Chapter 5 LIQUIDS AND SOLIDS INTERMOLECULAR FORCES 5









































- Slides: 78

Chapter 5. LIQUIDS AND SOLIDS INTERMOLECULAR FORCES 5. 1 The Origin of Intermolecular Forces 5. 2 Ion-Dipole Forces 5. 3 Dipole-Dipole Forces 5. 4 London Forces 5. 5 Hydrogen Bonding 5. 6 Repulsions LIQUID STRUCTURE 5. 7 Order in Liquids 5. 8 Viscosity and Surface Tension 2012 General Chemistry I 1

INTERMOLECULAR FORCES (Sections 5. 1 -5. 6) 5. 1 The Origin of Intermolecular Forces Ø Phase: uniform in both chemical composition and physical state - Condensed phase: simply a solid or liquid phase - Condensed phases form when attractive intermolecular forces between molecules pull them together; repulsions dominate at even shorter separations. 2

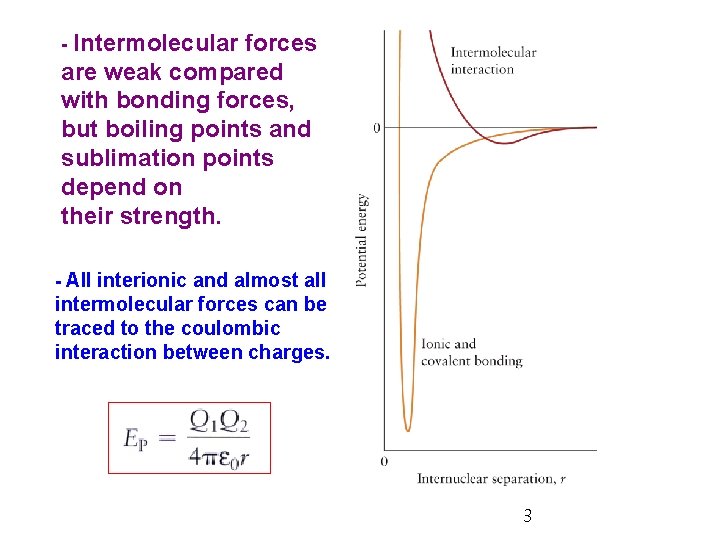
- Intermolecular forces are weak compared with bonding forces, but boiling points and sublimation points depend on their strength. - All interionic and almost all intermolecular forces can be traced to the coulombic interaction between charges. 3

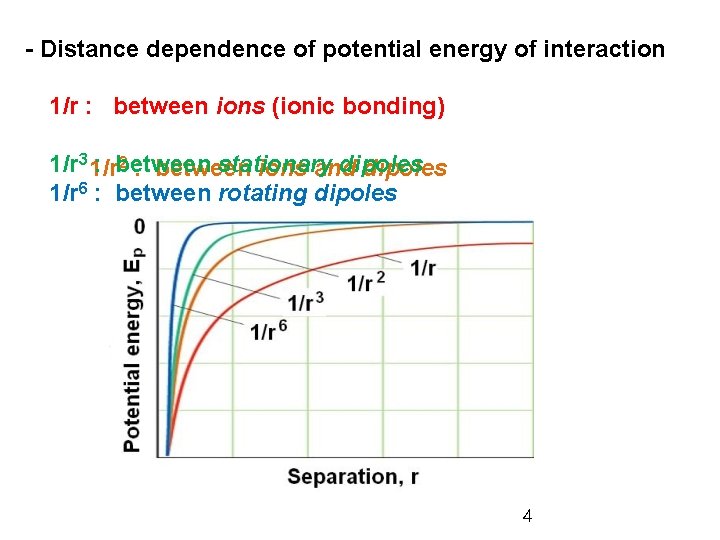
- Distance dependence of potential energy of interaction 1/r : between ions (ionic bonding) 2 : between 1/r 3 1/r : between stationary dipoles ions and dipoles 1/r 6 : between rotating dipoles 4

TABLE 5. 1 Interionic and Intermolecular Interactions 5

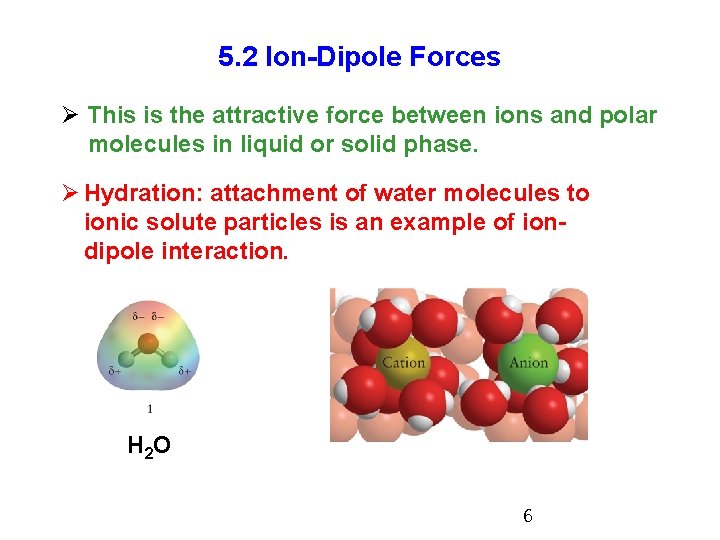
5. 2 Ion-Dipole Forces Ø This is the attractive force between ions and polar molecules in liquid or solid phase. Ø Hydration: attachment of water molecules to ionic solute particles is an example of iondipole interaction. H 2 O 6

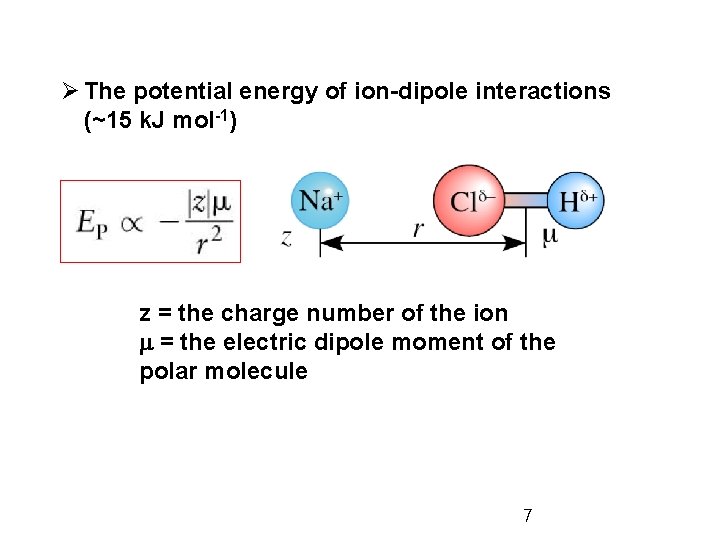
Ø The potential energy of ion-dipole interactions (~15 k. J mol-1) z = the charge number of the ion m = the electric dipole moment of the polar molecule 7

Ø Water of crystallization: smaller and highly charged cations strongly attract polar water molecules in the solid phase. - Note hydrated salts of Li and Na vs. anhydrous salts of K, Rb, Cs, and NH 4+ E. g. Na 2 CO 3. 10 H 2 O compared with K 2 CO 3 (Na+; 102 pm, K+; 138 pm) - Note Ba. Cl 2 · 2 H 2 O vs. anhydrous KCl (Ba 2+; 136 pm, K+; 138 pm) 8

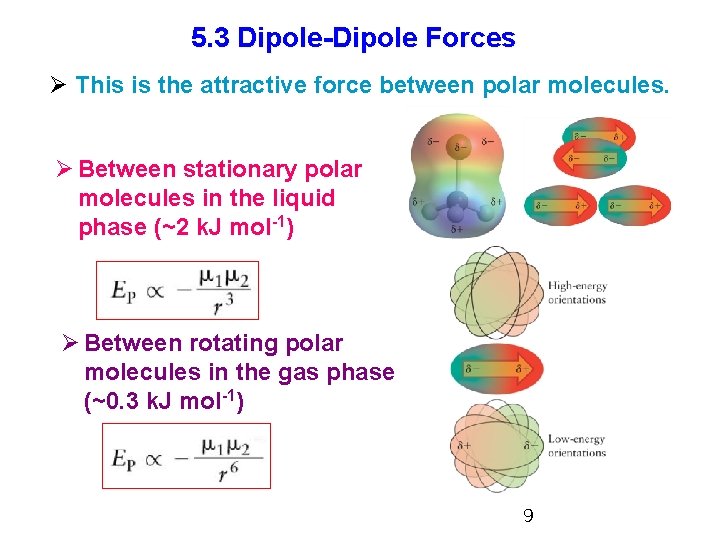
5. 3 Dipole-Dipole Forces Ø This is the attractive force between polar molecules. Ø Between stationary polar molecules in the liquid phase (~2 k. J mol-1) Ø Between rotating polar molecules in the gas phase (~0. 3 k. J mol-1) 9

10

5. 4 London Forces Ø London force (induce dipole-induced dipole force, dispersion force) ~2 k. Jmol-1: it exists between all molecules but is the only interaction between nonpolar molecules. - Attractive interactions due to instantaneous fleeting dipole moments - Fluctuation of the electron distribution in one molecule→ temporary dipole → second temporary dipole in the other molecule → ··· Time 11

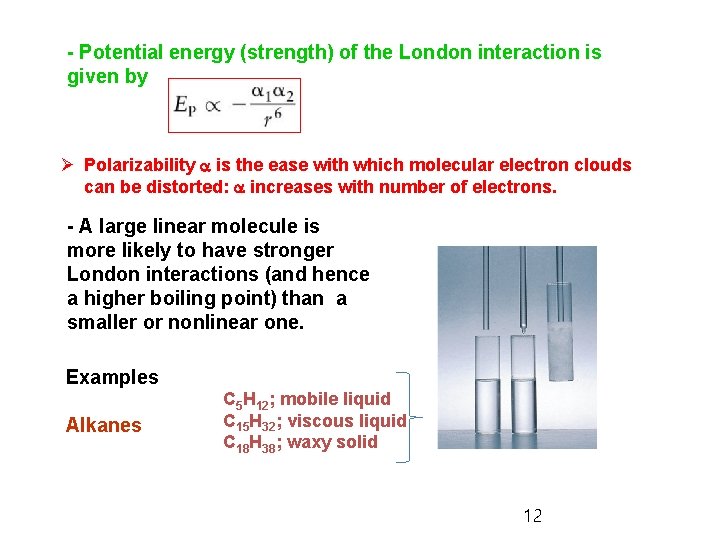
- Potential energy (strength) of the London interaction is given by Ø Polarizability a is the ease with which molecular electron clouds can be distorted: a increases with number of electrons. - A large linear molecule is more likely to have stronger London interactions (and hence a higher boiling point) than a smaller or nonlinear one. Examples Alkanes C 5 H 12; mobile liquid C 15 H 32; viscous liquid C 18 H 38; waxy solid 12

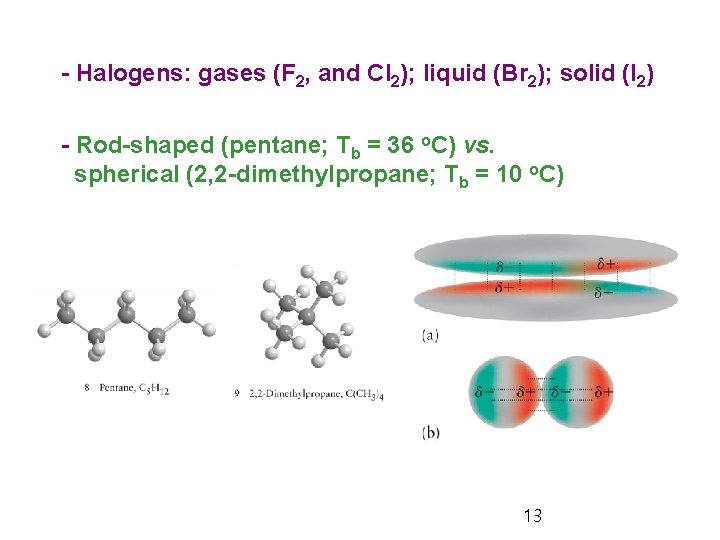
- Halogens: gases (F 2, and Cl 2); liquid (Br 2); solid (I 2) - Rod-shaped (pentane; Tb = 36 o. C) vs. spherical (2, 2 -dimethylpropane; Tb = 10 o. C) 13

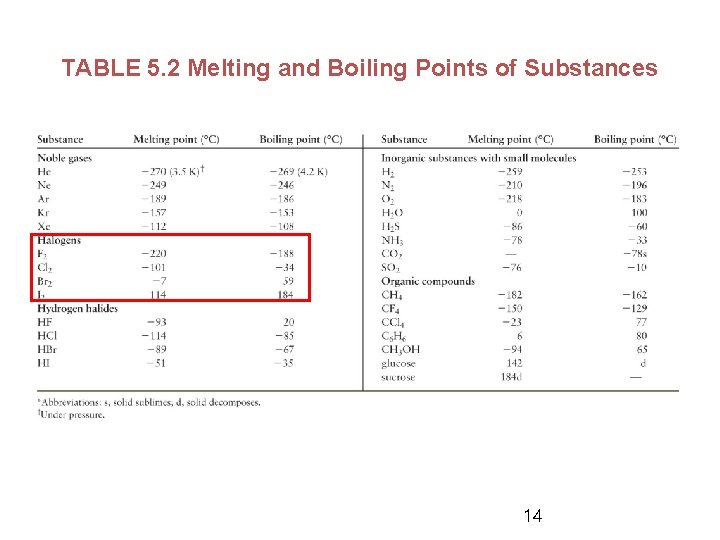
TABLE 5. 2 Melting and Boiling Points of Substances 14

Allied intermolecular interactions Ø Dipole-induced dipole interaction between a polar molecule and a nonpolar molecule (~2 k. J mol-1) – Van der Waals interactions is the collective name for dipole-dipole forces between rotating polar molecules, London forces, and dipole-induced dipole forces 15

EXAMPLE 5. 2 Explain the trend in the boiling points of the hydrogen halides: HCl, -85 o. C; HBr, -67 o. C; HI, -35 o. C. - Electronegativity differences: HCl > HBr > HI - Number of electrons and London forces: HCl < HBr < HI → not by dipole-dipole forces, but by London forces 16

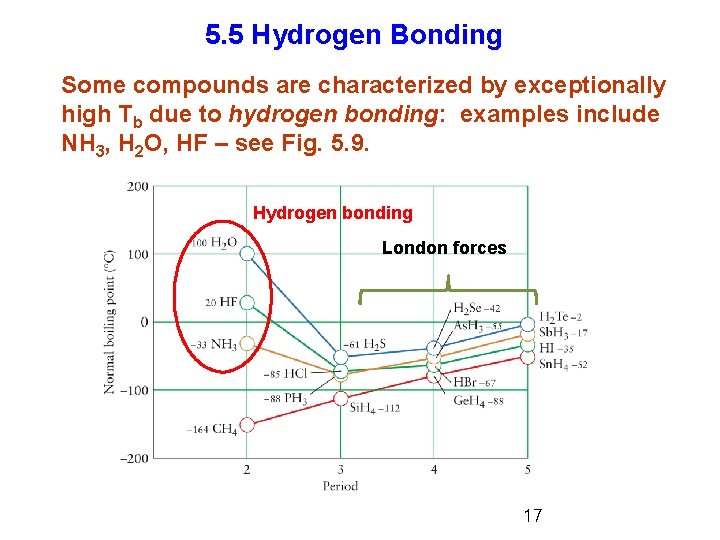
5. 5 Hydrogen Bonding Some compounds are characterized by exceptionally high Tb due to hydrogen bonding: examples include NH 3, H 2 O, HF – see Fig. 5. 9. Hydrogen bonding London forces 17

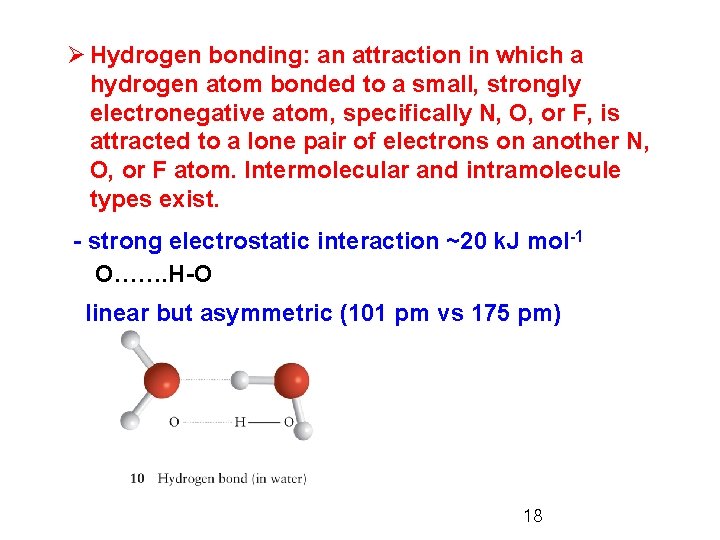
Ø Hydrogen bonding: an attraction in which a hydrogen atom bonded to a small, strongly electronegative atom, specifically N, O, or F, is attracted to a lone pair of electrons on another N, O, or F atom. Intermolecular and intramolecule types exist. - strong electrostatic interaction ~20 k. J mol-1 O……. H-O linear but asymmetric (101 pm vs 175 pm) 18

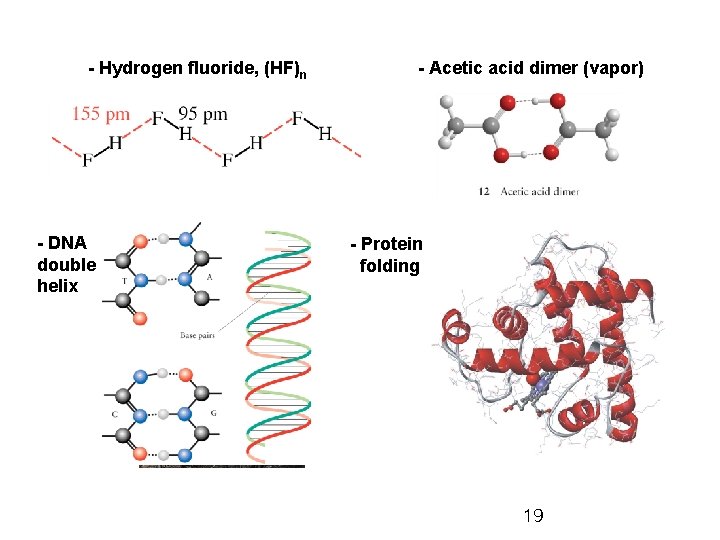
- Hydrogen fluoride, (HF)n - DNA double helix - Acetic acid dimer (vapor) - Protein folding 19

20

179 s Example 5. 1 Identify the kinds of intermolecular forces that might arise between molecules of each of the following substances: (a) NH 2 OH; (b) CBr 4; (c) H 2 Se. O 4; (d) SO 2 Solution 2013 General Chemistry I 21

179 s Example 5. 5 Suggest, giving reasons, which substance in each of the following pairs is likely to have the higher normal melting point (Lewis structures may help your arguments): (a) HCl or Na. Cl; (b) C 2 H 5 OC 2 H 5 (diethyl ether) or C 4 H 9 OH (butanol); (c) CHI 3 or CHF 3; (d) C 2 H 4 or CH 3 OH. Solution 2013 General Chemistry I 22

5. 6 Repulsions -Intermolecular repulsions arise from the overlap of orbitals on neighboring molecules and the requirements of the Pauli exclusion principle. - They are important only at very short distances: 23

LIQUID STRUCTURE (Sections 5. 7 -5. 8) 5. 7 Order in Liquids - The liquid phase lies between the extremes of the gas and solid phases. gas phase: moving with almost complete freedom minimal intermolecular forces solid phase: locked in place by intermolecular forces oscillate around an average location 24

- In the liquid phase, molecules have short-range order but not long-range order. - Water loses only 10% of hydrogen bonds upon melting and the rest are continuously broken and reformed. 25

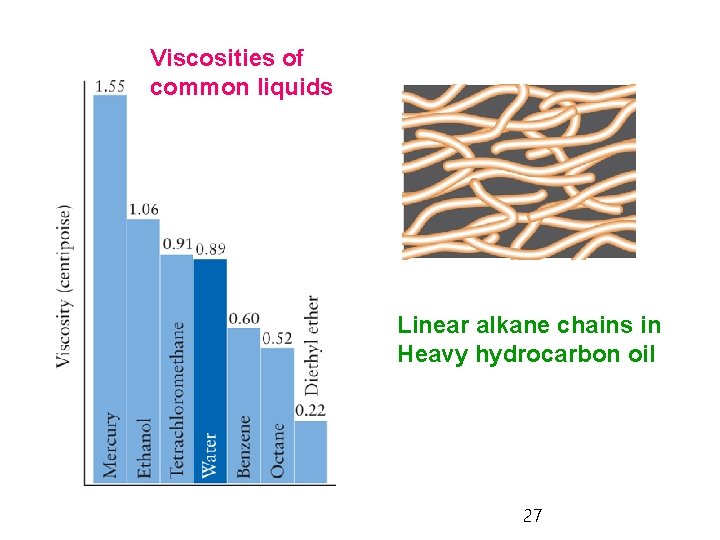
5. 8 Viscosity and Surface Tension Ø Viscosity: resistance to flow, indication of the intermolecular force strength - Water and glycerol: very viscous due to hydrogen bonding - Hydrocarbon oils and grease: viscous due to tangling long chains - Viscosity usually decreases with temperature due to higher energy of molecules. 26

Viscosities of common liquids Linear alkane chains in Heavy hydrocarbon oil 27

Ø Surface tension: the net inward pull, an indication of the intermolecular force strength - Water: three times larger than many other liquids, due to hydrogen bonds - Mercury: more than six times that of water, partially covalent 28

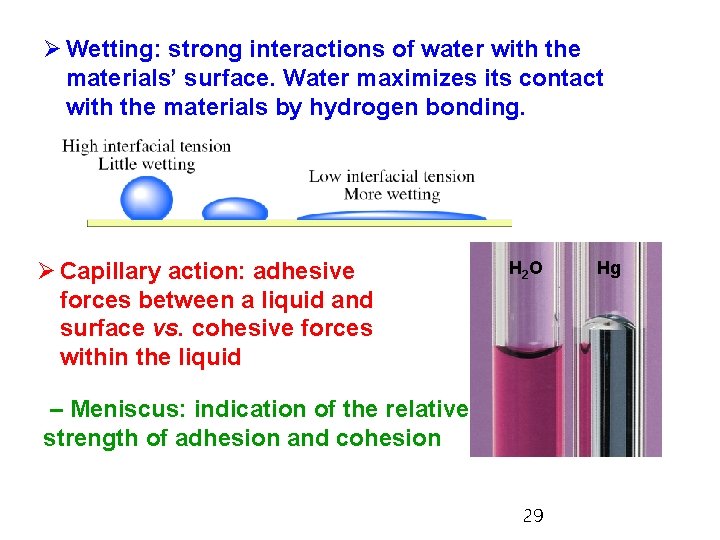
Ø Wetting: strong interactions of water with the materials’ surface. Water maximizes its contact with the materials by hydrogen bonding. Ø Capillary action: adhesive forces between a liquid and surface vs. cohesive forces within the liquid H 2 O – Meniscus: indication of the relative strength of adhesion and cohesion 29 Hg

181 s Example 5. 21 Predict how each of the following properties of a liquid varies as the strength of intermolecular forces increases and explain your reasoning: (a) boiling point; (b) viscosity; (c) surface tension. Solution 2013 General Chemistry I 30

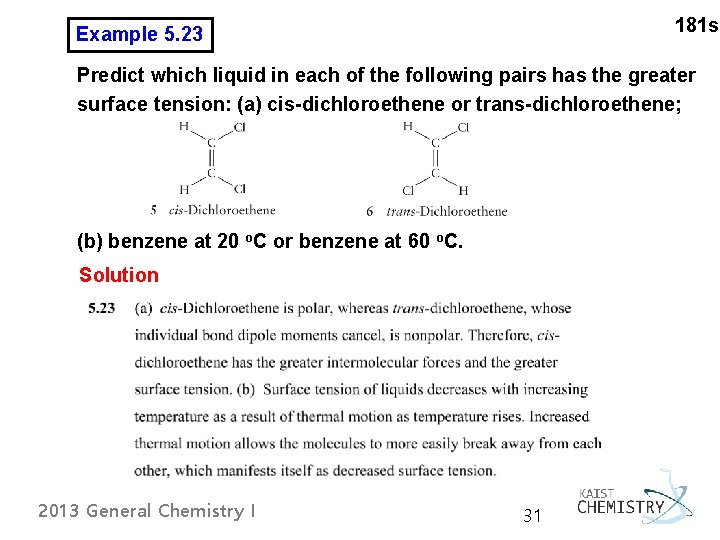
181 s Example 5. 23 Predict which liquid in each of the following pairs has the greater surface tension: (a) cis-dichloroethene or trans-dichloroethene; (b) benzene at 20 o. C or benzene at 60 o. C. Solution 2013 General Chemistry I 31

181 s Example 5. 25 Rank the following molecules in order of increasing viscosity at 50 o. C: C 6 H 5 SH, C 6 H 5 OH, C 6 H 6. Solution 2013 General Chemistry I 32

Chapter 5. LIQUIDS AND SOLIDS SOLID STRUCTURES 5. 9 Classification of Solids 5. 10 Molecular Solids 5. 11 Network Solids 5. 12 Metallic Solids 5. 13 Unit Cells 5. 14 Ionic Structures THE IMPACT ON MATERIALS 5. 15 Liquid Crystals 5. 16 Ionic Liquids 2012 General Chemistry I 33

SOLID STRUCTURES (Sections 5. 9 -5. 14) 5. 9 Classification of Solids Ø Crystalline solid: a solid in which the atoms, ions, or molecules lie in an orderly array with crystal faces Ø Amorphous solid: one in which the atoms, ions, or molecules lie in a random jumble quartz amorphous silica 34

Classification of Crystalline Solids According to the bonds that hold their atoms, ions, or molecules in place: Metallic: consisting of cations held together by a sea of electrons Ionic: built from the mutual attractions of cations and anions Molecular: assemblies of discrete molecules held in place by intermolecular forces Network: consisting of atoms covalently bonded to their neighbors throughout the extent of the solid 35

36

182 s Example 5. 35 Classify each of the following solids as ionic, network, metallic, or molecular: (a) quartz, Si. O 2; (b) limestone, Ca. CO 3; (c) dry ice, CO 2; (d) sucrose, C 12 H 22 O 11; (e) polyethylene, a polymer with molecules consisting of chains of thousands of repeating –CH 2 - units. Solution 2013 General Chemistry I 37

5. 10 Molecular Solids Molecular solids consist of molecules held together by intermolecular forces; physical properties depend on the strengths of those forces. Amorphous molecular solids: as soft as paraffin wax Crystalline molecular solids: - sucrose: numerous hydrogen bonds between OH groups account for high melting point at 184 o. C - ultrahigh-density polyethylene: smooth yet tough 38

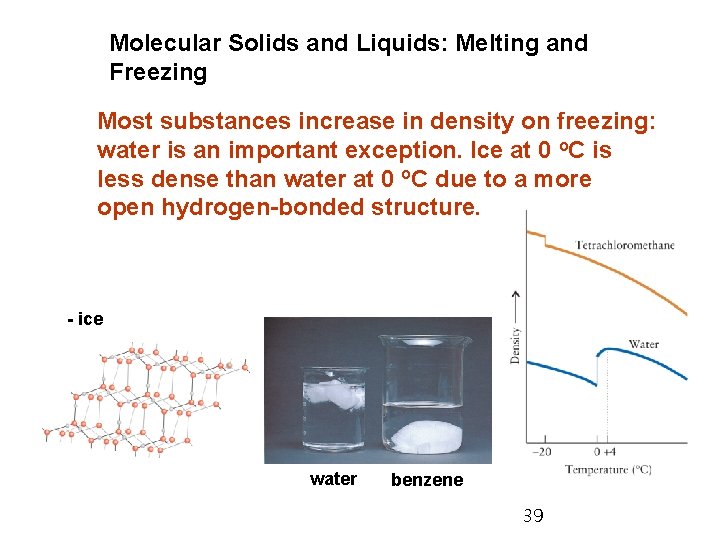
Molecular Solids and Liquids: Melting and Freezing Most substances increase in density on freezing: water is an important exception. Ice at 0 o. C is less dense than water at 0 o. C due to a more open hydrogen-bonded structure. - ice water benzene 39

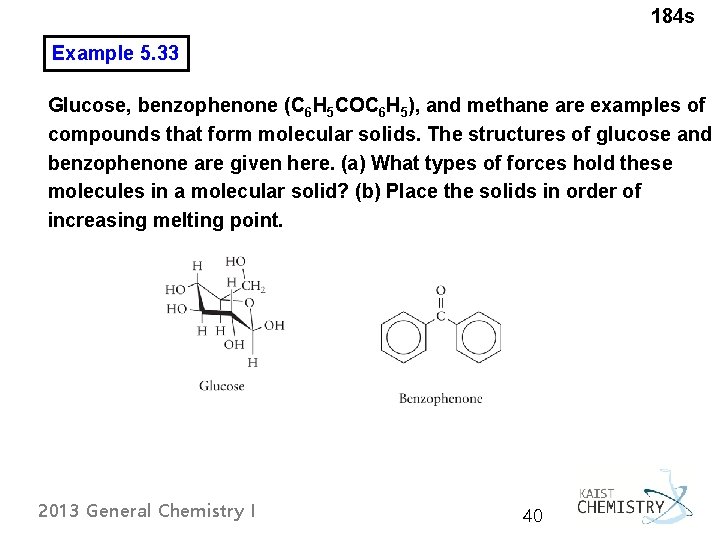
184 s Example 5. 33 Glucose, benzophenone (C 6 H 5 COC 6 H 5), and methane are examples of compounds that form molecular solids. The structures of glucose and benzophenone are given here. (a) What types of forces hold these molecules in a molecular solid? (b) Place the solids in order of increasing melting point. 2013 General Chemistry I 40

184 s Solution 2013 General Chemistry I 41

5. 11 Network Solids Network solids are characterized by a strong covalent bond network throughout the crystal: they are very hard and rigid, with high Tm and Tb E. g. Two common allotropes (forms of an element that differ in the way in which the atoms are linked) of carbon have very different network structures. Diamond, with an sp 3 hybrid s-bonding framework, is one of the hardest substances 42

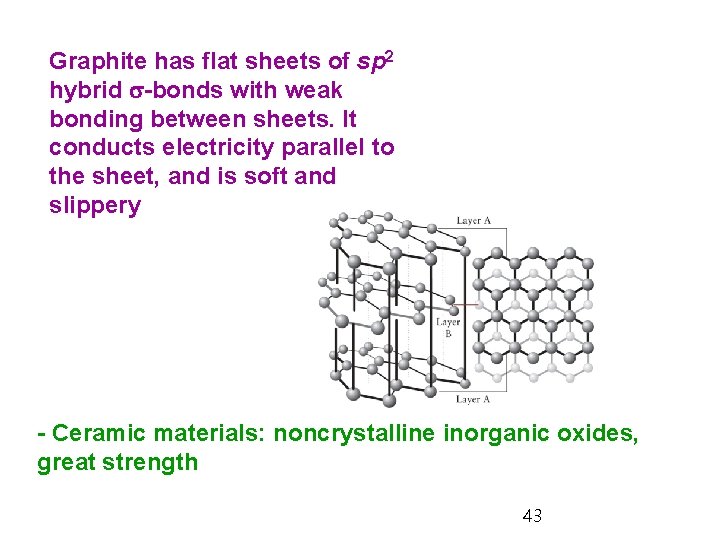
Graphite has flat sheets of sp 2 hybrid s-bonds with weak bonding between sheets. It conducts electricity parallel to the sheet, and is soft and slippery - Ceramic materials: noncrystalline inorganic oxides, great strength 43

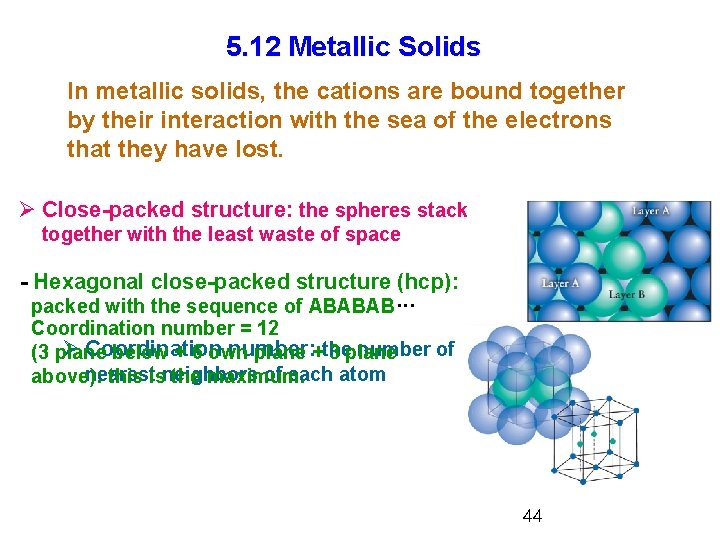
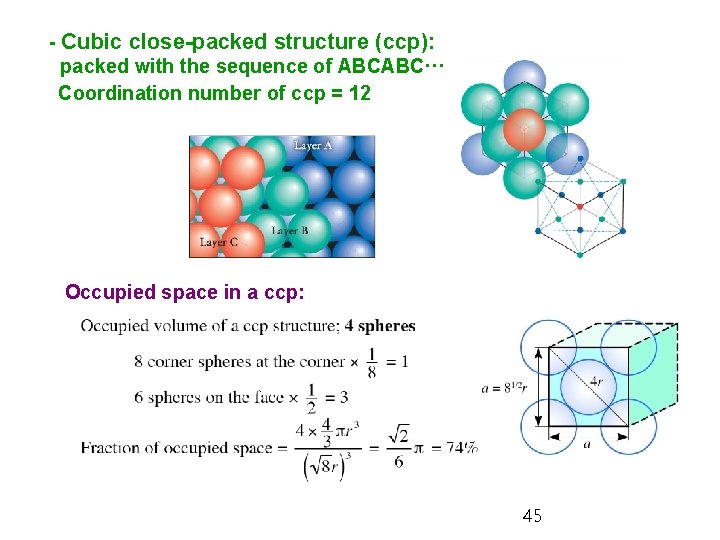
5. 12 Metallic Solids In metallic solids, the cations are bound together by their interaction with the sea of the electrons that they have lost. Ø Close-packed structure: the spheres stack together with the least waste of space - Hexagonal close-packed structure (hcp): packed with the sequence of ABABAB··· Coordination number = 12 Ø Coordination number: number of (3 plane below + 6 own plane +the 3 plane nearest of each atom above): this isneighbors the maximum. 44

- Cubic close-packed structure (ccp): packed with the sequence of ABCABC··· Coordination number of ccp = 12 Occupied space in a ccp: 45

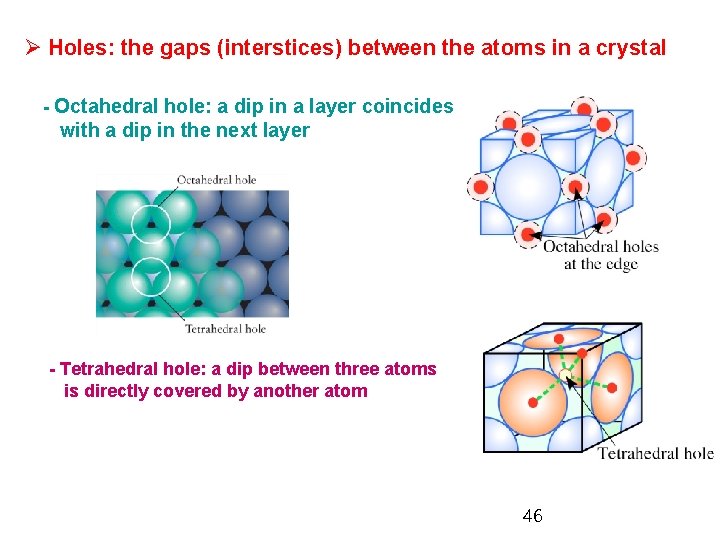
Ø Holes: the gaps (interstices) between the atoms in a crystal - Octahedral hole: a dip in a layer coincides with a dip in the next layer - Tetrahedral hole: a dip between three atoms is directly covered by another atom 46

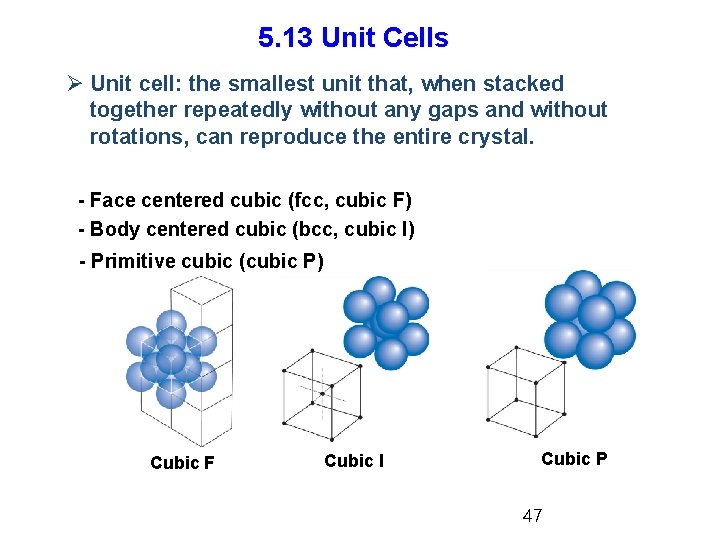
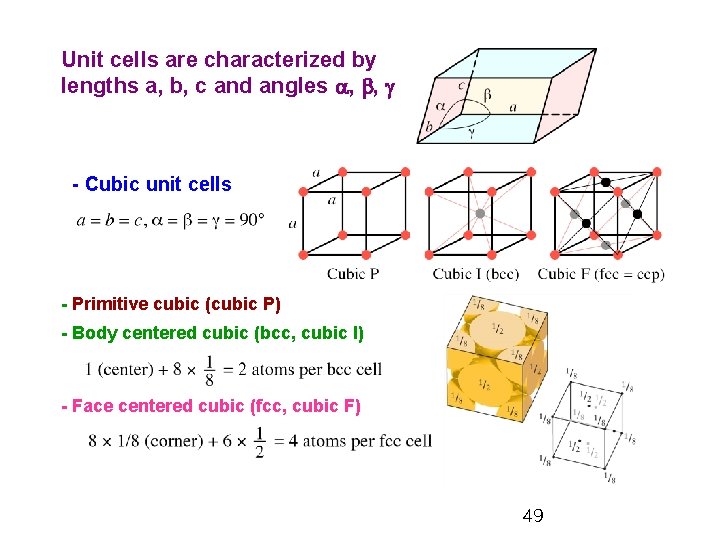
5. 13 Unit Cells Ø Unit cell: the smallest unit that, when stacked together repeatedly without any gaps and without rotations, can reproduce the entire crystal. - Face centered cubic (fcc, cubic F) - Body centered cubic (bcc, cubic I) - Primitive cubic (cubic P) Cubic F Cubic I Cubic P 47

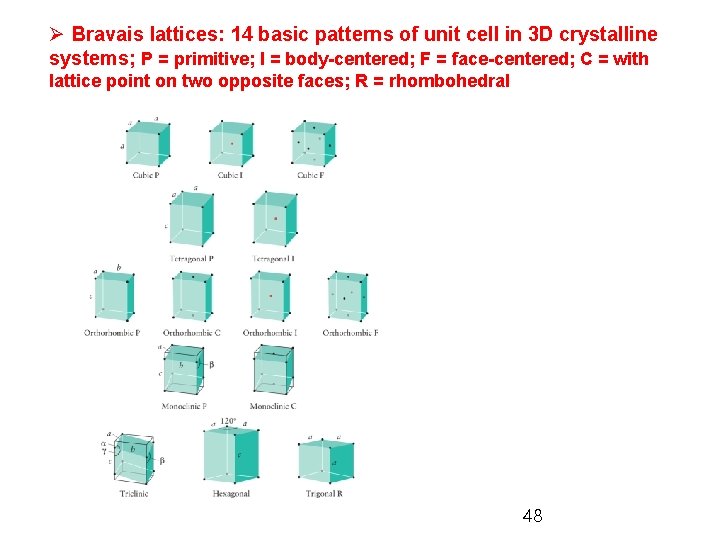
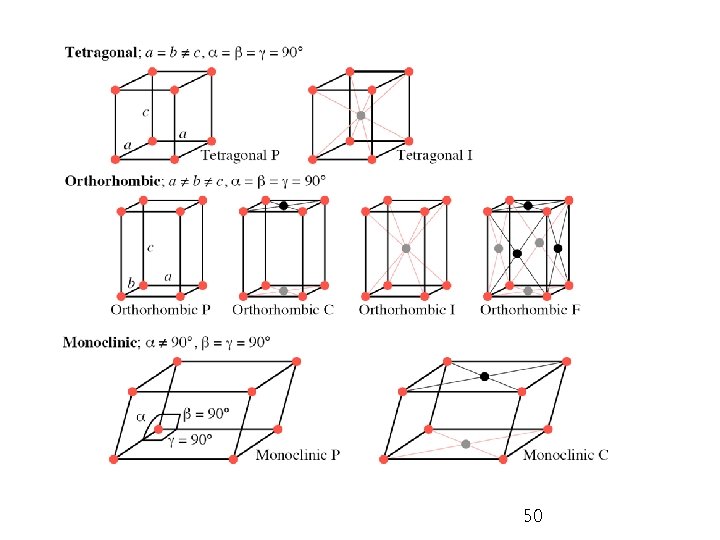
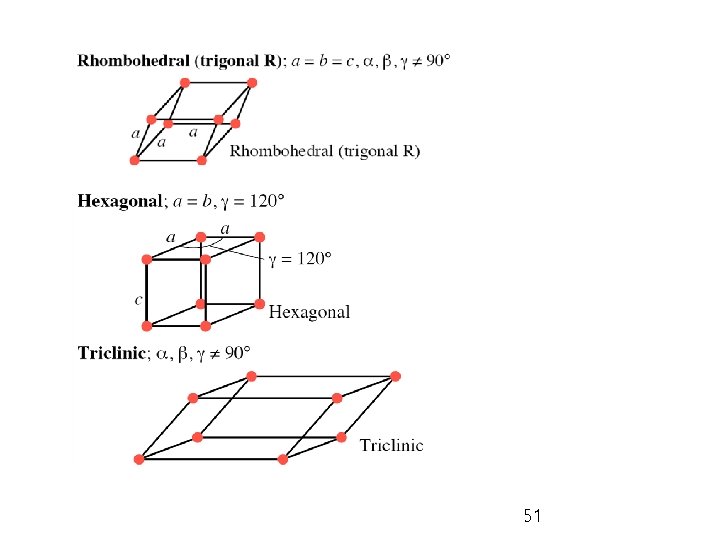
Ø Bravais lattices: 14 basic patterns of unit cell in 3 D crystalline systems; P = primitive; I = body-centered; F = face-centered; C = with lattice point on two opposite faces; R = rhombohedral 48

Unit cells are characterized by lengths a, b, c and angles a, b, g - Cubic unit cells - Primitive cubic (cubic P) - Body centered cubic (bcc, cubic I) - Face centered cubic (fcc, cubic F) 49

50

51

52

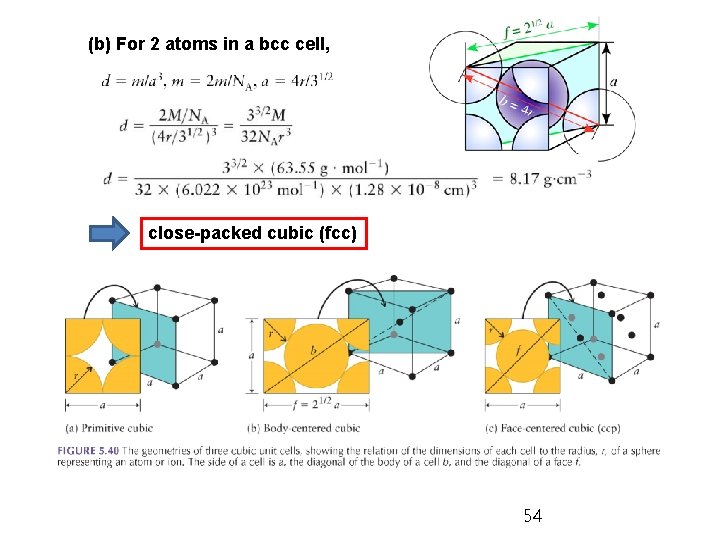
EXAMPLE 5. 3 The density of copper is 8. 93 gcm-3 and its atomic radius is 128 pm. Is the metal (a) close-packed or (b) body-centered cubic? (a) Fcc (ccp) and hcp cannot be distinguished by density only. For 4 atoms in a fcc cell, 53

(b) For 2 atoms in a bcc cell, close-packed cubic (fcc) 54

55

190 s Example 5. 47 One form of silicon has density of 2. 33 gcm-3 and crystallizes in a cubic Lattice with a unit cell edge of 543 pm. (a) What is the mass of each unit cell? (b) How many silicon atoms does one unit cell contain? Solution 2013 General Chemistry I 56

190 s Example 5. 49 What percentage of space is occupied by close-packed cylinders of length l and radius r ? Solution 2013 General Chemistry I 57

5. 14 Ionic Structures - Ionic structures are (like metallic structures) close packed, with anions forming a slightly expanded close-packed structure with smaller cations occupying some of the enlarged holes in the expanded lattice. - Smaller tetrahedral hole for small cations, larger octahedral hole for somewhat bigger cations Ø Coordination number: the number of ions of opposite charge immediately surrounding a specific ion Ø Radius ratio (r) 58

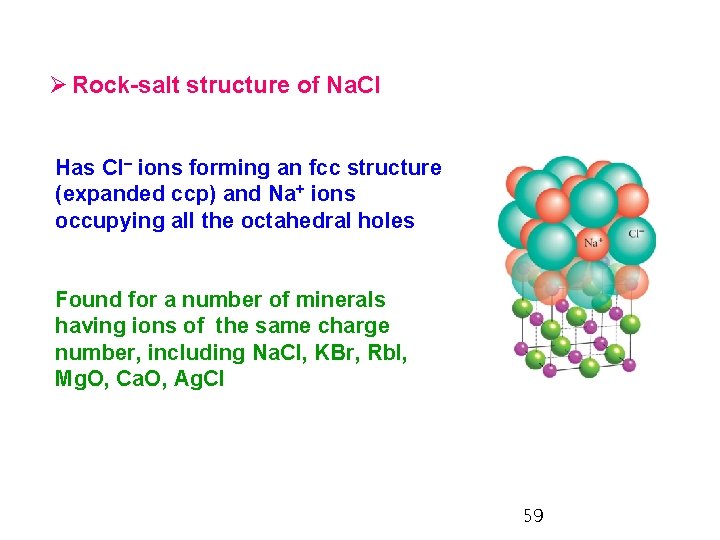
Ø Rock-salt structure of Na. Cl Has Cl– ions forming an fcc structure (expanded ccp) and Na+ ions occupying all the octahedral holes Found for a number of minerals having ions of the same charge number, including Na. Cl, KBr, Rb. I, Mg. O, Ca. O, Ag. Cl 59

- Rock-salt (fcc) structure: (6, 6) coordination, the coordination numbers of the cations and the anions are both 6. - Common whenever the cations and the anions have very different radii; the cations can fit into the octahedral holes in a fcc array of anions; 0. 4 < r < 0. 7 60

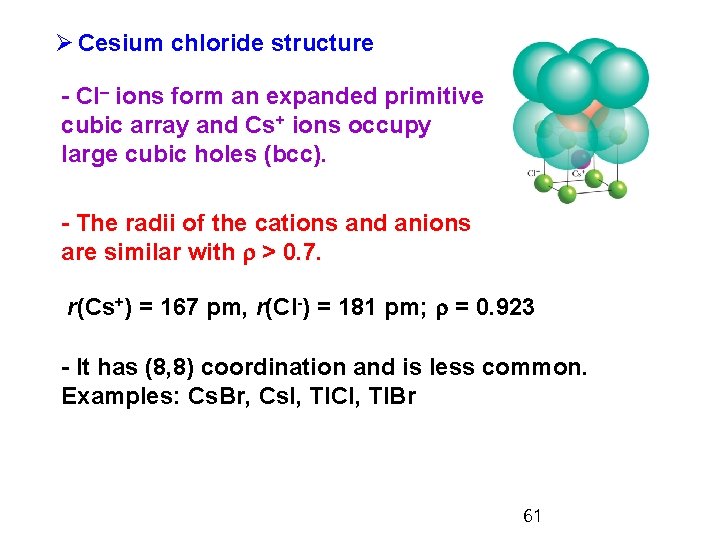
Ø Cesium chloride structure - Cl– ions form an expanded primitive cubic array and Cs+ ions occupy large cubic holes (bcc). - The radii of the cations and anions are similar with r > 0. 7. r(Cs+) = 167 pm, r(Cl-) = 181 pm; r = 0. 923 - It has (8, 8) coordination and is less common. Examples: Cs. Br, Cs. I, Tl. Cl, Tl. Br 61

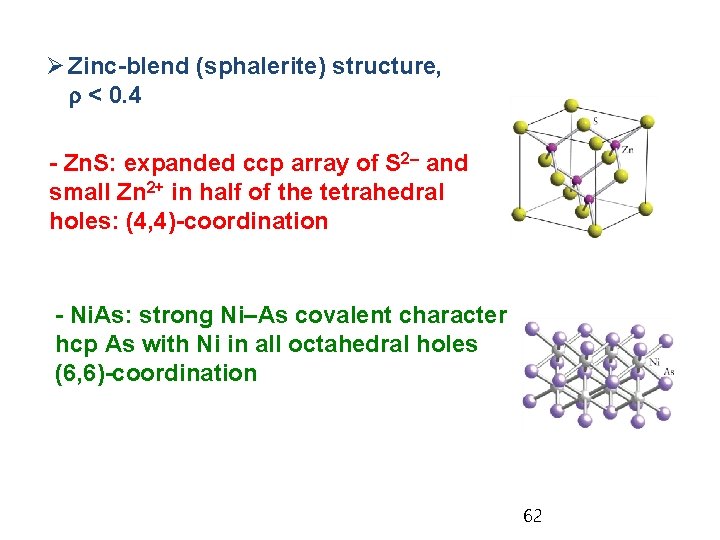
Ø Zinc-blend (sphalerite) structure, r < 0. 4 - Zn. S: expanded ccp array of S 2– and small Zn 2+ in half of the tetrahedral holes: (4, 4)-coordination - Ni. As: strong Ni–As covalent character hcp As with Ni in all octahedral holes (6, 6)-coordination 62

63

64

192 s Example 5. 53 Calculate the number of cations, and formula units per unit cell in each of the following solids: (a) the cesium chloride unit cell Solution 2013 General Chemistry I 65

192 s (b) The rutile (Ti. O 2) unit cell (c) What are the coordination numbers of the ions in rutile? Solution 2013 General Chemistry I 66

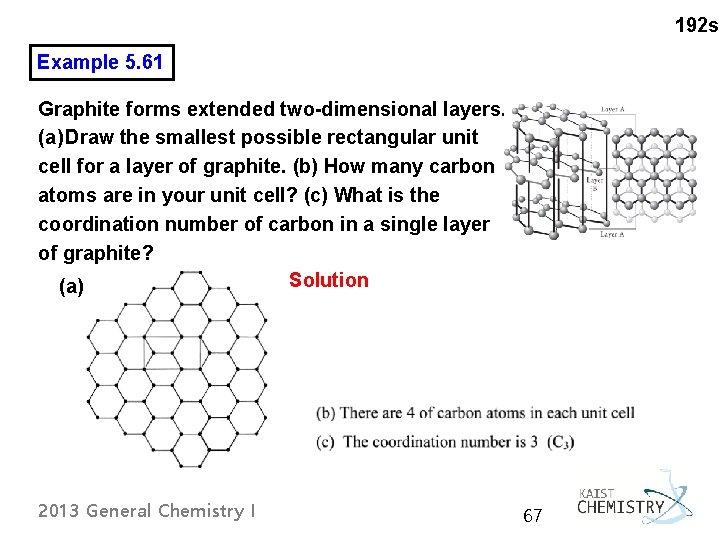
192 s Example 5. 61 Graphite forms extended two-dimensional layers. (a) Draw the smallest possible rectangular unit cell for a layer of graphite. (b) How many carbon atoms are in your unit cell? (c) What is the coordination number of carbon in a single layer of graphite? Solution (a) 2013 General Chemistry I 67

190 s Example 5. 63 If the edge length of a fcc unit cell of Rb. I is 732. 6 pm, how long would the edge of a cubic single crystal of Rb. I be that contains 1. 00 mol Rb. I? Solution 2013 General Chemistry I 68

THE IMPACT ON MATERIALS (Sections 5. 15 -5. 16) 5. 15 Liquid Crystals Ø Liquid crystals are substances that flow like viscous liquids, but their molecules lie in a moderately orderly array. - mesophase: an intermediate state of matter with the fluidity of a liquid and some of the molecular order of a solid - responsive to changes in temperature and electric fields - isotropic vs. anisotropic (due to ordering of rodlike molecules) - p-azoxyanisole, a long and rodlike liquid crystal molecule 69

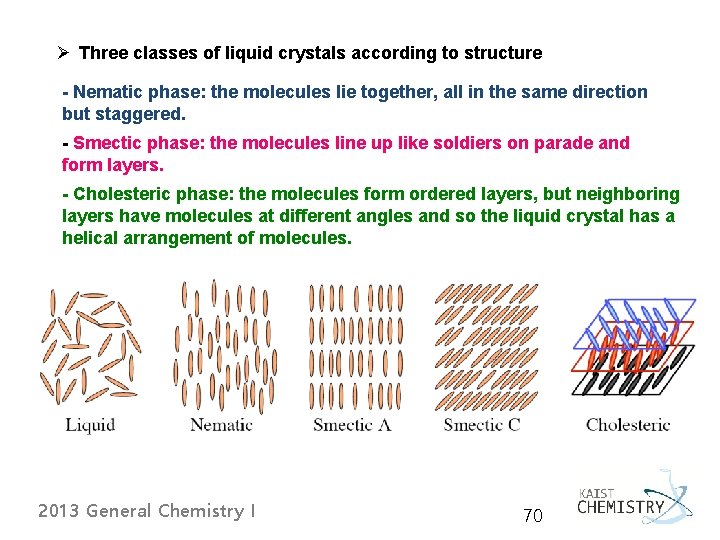
Ø Three classes of liquid crystals according to structure - Nematic phase: the molecules lie together, all in the same direction but staggered. - Smectic phase: the molecules line up like soldiers on parade and form layers. - Cholesteric phase: the molecules form ordered layers, but neighboring layers have molecules at different angles and so the liquid crystal has a helical arrangement of molecules. 2013 General Chemistry I 70

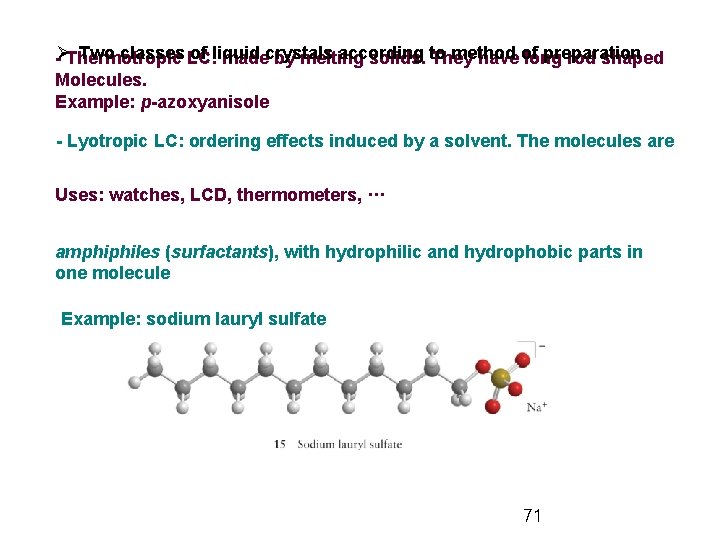
Two classes LC: of liquid according method preparation -ØThermotropic madecrystals by melting solids. to They have of long rod shaped Molecules. Example: p-azoxyanisole - Lyotropic LC: ordering effects induced by a solvent. The molecules are Uses: watches, LCD, thermometers, ··· amphiphiles (surfactants), with hydrophilic and hydrophobic parts in one molecule Example: sodium lauryl sulfate 71

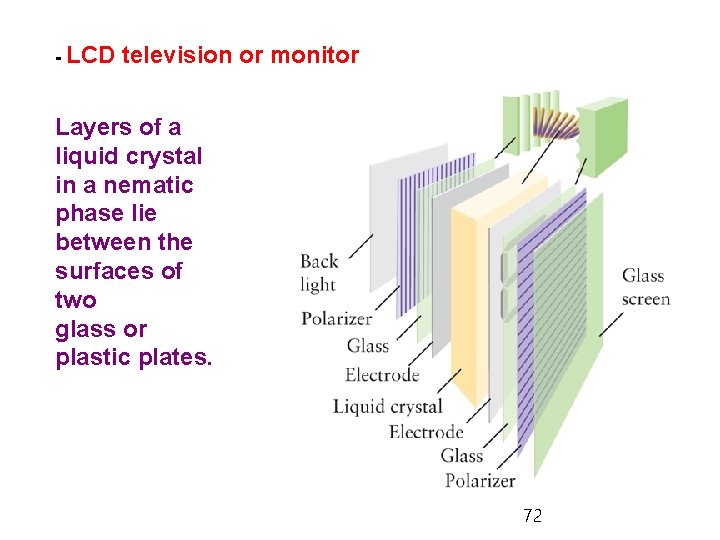
- LCD television or monitor Layers of a liquid crystal in a nematic phase lie between the surfaces of two glass or plastic plates. 72

190 s Example 5. 65 Why do long hydrocarbon molecules that do not have multiple bonds, such as decane, CH 3(CH 2)8 CH 3, not form liquid crystals? Solution 2013 General Chemistry I 73

190 s Example 5. 67 When long surfactant molecule having a polar headgroup and a nonpolar “tail” are placed into water, micelles are formed in which the nonpolar tails aggregate, with the polar headgroups pointing out toward the solvent. Inverse micelles are similar but have the nonpolar regions pointing outward. How can inverse micelles be placed? Solution 2013 General Chemistry I 74

5. 16 Ionic Liquids - A liquid at room temperature is likely to be a molecular substance. There will be nonionic, weak intermolecular interactions and it will have a relatively high vapor pressure. Ø Ionic liquids: These are characterized by a relatively small anions (BF 4–) + large organic cation (e. g. 1 -butyl-3 methylimidazolium ion), preventing crystallization. Low vapor pressure, novel solvent properties: reducing pollution 75

X-RAY DIFFRACTION Ø The Technique - interference: When two or more waves pass through the same region, interference is observed as an increase (constructive) or a decrease (destructive) in the total amplitude of the wave. - diffraction: interference between waves that arises when there is an object in their path - Regular layers of atoms in a crystal giving a diffraction pattern 76

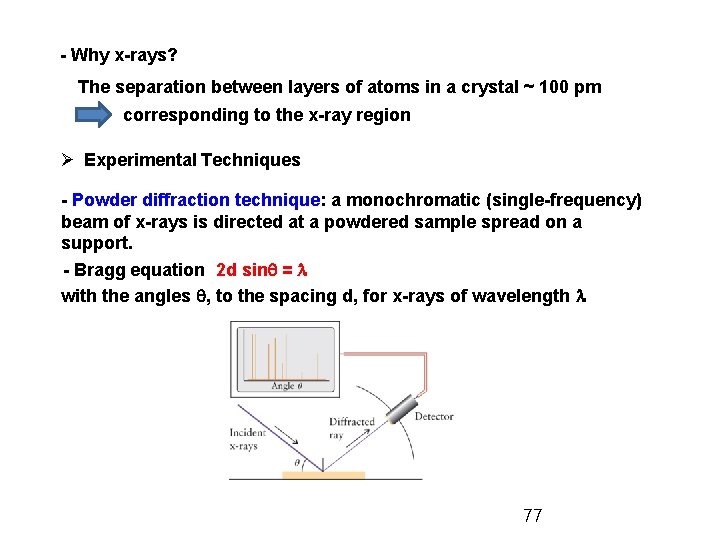
- Why x-rays? The separation between layers of atoms in a crystal ~ 100 pm corresponding to the x-ray region Ø Experimental Techniques - Powder diffraction technique: a monochromatic (single-frequency) beam of x-rays is directed at a powdered sample spread on a support. - Bragg equation 2 d sinq = l with the angles q, to the spacing d, for x-rays of wavelength l 77

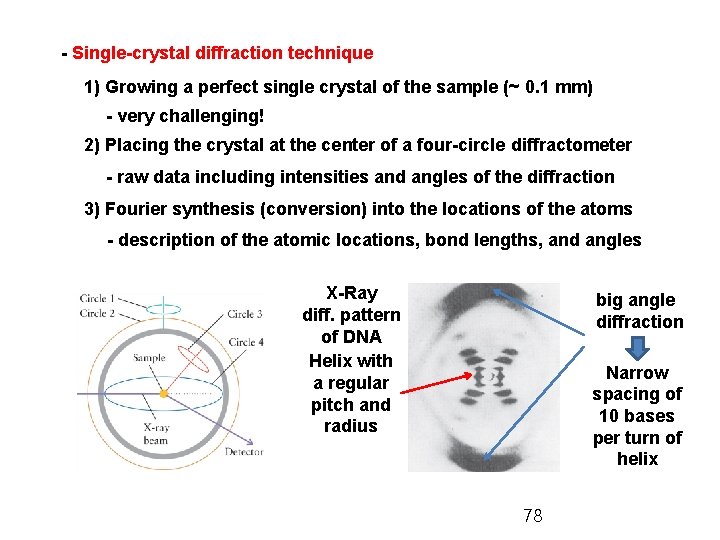
- Single-crystal diffraction technique 1) Growing a perfect single crystal of the sample (~ 0. 1 mm) - very challenging! 2) Placing the crystal at the center of a four-circle diffractometer - raw data including intensities and angles of the diffraction 3) Fourier synthesis (conversion) into the locations of the atoms - description of the atomic locations, bond lengths, and angles X-Ray diff. pattern of DNA Helix with a regular pitch and radius big angle diffraction Narrow spacing of 10 bases per turn of helix 78