CHAPTER 5 MICROBIAL METABOLISM Energy Chemical work Transport

CHAPTER 5 MICROBIAL METABOLISM

• Energy – Chemical work – Transport work – Mechanical work • Laws of thermodynamics – 1 st – 2 nd – entropy

• Two fundamental tasks required for growth and reproduction – Catabolism – Anabolism

• Metabolism – Amazing diversity but also unity • Ordered, enzyme-mediated pathways • ATP • Redox

• Catabolic reactions – hydrolytic, exergonic (-ΔG) – Keq>1, spontaneous – Cellular respiration • Provides precursor molecules and energy for anabolic reactions
• Anabolic reactions – dehydration synthesis, endergonic (+ΔG) – Keq<1, not favorable – protein synthesis • Consumes energy and precursor molecules in the biosynthesis of macromolecules
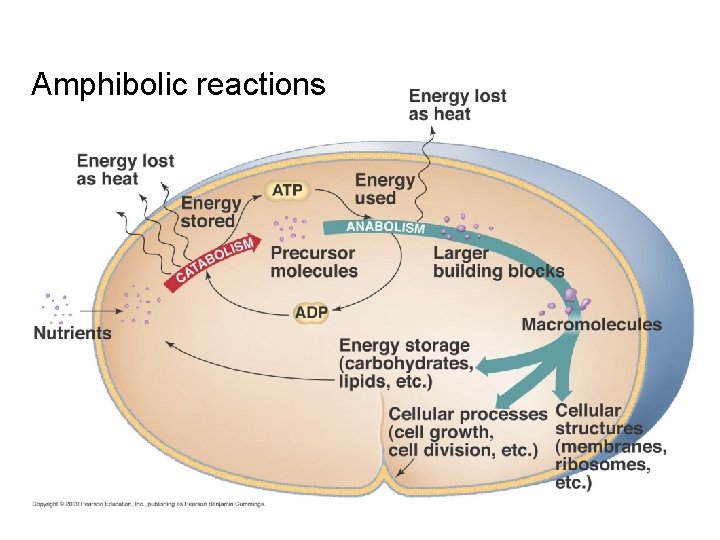
Amphibolic reactions

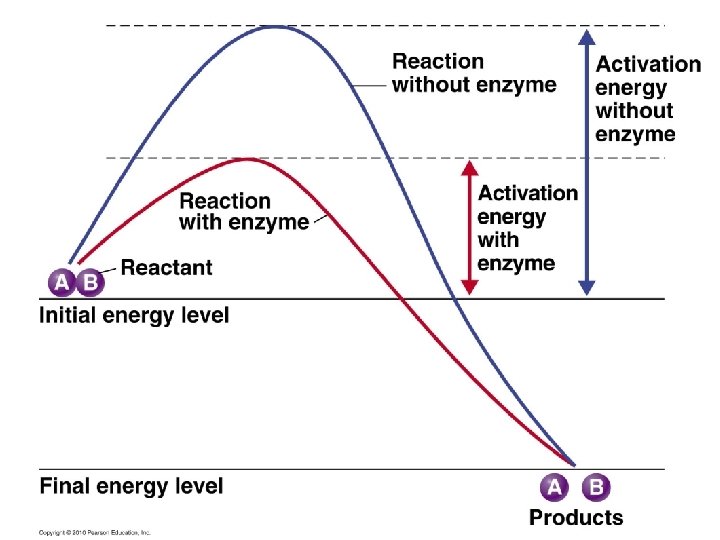
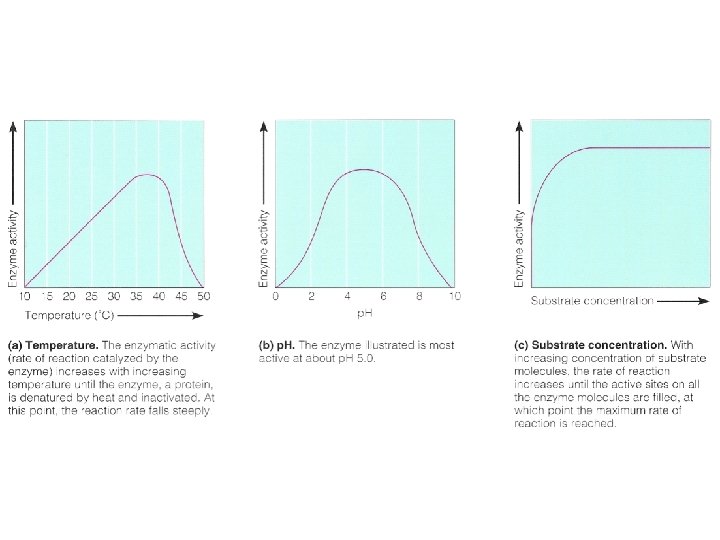
Energy of Activation (EA) • Catalysts (influence reaction rate): – Temperature – Substrate Concentration – Enzymes – biological catalysts
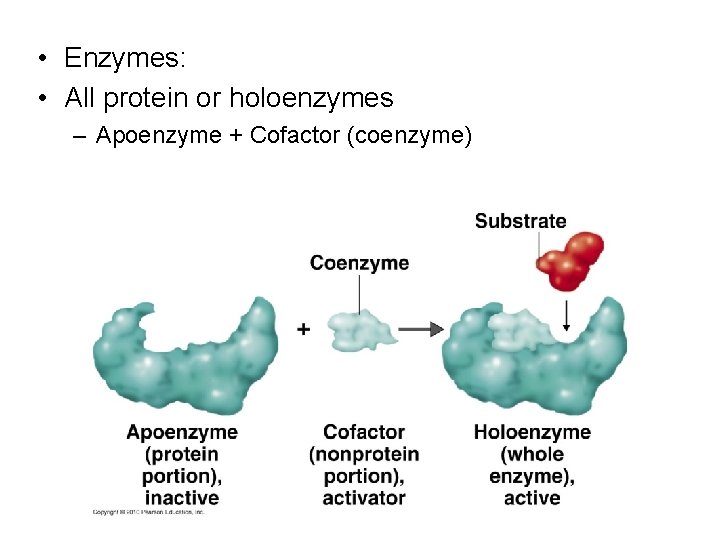
• Enzymes: • All protein or holoenzymes – Apoenzyme + Cofactor (coenzyme)

• Characteristics of enzymes – do not make reactions happen that could not happen on their own – not permanently altered or used up – substrate-specific – Function is based on structure
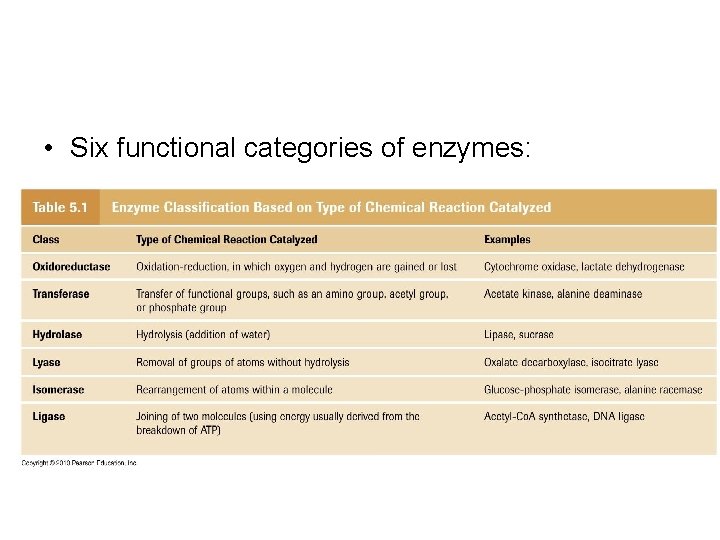
• Six functional categories of enzymes:

Unconventional Enzymes • Ribozymes – Novel type of RNA • Extremozymes – Have molecular applications
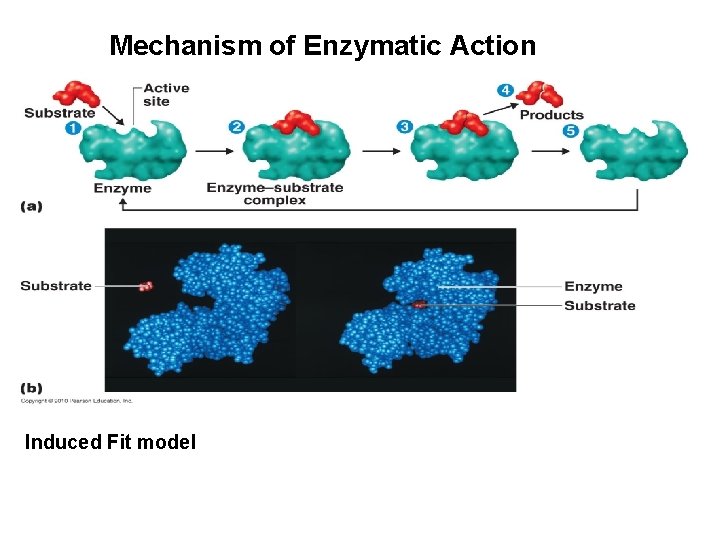
Mechanism of Enzymatic Action Induced Fit model
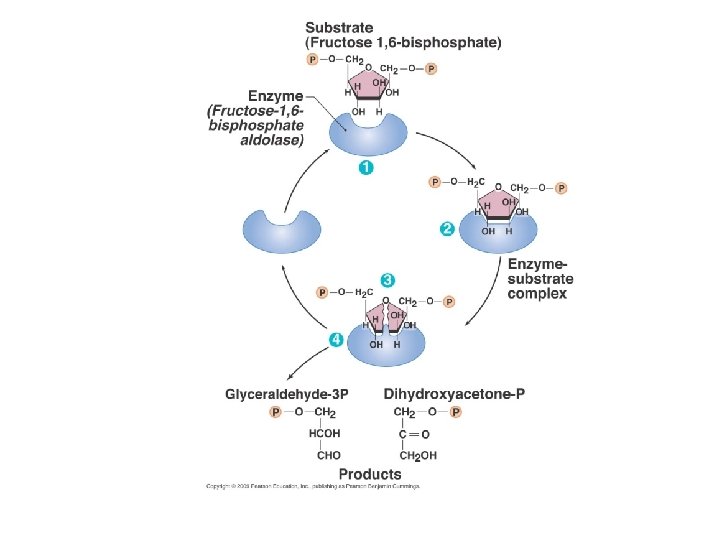
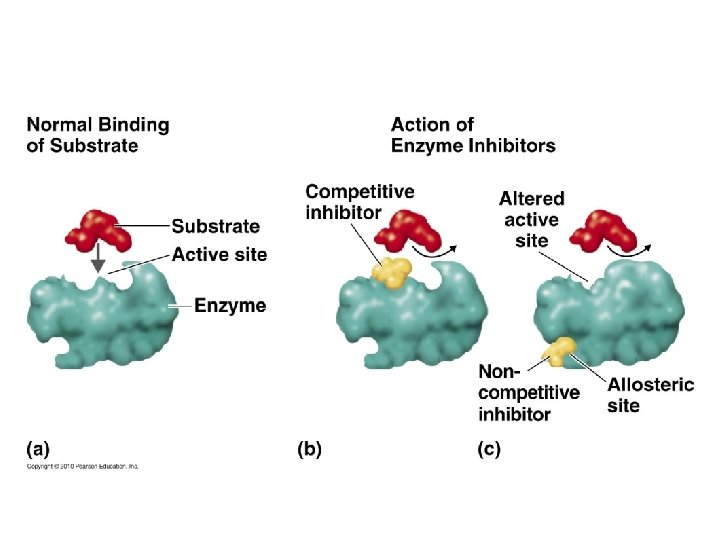
Factors influencing enzyme activity • Denaturing stresses – Heat, p. H, UV radiation, chemicals • Substrate concentration • Competitive inhibition • Non-competitive (allosteric) inhibition
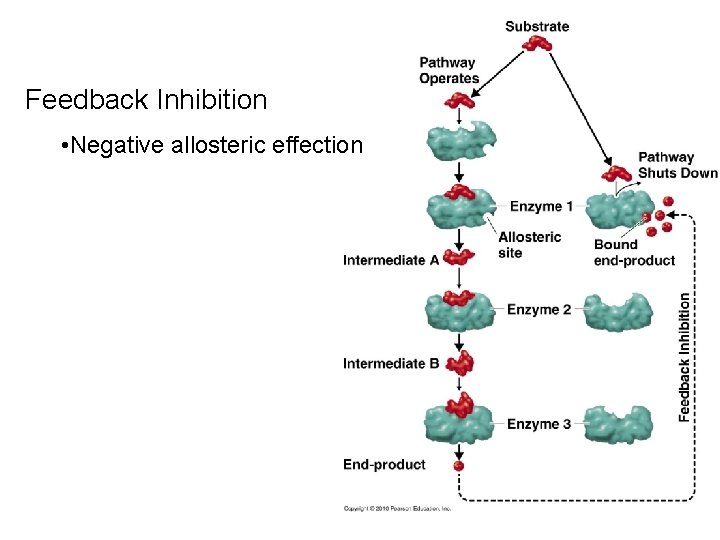
Feedback Inhibition • Negative allosteric effection

Reduction-Oxidation Reactions • Redox reactions liberate energy – always coupled – oxidation (electron donor) – reduction (electron acceptor) • Standard reduction potential (E`O) • Reducing power (potential energy)
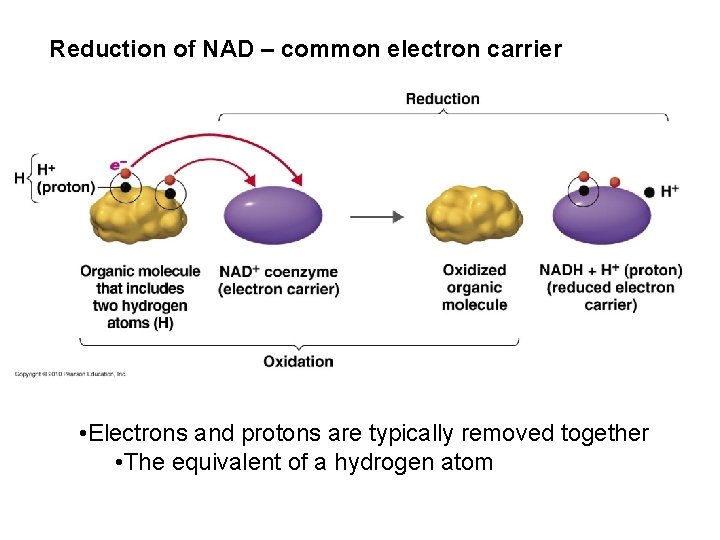
Reduction of NAD – common electron carrier • Electrons and protons are typically removed together • The equivalent of a hydrogen atom
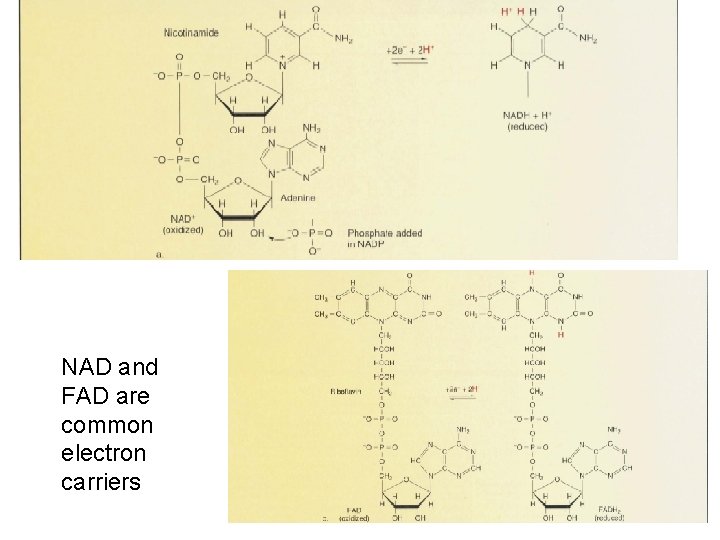
NAD and FAD are common electron carriers
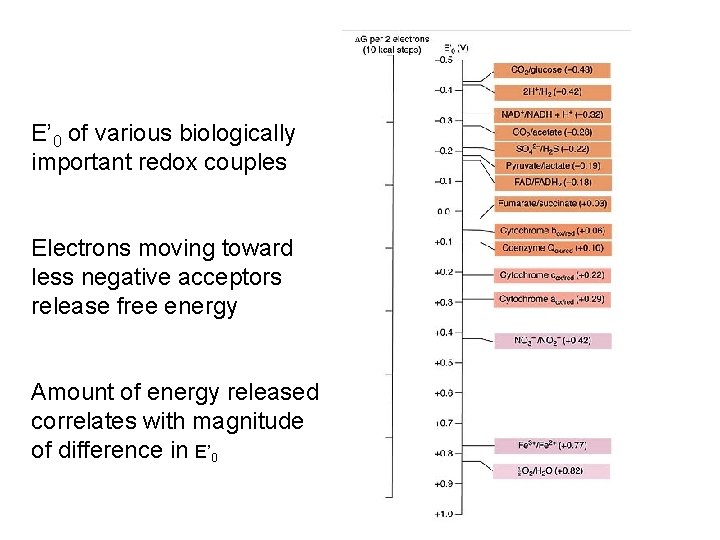
E’ 0 of various biologically important redox couples Electrons moving toward less negative acceptors release free energy Amount of energy released correlates with magnitude of difference in E’ 0
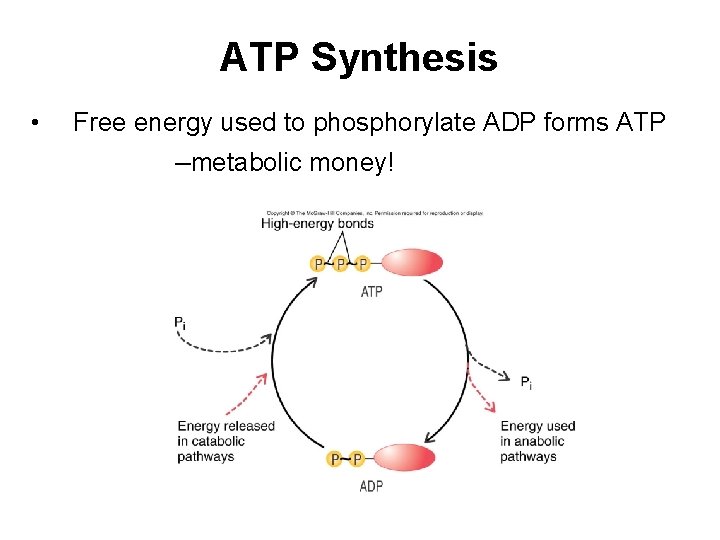
ATP Synthesis • Free energy used to phosphorylate ADP forms ATP –metabolic money!
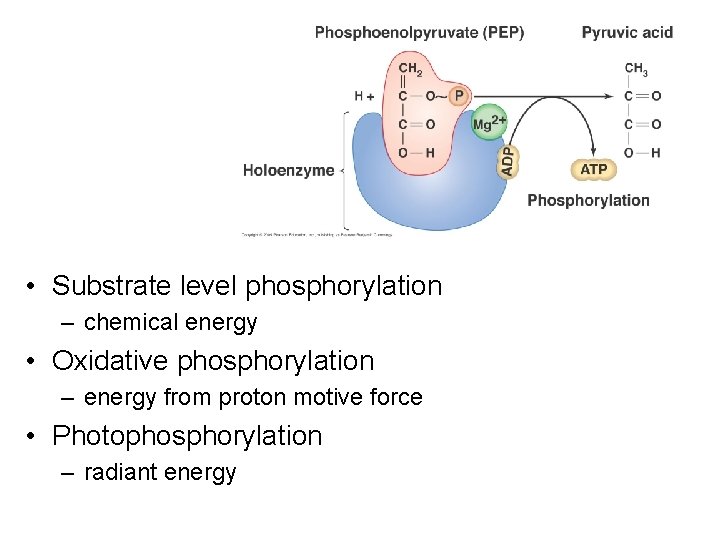
• Substrate level phosphorylation – chemical energy • Oxidative phosphorylation – energy from proton motive force • Photophosphorylation – radiant energy

Heterotrophic Metabolism • Oxidize energy (electron) rich organic molecules • Typically utilize carbohydrates – Glucose (C 6 H 12 O 6) is #1 source

• Three possible pathways based on final electron receptor – Aerobic respiration – exogenous (oxygen) – Anaerobic respiration – exogenous – Fermentation – endogenous organic
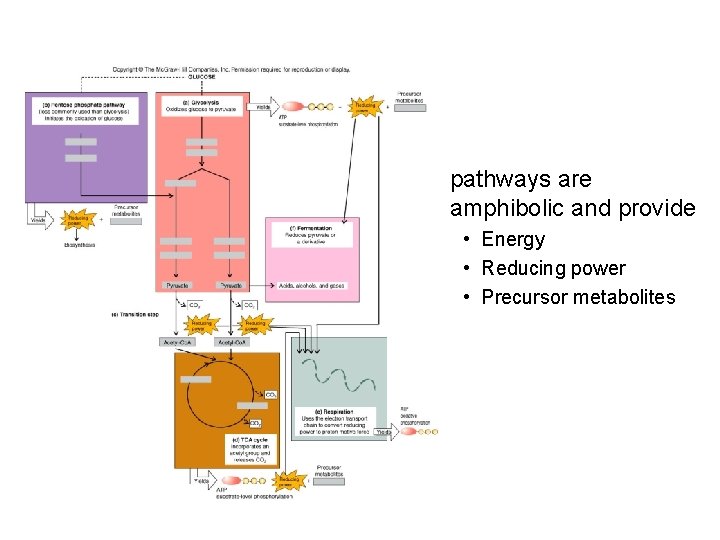
pathways are amphibolic and provide • Energy • Reducing power • Precursor metabolites

– Respiration uses reducing power to generate ATP • NADH and FADH 2 provide electrons to power proton motive force • Terminal electron acceptor varies – Oxygen in aerobic respiration – Anaerobic respiration uses alternate inorganic molecule
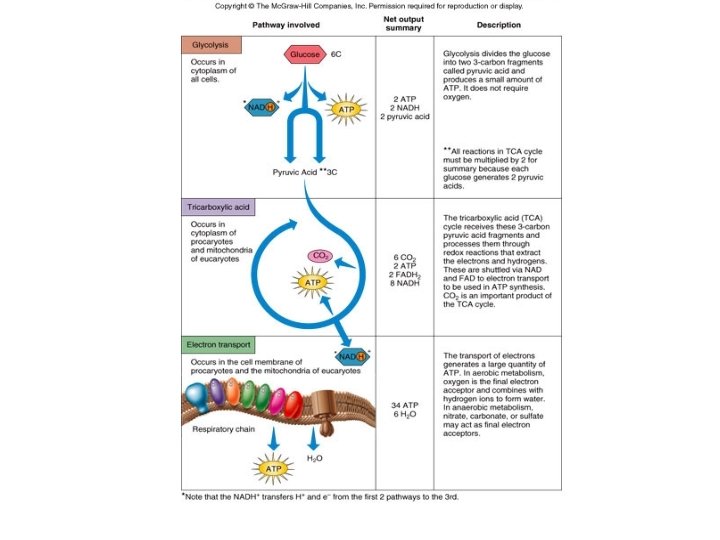

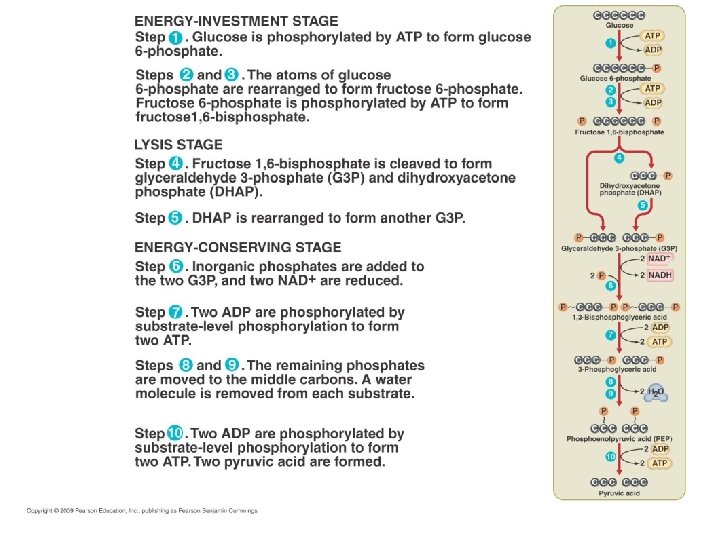
GLYCOLYSIS • Embden-Meyerhof Pathway • Common pathway • Glucose (6 C) partially broken down into 2 molecules of pyruvate (3 C) – Anerobic; cytoplasm • 2 NADH; 4 ATP • 2 ATP consumed; so net gain of 2 ATP – Substrate level phosphorylation


• Pentose phosphate pathway – Produces many intermediate materials for other pathways • glyceraldehyde 3 -phosphate, fructose 6 phosphate, ribulose 5 -phosphate • If only 5 carbon sugars are available it can biosynthesize 6 carbon sugars – Major contributor to biosynthesis • reducing power in NADPH • vital precursor metabolites for anabolic pathways • intermediates may be used to generate ATP
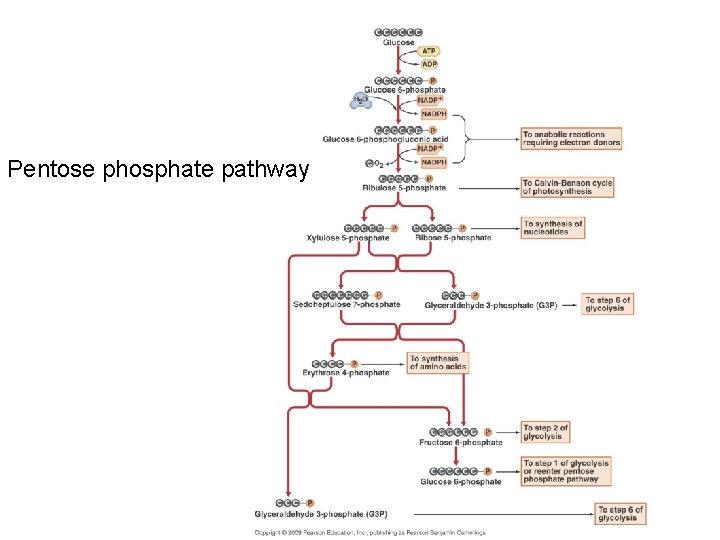
Pentose phosphate pathway

• Entner-Doudoroff pathway – Alternate pathway to glycolysis – typically not seen in G+ bacteria – major contributor to biosynthesis • reducing power as NADH and NADPH • vital precursor metabolites for anabolic pathways
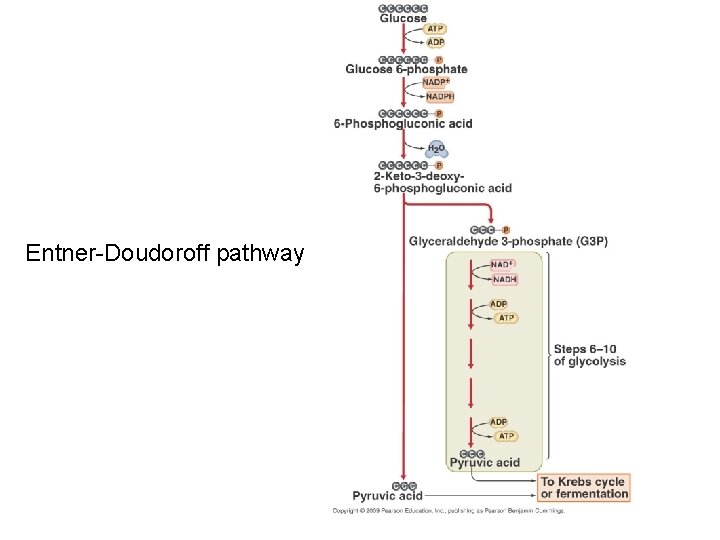
Entner-Doudoroff pathway
- Slides: 37