(+1) = +2 CO 3 = (1)∙(-2) = -2 62

Practice - What are the formulas for the following acids? • chlorous acid • phosphoric acid • hydrobromic acid 63

Practice - What are the formulas for the following acids? • H+ with Cl. O 2– HCl. O 2 • H+ with PO 43– H 3 PO 4 • H+ with Br– HBr 64

Formula Mass • the mass of an individual molecule or formula unit • also known as molecular mass or molecular weight • sum of the masses of the atoms in a single molecule or formula unit üwhole = sum of the parts! mass of 1 molecule of H 2 O = 2(1. 01 amu H) + 16. 00 amu O = 18. 02 amu 65