Chapter 5 Matter Properties and Changes Introductory Lab

Chapter 5 Matter– Properties and Changes

Introductory Lab Your science team will be given a pingpong ball and a golf ball You will write down their similarities and differences Question: Since both balls are made of plastic, will both balls float in water? Hypothesis: If…then…because

Key Vocabulary Physical property Density State of matter Size dependent property Size independent property Chemical property salts Physical Change Chemical Change Reactivity

Physical Properties A characteristic of matter than can be observed without changing the identity of the matter Common Examples: – – – – – Mass Volume Density Melting point Boiling point Freezing point Color Shape Smell Taste

Density The amount of mass in a given volume A golf ball is more dense than a ping-pong ball so the golf ball sinks in water and the ping-pong ball floats Density = mass/volume

State of Matter Whether matter is in solid, liquid, gas, or plasma form

Properties related to size Size-dependent properties- change with the size of a substance – Volume and mass Size-Independent properties- do not change with the change in size of a substance – density

p. H Scale p. H is the measure of Hydronium Ions present in a substance…sounds more complicated than it is… p. H is measured on a scale of 1 -14 – 1 is extremely acidic and 14 is extremely basic – 7 is neutral Each integer is an increase of 10 X

Properties of Acids What do you think of when you hear the word ACID? Acids are in some foods we eat and others can severely harm you Every time you eat citrus you eat acid (ascorbic acid) What taste does a lemon have?

Properties of Bases Have you ever accidentally eaten soap? – What did it taste like? Bases have a bitter taste and are slippery

Chemical Properties Characteristic of matter that allows it to change to a different type of matter What happens when you strike a match? What happens when you leave an apple on the counter?

Reactivity How easily a chemical reacts with another chemical Iron rusts easily due to its high reactivity with oxygen Gold and silver have low reactivity and therefore are ideal for jewelry

Chemical Properties of Acids React with/corrode metals Can harm body tissue
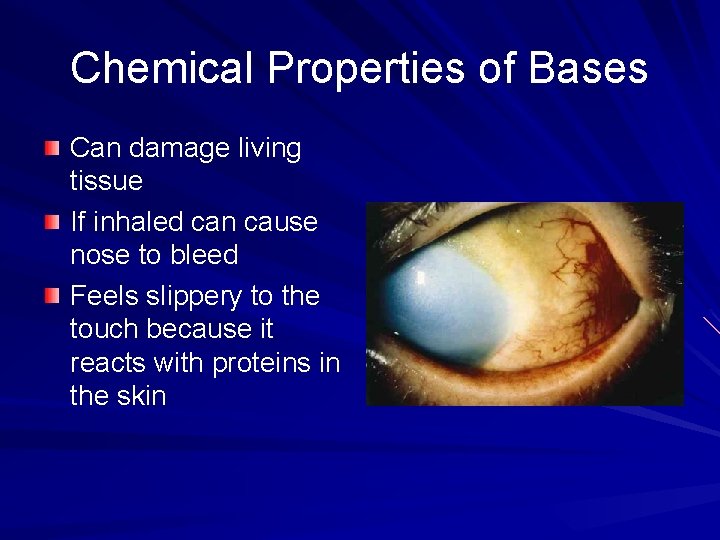
Chemical Properties of Bases Can damage living tissue If inhaled can cause nose to bleed Feels slippery to the touch because it reacts with proteins in the skin

Salts Compounds made of a metal and nonmetal that is a result of an acid-base reaction Table Salt (sodium chloride) is made from a reaction of Hydrochloric acid and Sodium Hydroxide

Foldable Make a Shutter-Fold Cut 6 shutters on each side High-five the first shutter closed Label that shutter “ACIDS” on the left and “BASES” on the right Copy down Table 2 on page 141 in your foldable

Physical Change Any change in shape, size, form, or state where the identity of matter remains the same

Chemical Change Occurs when one type of matter changes into another kind of matter with new properties Examples: Photosynthesis, rust, silver tarnishing,

Signs of a chemical change Light Heat Sound Gas bubbles Visible gas Precipitate- a solid that forms as a result of a chemical reaction

Changes in Nature Physical Changes – Physical weathering Responsible for most of the shape of Earth’s surface Gravity, plants, animals, and earthquakes all contribute to physical weathering Chemical Changes – Chemical weathering Formation of stalactites Acid rain

p. H Scale Diagram Acids have a p. H ranging from 0 -7 7 is neutral Bases have a p. H ranging from 7 -14 Now, make a poster using what you know about the p. H scale. Be sure to label acids, bases, and neutral. Give examples of several known p. H readings.
- Slides: 21