CHAPTER 5 Leukocytic Disorders 1 Acknowledgements Addisa Ababa

CHAPTER 5 Leukocytic Disorders 1

Acknowledgements • • Addisa Ababa University Jimma University Haramaya University Hawassa University of Gondar American Society for Clinical Pathology Center for Disease Control and Prevention-Ethiopia 2

Learning Objectives At the end of this session, students will be able to: • • Describe the classification of leukocytic disorders Describe quantitative leukocytic disorders Describe qualitative/functional leukocytic disorders Identify the morphologic characteristics of normal and abnormal granulocytes 3

Outline • • Definition of terms Classification of leukocytic disorders Non malignant leukocytic disorders Malignant leukocytic disorders 4

Definition of Terms • Leucocytosis: an increased number of leucocytes in the peripheral blood • Cytopenia: reduction in the number of cells in the peripheral blood, e. g. neutropenia, thrombocytopenia, pancytopenia • Leukemoid reaction: leukemoid reaction a reactive condition which simulates or closely resembles leukemia • Leukaemia: a myeloid or lymphoid neoplasm, characterized by circulating neoplastic cells but also encompassing similar cases in which there are neoplastic cells in the bone marrow but not in the peripheral blood • Vacuole: a fluid-filled cavity within a cell which, in the case of phagocytes, is formed by invagination of the surface membrane • Toxic granulation: increased staining of neutrophil granules occurring as a response to infection and inflammation but also as a physiological change during pregnancy 5

Definition of Terms Cont’d • Auer rod: a rod-shaped crystalline structure derived from primary granules, found in the cytoplasm of cells of granulocytic and, less often, monocytic lineage, observed in acute myeloid and monocytic leukemias • Döhle body: a weakly basophilic inclusion within the cytoplasm of a neutrophil composed of ribosomes toxic granulation increased staining of neutrophil granules occurring as a response to infection and inflammation but also as a physiological change during pregnancy • Malignant: very injurious; in relation to neoplasms, aggressive and characterized by local invasion, distant spread (metastasis) and cytological differences from the equivalent normal tissue 6

Leukocyte disorders • Classification: 1. Non-Malignant 2. Malignant • 1. Non-malignant (Reactive) Quantitative Disorders • Leukocytosis Vs Leukopenia • Leukocytosis occurs mostly due to Neutrophilia because of their predominance in the circulating blood 7

Reactive Leukocyte disorders cont’d Non-malignant leukocytosis may be caused by: • Increased movement of immature cells out of the bone marrow proliferation compartment • Increased mobilization of cells from the maturation storage compartment of the bone marrow to the peripheral blood • Decreased movement of mature cells from the circulation into tissues 8

Reactive Neutrophilia § Assess for toxic changes and multilineage abnormalities. Toxic changes such as Toxic granulation, Dohle bodies, prominent cytoplasmic Vacoules, may be found when sepsis is present and in exceptional cases, intra- and extracellular bacteria and fungi may be identified. § Evaluate for bacterial infections § Blood findings with clinical features & chemical analysis, cytogenetic analysis if a neoplastic disorder is suspected as the presumptive diagnostic consideration 9

Quantitative Leukocyte Disorders • Neutrophil leukocytosis – Infections, drugs, inflammation • Note: Leukemoid Reaction needs to be differentiated from Chronic Myeloid Leukemia – Leukocytosis with marked left shift of immature neutrophils; bands, myelocyte, metamyelocyte – Infection, malignancy, burn, hemolysis, hemorrhage – Differential: chronic myelocytic leukemia (CML) • Increase in granulocytes, Toxic granulation, Dohle body, vaculation in granulocytes, increased Leukocyte Alkaline Phosphatase (LAP) score

Quantitative Leukocyte Disorders Cont’d • Neutropenia: decrease in neutrophils • May be acquired (e. g. overwhelming sepsis, drugs, viral, radiation, malignancies • May be inherited (e. g. cyclic neutropenia)

Quantitative Leukocyte Disorders • Eosinophilia – Allergy, parasites, Chronic Myelocytic Leukemia (CML), Hodgkin’s lymphoma • Basophilia – Myeloproliferative disorder (CML) • Lymphocytosis – Viral infection, autoimmune disease – Chronic lymphocytic leukemia (CLL) • Monocytosis – Infection, autoimmune disease, – Acute monocytic leukemia • Lymphopenia – Viral infection

Morphological abnormalities of mature granulocytes (especially neutrophils) Frequently observed morphological abnormalities: are acquired • • Toxic granulation Döhle bodies (Inclusion of RNA origin) Vacuolation Hypersegmentation Rarely observed genetic disorders • Pelger-Hu acquired Pelger–Huët anomaly (bi-lobed, or non-segmented nuclei)-no significant functional abnormalities • May-Hegglin anomaly (large blue cytoplasmic inclusion resembling Döhle bodies) • Chediak-Higashi syndrome (giant cytoplasmic granules)13
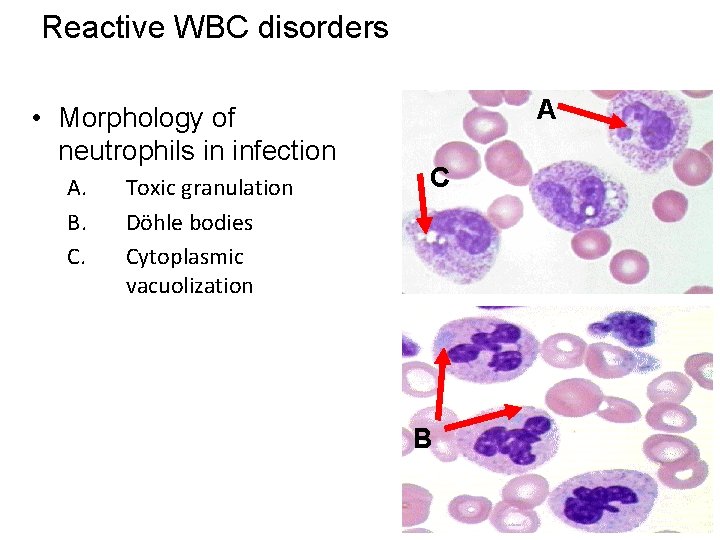
Reactive WBC disorders • Morphology of neutrophils in infection A. B. C. Toxic granulation Döhle bodies Cytoplasmic vacuolization A C B
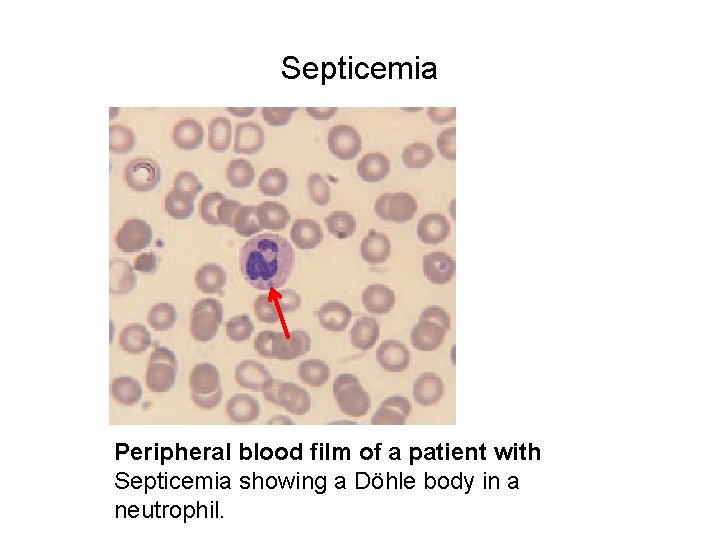
Septicemia Peripheral blood film of a patient with Septicemia showing a Döhle body in a neutrophil.
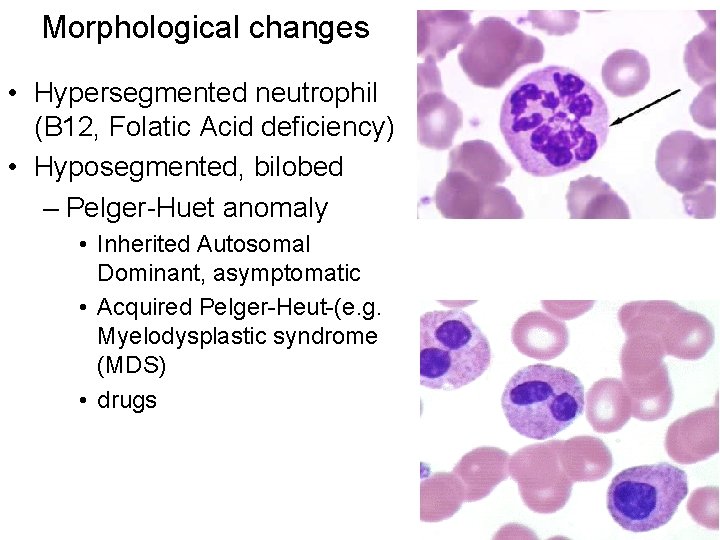
Morphological changes • Hypersegmented neutrophil (B 12, Folatic Acid deficiency) • Hyposegmented, bilobed – Pelger-Huet anomaly • Inherited Autosomal Dominant, asymptomatic • Acquired Pelger-Heut-(e. g. Myelodysplastic syndrome (MDS) • drugs
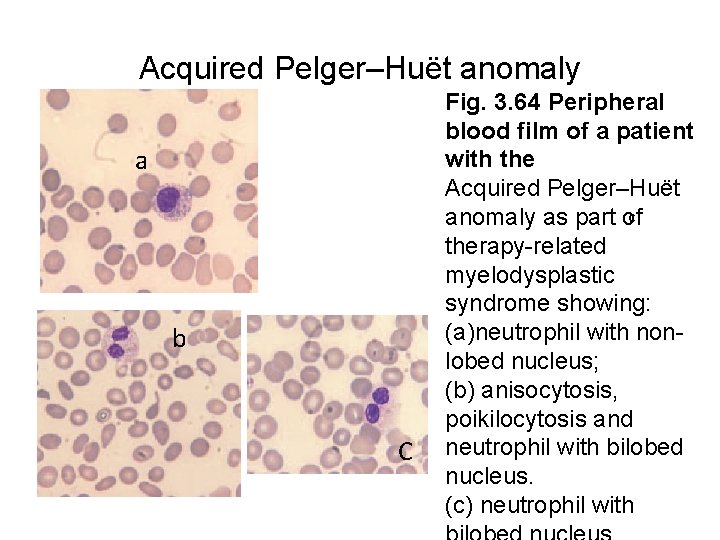
Acquired Pelger–Huët anomaly a b C Fig. 3. 64 Peripheral blood film of a patient with the Acquired Pelger–Huët c anomaly as part of therapy-related myelodysplastic syndrome showing: (a)neutrophil with nonlobed nucleus; (b) anisocytosis, poikilocytosis and neutrophil with bilobed nucleus. (c) neutrophil with
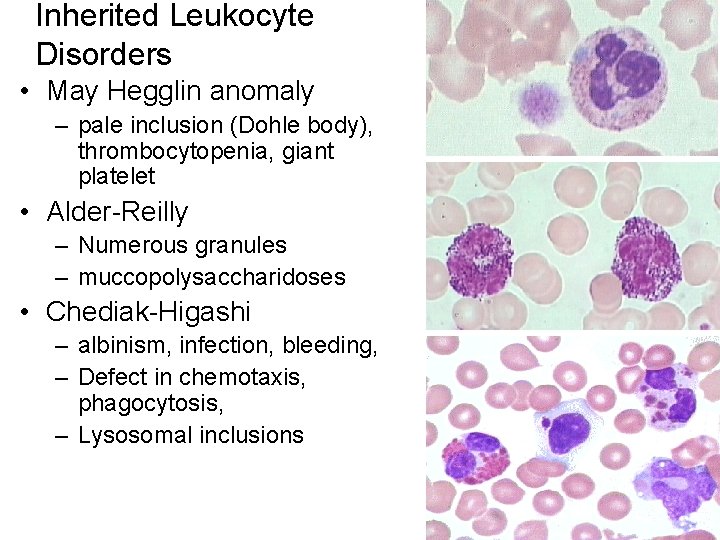
Inherited Leukocyte Disorders • May Hegglin anomaly – pale inclusion (Dohle body), thrombocytopenia, giant platelet • Alder-Reilly – Numerous granules – muccopolysaccharidoses • Chediak-Higashi – albinism, infection, bleeding, – Defect in chemotaxis, phagocytosis, – Lysosomal inclusions
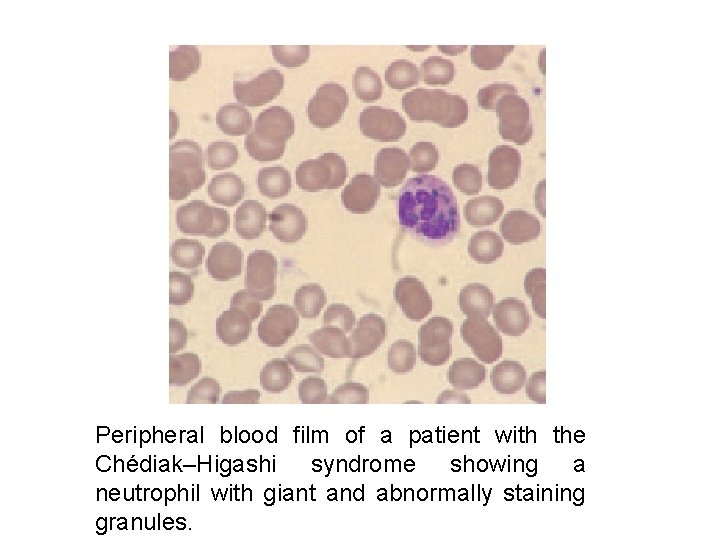
Peripheral blood film of a patient with the Chédiak–Higashi syndrome showing a neutrophil with giant and abnormally staining granules.
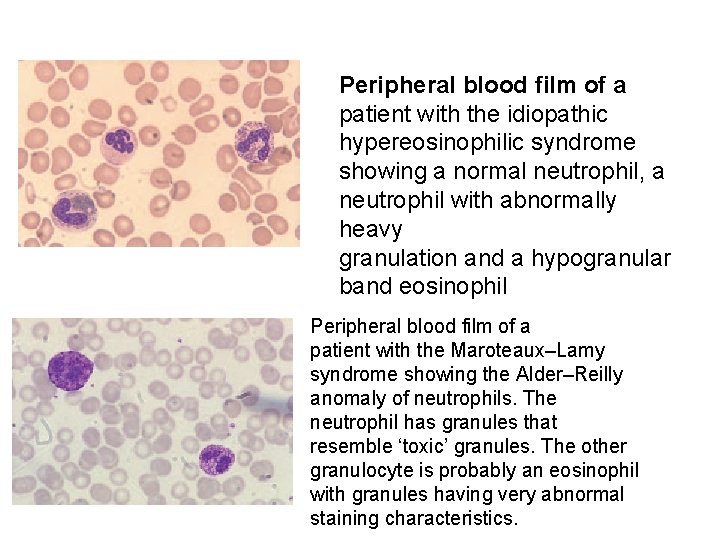
Peripheral blood film of a patient with the idiopathic hypereosinophilic syndrome showing a normal neutrophil, a neutrophil with abnormally heavy granulation and a hypogranular band eosinophil Peripheral blood film of a patient with the Maroteaux–Lamy syndrome showing the Alder–Reilly anomaly of neutrophils. The neutrophil has granules that resemble ‘toxic’ granules. The other granulocyte is probably an eosinophil with granules having very abnormal staining characteristics.
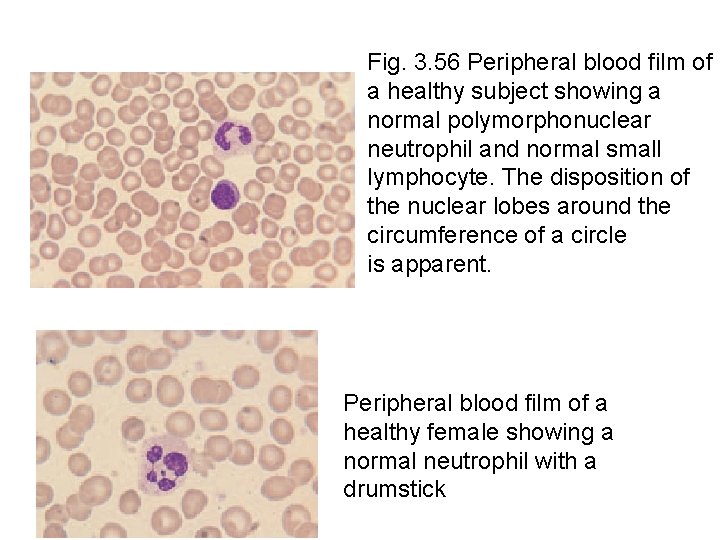
Fig. 3. 56 Peripheral blood film of a healthy subject showing a normal polymorphonuclear neutrophil and normal small lymphocyte. The disposition of the nuclear lobes around the circumference of a circle is apparent. Peripheral blood film of a healthy female showing a normal neutrophil with a drumstick
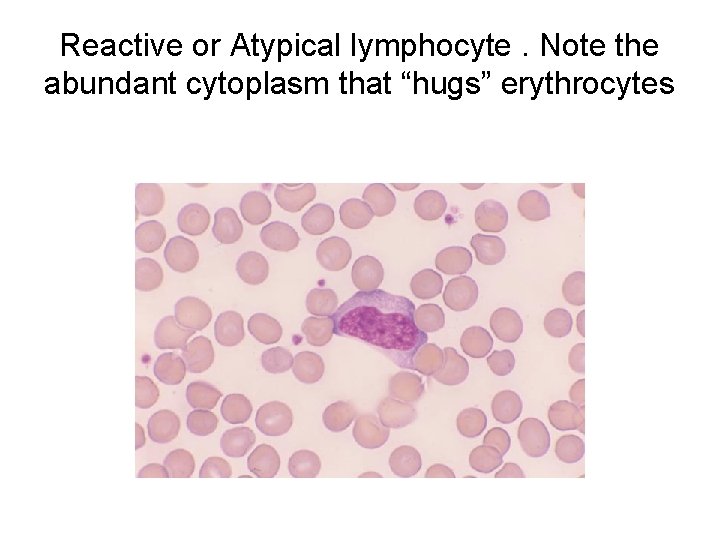
Reactive or Atypical lymphocyte. Note the abundant cytoplasm that “hugs” erythrocytes
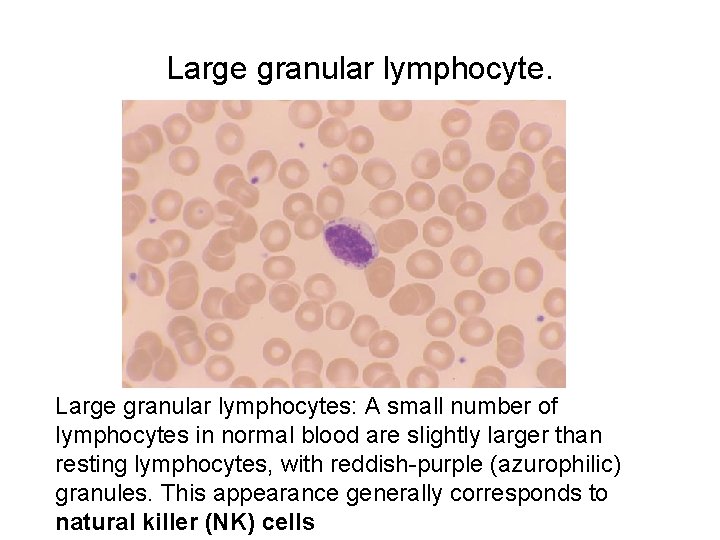
Large granular lymphocyte. Large granular lymphocytes: A small number of lymphocytes in normal blood are slightly larger than resting lymphocytes, with reddish-purple (azurophilic) granules. This appearance generally corresponds to natural killer (NK) cells
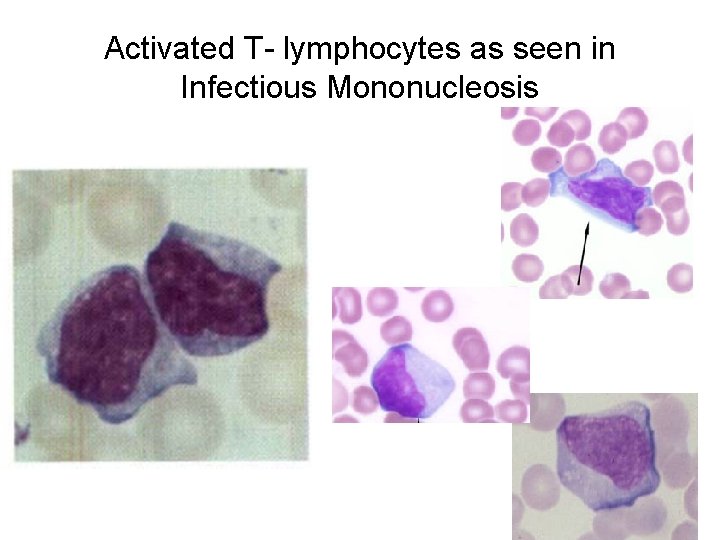
Activated T- lymphocytes as seen in Infectious Mononucleosis

HEMATOLOGIC MALIGNANCIES 25

Objectives At the end of this chapter, the student shall be able to: q Define hematologic malignancies q Explain the mechanisms of malignant transformation q Describe the classification of leukemia q Explain the diagnostic methodologies for the leukemias q Describe myelodysplastic syndromes q Define malignant lymphoma q Characterize multiple myeloma q Describe myeloproliferative disorders 26

Introduction q Hematological malignancies are clonal diseases that derive from a single cell in the marrow or peripheral lymphoid tissue which has undergone a genetic alteration q A combination of genetic and environmental factors determine the risk of developing malignancy: q. Inherited factors – genetic diseases increase the incidence of leukemia q. Down’s syndrome, Bloom’s syndrome, Fanconi’s anemia, ataxia telangiectasia q. Environmental influences q. Chemicals, drugs, radiation, infection 27

The genetics of malignant transformation q Malignant transformation - accumulation of genetic mutations in cellular genes q The genes involved in the development of cancer are divided broadly into two groups: q. Oncogenes q. Arise because of gain-of-function mutations in normal cellular genes called proto-oncogenes q. Oncogenic versions are generated when the activity of proto-oncogenes is increased or they acquire a novel function through: q. Translocation q. Mutation q. Duplication q. Tumour suppressor genes q. May acquire loss-of-function point mutation or deletion leading to malignant transformation 28

Leukemia q The leukemias are a group of disorders characterized by the accumulation of abnormal white cells in the bone marrow q These abnormal cells may cause: q. Bone marrow failure q. A raised circulating white cell count q. Infiltration of organs q Thus common but not essential features include: q. Abnormal white cells in the peripheral blood q. A raised total white cell count q. Evidence of bone marrow failure (e. g. anemia, neutropenia, thrombocytopenia) in the acute leukemias q. Involvement of other organs (e. g. , liver, spleen, lymph 29 nodes, meninges, brain, skin or testes)

Leukemia cont’d q Although viruses cause several forms of leukemia in animals, their role in humans is uncertain q. Only two viral associations are identified q. Epstein-Barr virus, a DNA virus, associated with Burkitt's lymphoma q. Human T-cell lymphotropic virus type I, called human T-cell leukemia/lymphoma virus, an RNA retrovirus, associated with some T-cell leukemias and lymphomas q Exposure to ionizing radiation and certain chemicals (e. g. , benzene, some anti-neoplastic drugs) is associated with an increased risk of leukemia 30

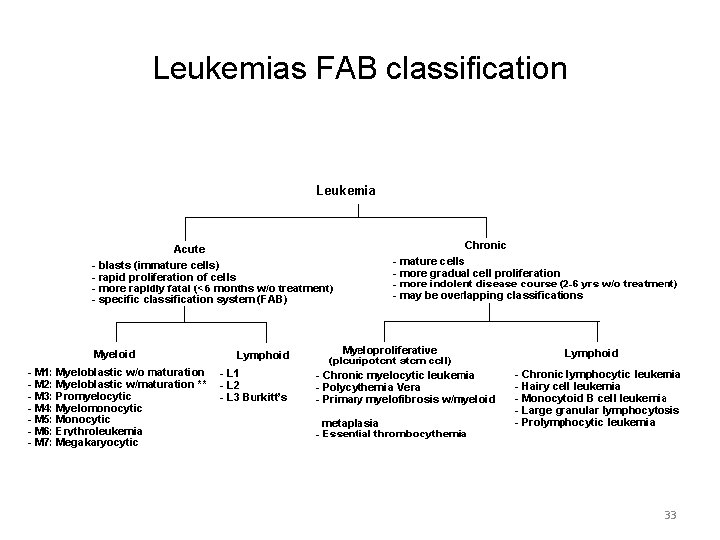
Leukemia cont’d q Some genetic defects (e. g. , Down syndrome, Fanconi's anemia) also predispose to leukemia Classification of leukemia q The main classification is into acute and chronic leukemia q On the basis of morphology and cytochemistry, acute leukemia is further subdivided into: q Acute myeloid (myeloblastic/myelogenous) leukemia (AML) q. Acute lymphoblastic (lymphocytic) leukemia (ALL) q AML is further subdivided into eight variants on a morphological basis according to the French-American. British (FAB) scheme (M 0 – M 7) 31

Classification of Leukemia cont’d q ALL is subdivided on a morphological basis according to the French-American-British (FAB) classification into L 1, L 2, and L 3 q The chronic leukemias comprise two main types: q. Chronic myeloid leukemia (CML) q. Chronic lymphocytic (lymphatic) leukemia (CLL) q Other chronic types include: q. Hairy cell leukemia q. Prolymphocytic leukemia q. Various leukemia/lymphoma syndromes 32
Leukemias FAB classification 33
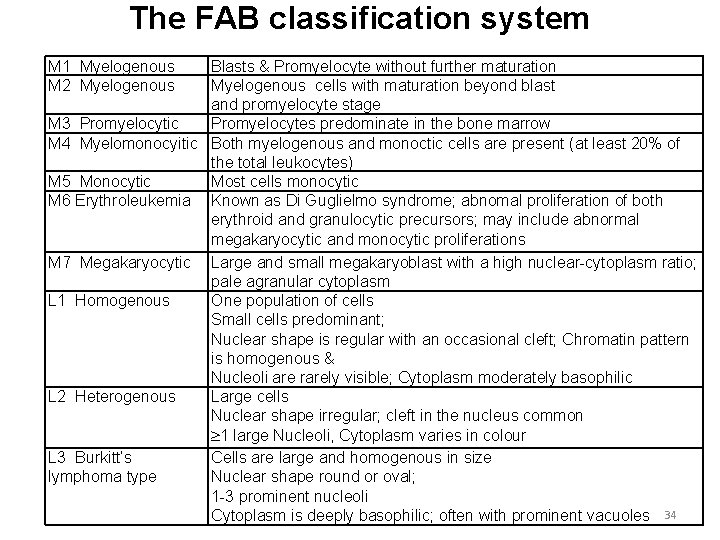
The FAB classification system M 1 Myelogenous M 2 Myelogenous Blasts & Promyelocyte without further maturation Myelogenous cells with maturation beyond blast and promyelocyte stage M 3 Promyelocytic Promyelocytes predominate in the bone marrow M 4 Myelomonocyitic Both myelogenous and monoctic cells are present (at least 20% of the total leukocytes) M 5 Monocytic Most cells monocytic M 6 Erythroleukemia Known as Di Guglielmo syndrome; abnomal proliferation of both erythroid and granulocytic precursors; may include abnormal megakaryocytic and monocytic proliferations M 7 Megakaryocytic Large and small megakaryoblast with a high nuclear-cytoplasm ratio; pale agranular cytoplasm L 1 Homogenous One population of cells Small cells predominant; Nuclear shape is regular with an occasional cleft; Chromatin pattern is homogenous & Nucleoli are rarely visible; Cytoplasm moderately basophilic L 2 Heterogenous Large cells Nuclear shape irregular; cleft in the nucleus common 1 large Nucleoli, Cytoplasm varies in colour L 3 Burkitt’s Cells are large and homogenous in size lymphoma type Nuclear shape round or oval; 1 -3 prominent nucleoli Cytoplasm is deeply basophilic; often with prominent vacuoles 34

The Acute Leukemias q The leukemic cell population in ALL and AML probably result from clonal proliferation by successive divisions form a single abnormal stem or progenitor cell q There are over 50% myeloblasts or lymphoblasts in the bone marrow at clinical presentation, and these blast cells fail to differentiate normally but are capable of further divisions q. Replacement of the normal hemopoietic precursor cells of the bone marrow by myeloblasts or lymphoblasts and, ultimately in bone marrow failure 35
The Acute Leukemias cont’d q The clinical condition of the patient can be correlated with the total number of leukemic cells in the body q. When the abnormal cell number approaches 1012 the patient is usually gravely ill with severe bone marrow failure q Peripheral blood involvement by the leukemic cells and infiltration of organs such as the spleen, liver and lymph nodes may not occur until the leukemic cell population comprised 60% or more of the marrow cell total 36

The Acute Leukemias cont’d q The disease may be recognized by conventional morphology only when blast (leukemic) cells in the marrow exceed 5% of the cell total unless the blast cells have some particular abnormal feature(e. g. Auer rods in myeloblasts and monoblasts) q. This corresponds to a total cell count in excess of 108 q The clinical presentation and mortality in acute leukemia arises mainly from: q. Neutropenia q. Thrombocytopenia, and q. Anemia because of bone marrow failure q. Organ infiltration (e. g. meninges or testes , less common) 37

The Acute Leukemias cont’d q The acute leukemias comprise over half of the leukemias seen in clinical practice q ALL is the common form in children q. Its incidence is highest at 3 -4 years q. Falls off by 10 years q. There is a lower frequency of ALL after 10 years of age with a secondary rise after the age of 40 q AML occurs in all age groups q. It is the common form of acute leukemia in adults including the elderly 38

Laboratory Features in Acute Leukemias q A normochromic normocytic anemia q The total white cell count may be decreased, normal or increased up to 200 x 109/l or more q Thrombocytopenia in most cases, often extreme in AML q Blood film examination typically shows variable numbers of blast cells q. In AML, the blasts my contain Auer rods and other abnormal cells may be present, e. g. , promyelocytes, agranular neutrophils, pseudo-Pelger cells or myelomonocytic cells q ALL must be differentiated from infectious mononucleosis and other causes of lymphocytosis 39
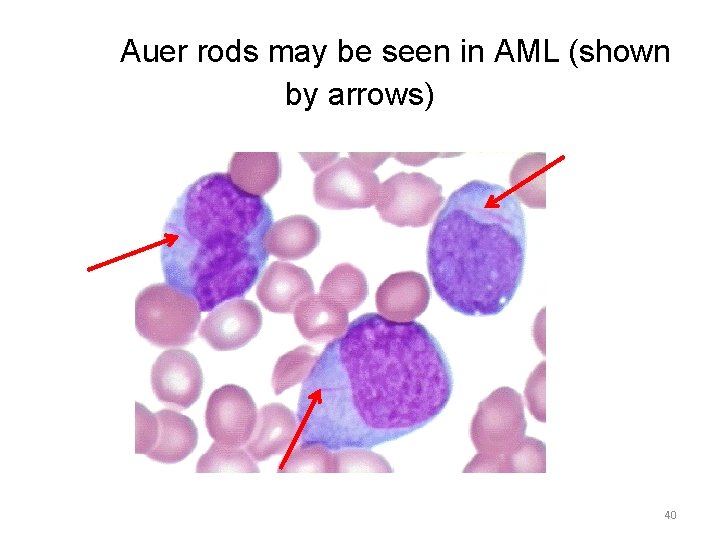
Auer rods may be seen in AML (shown by arrows) 40

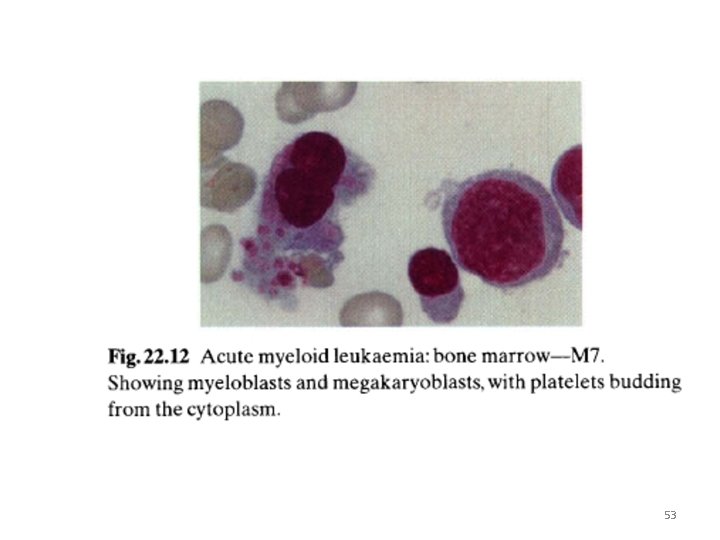
Laboratory features of Acute Myeloid Leukemias cont’d q In AML M 6 (erythroleukemia) many erythroblasts may be found and these may also be seen in smaller numbers in other forms. Megaloblastic changes may also be seen in bone marrow q The bone marrow is hypercellular with a marked proliferation of leukemic blast cells which amount to over 50% and typically over 75% of the marrow cell total q In ALL the marrow may be difficult to aspirate because of increased reticulum fiber q In AML M 7 the patient typically has an acute onset of pancytopenia with marrow fibrosis 41
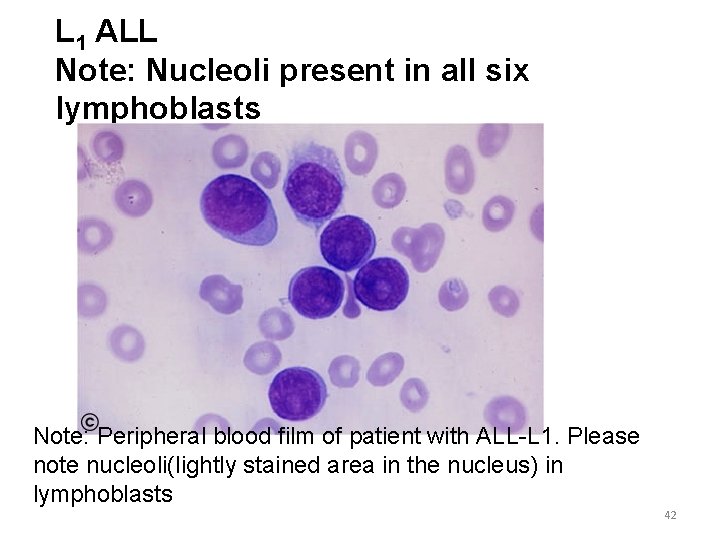
L 1 ALL Note: Nucleoli present in all six lymphoblasts Note: Peripheral blood film of patient with ALL-L 1. Please note nucleoli(lightly stained area in the nucleus) in lymphoblasts 42
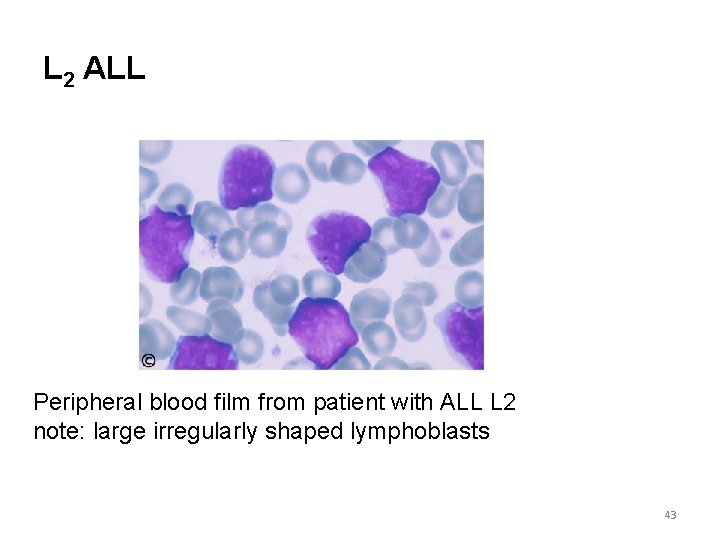
L 2 ALL Peripheral blood film from patient with ALL L 2 note: large irregularly shaped lymphoblasts 43
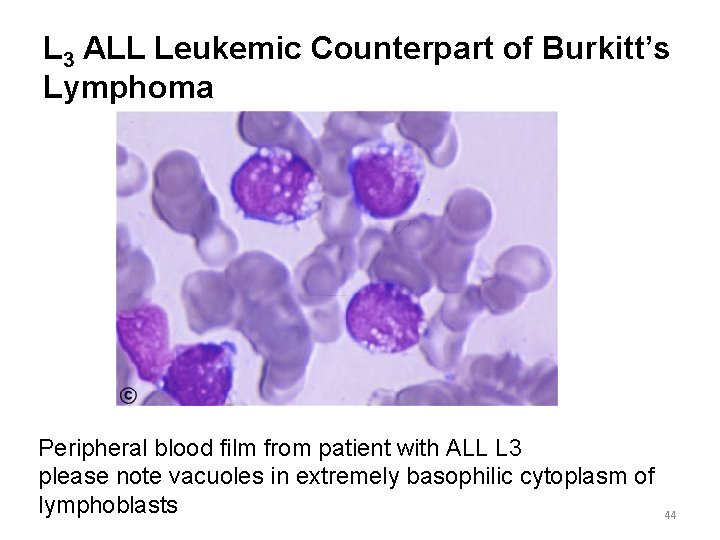
L 3 ALL Leukemic Counterpart of Burkitt’s Lymphoma Peripheral blood film from patient with ALL L 3 please note vacuoles in extremely basophilic cytoplasm of lymphoblasts 44
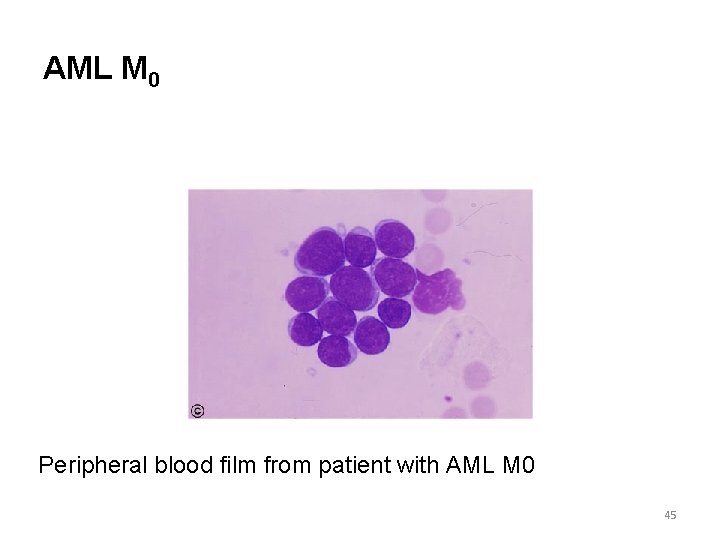
AML M 0 Peripheral blood film from patient with AML M 0 45
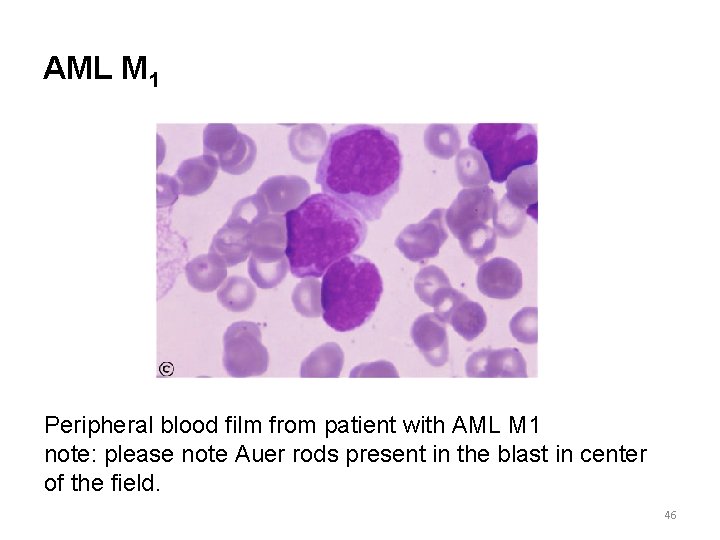
AML M 1 Peripheral blood film from patient with AML M 1 note: please note Auer rods present in the blast in center of the field. 46
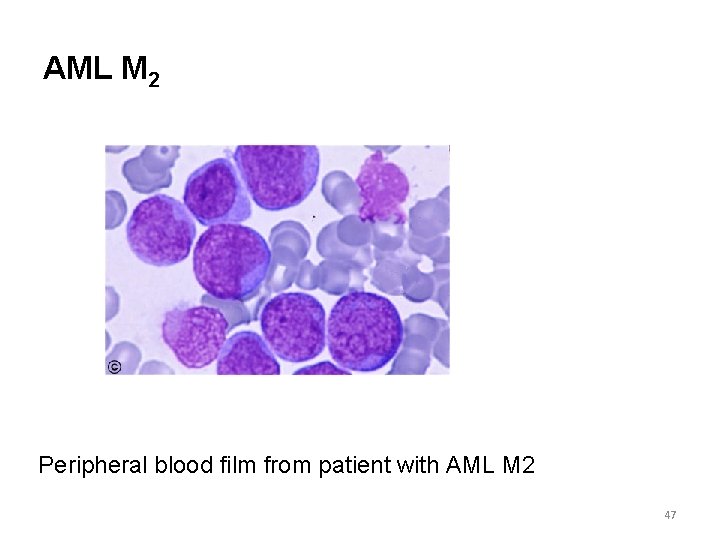
AML M 2 Peripheral blood film from patient with AML M 2 47
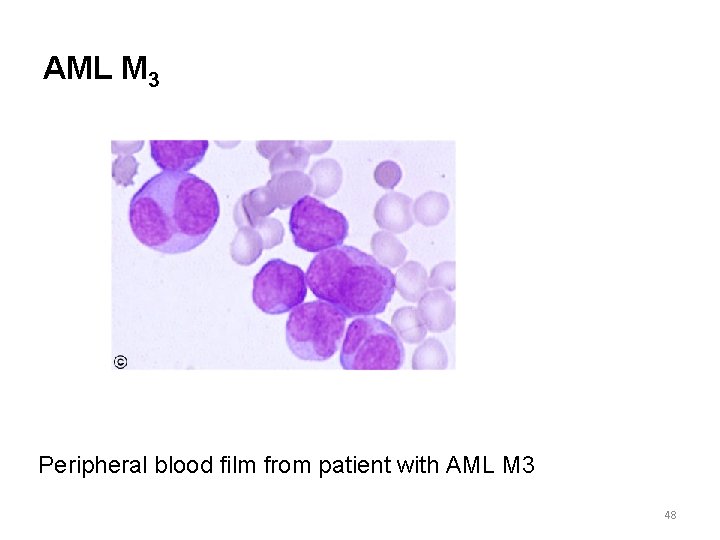
AML M 3 Peripheral blood film from patient with AML M 3 48
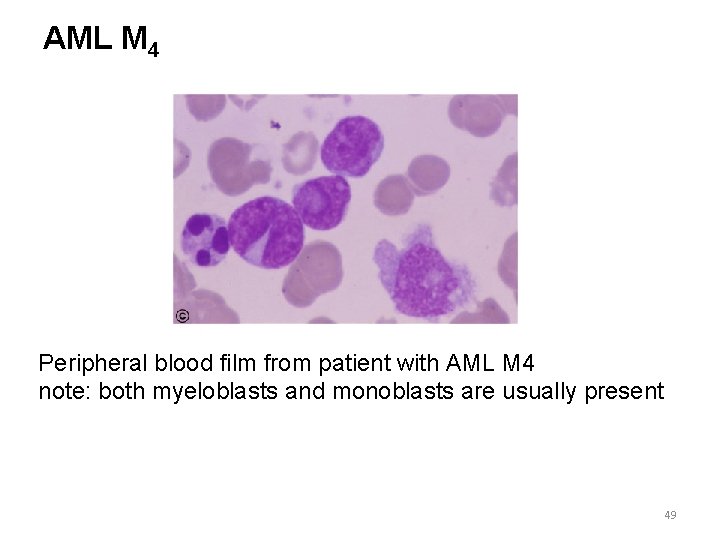
AML M 4 Peripheral blood film from patient with AML M 4 note: both myeloblasts and monoblasts are usually present 49
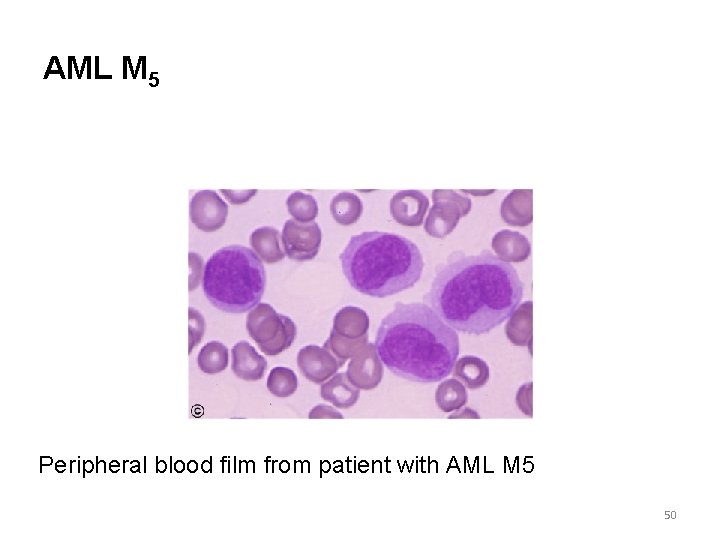
AML M 5 Peripheral blood film from patient with AML M 5 50
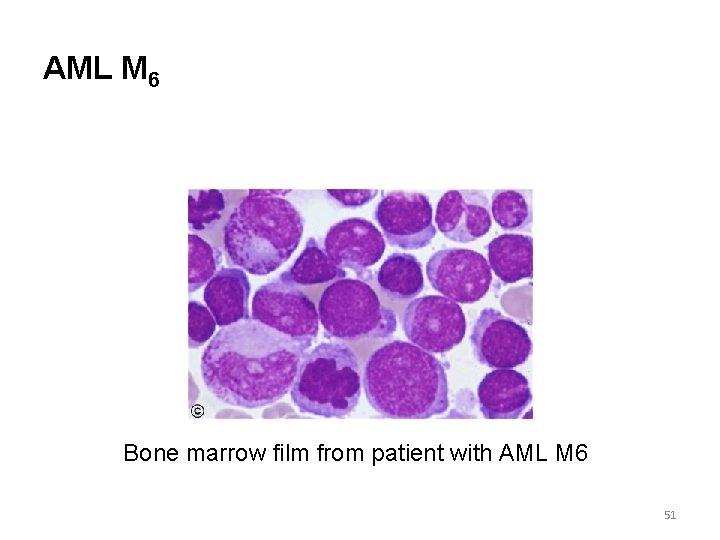
AML M 6 Bone marrow film from patient with AML M 6 51
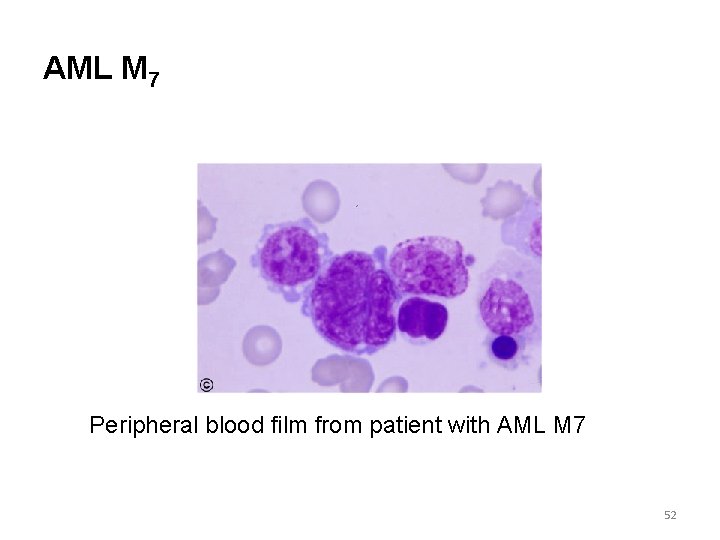
AML M 7 Peripheral blood film from patient with AML M 7 52
53
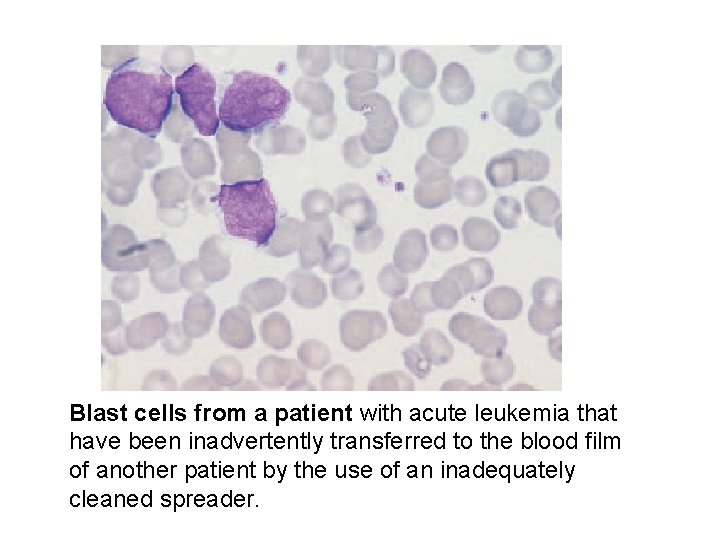
Blast cells from a patient with acute leukemia that have been inadvertently transferred to the blood film of another patient by the use of an inadequately cleaned spreader.
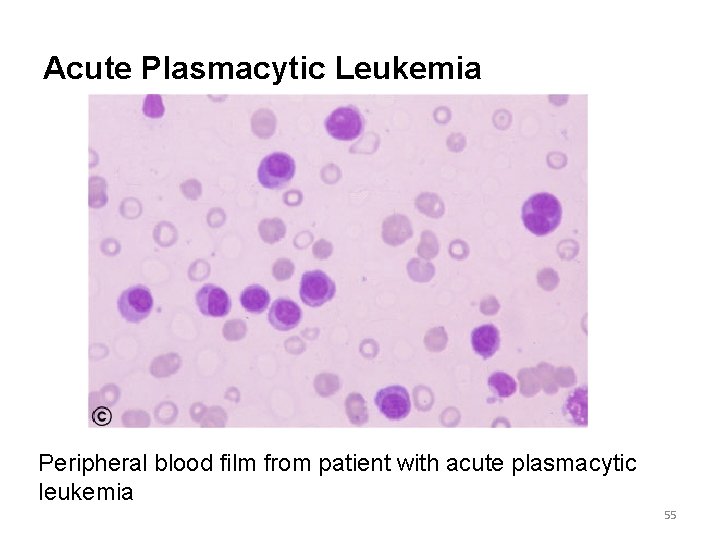
Acute Plasmacytic Leukemia Peripheral blood film from patient with acute plasmacytic leukemia 55

Differentiation of ALL from AML q In most cases, the clinical features and morphology on routine staining separate ALL from AML q. In ALL the blasts show no differentiation (with the exception of B-cell ALL) q. In AML some evidence of differentiation to granulocytes or monocytes is seen in the blasts or their progeny q Special test (e. g. , cytochemistry, gene rearrangement studies and chromosome analysis) are needed when the cells are undifferentiated to: q. Confirm the diagnosis of AML or ALL, and q. Subdivide cases of AML or ALL into their different 56 subtypes
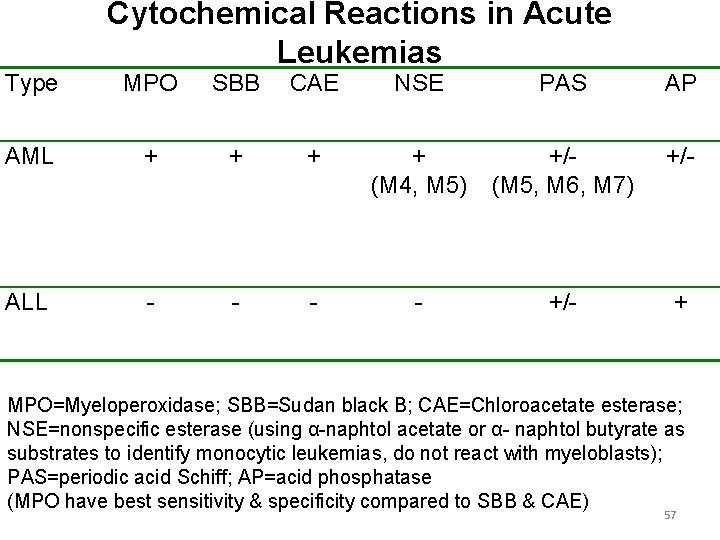
Type Cytochemical Reactions in Acute Leukemias MPO SBB CAE NSE PAS AP AML + + (M 4, M 5) +/(M 5, M 6, M 7) +/- ALL - - +/- + MPO=Myeloperoxidase; SBB=Sudan black B; CAE=Chloroacetate esterase; NSE=nonspecific esterase (using α-naphtol acetate or α- naphtol butyrate as substrates to identify monocytic leukemias, do not react with myeloblasts); PAS=periodic acid Schiff; AP=acid phosphatase (MPO have best sensitivity & specificity compared to SBB & CAE) 57
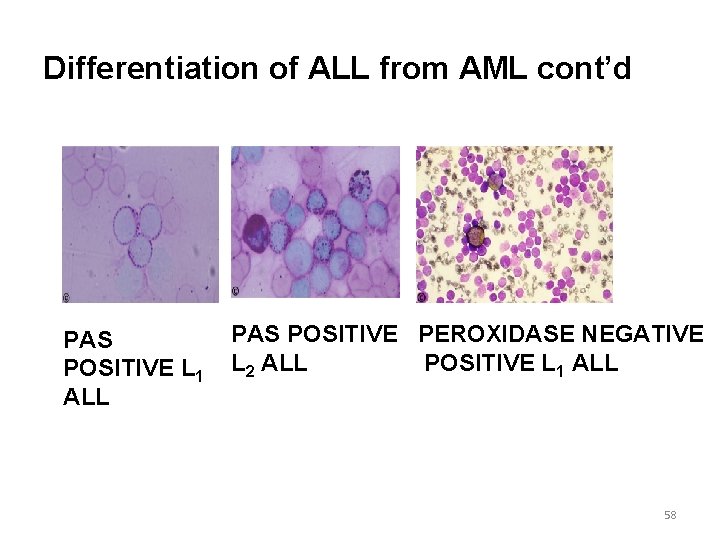
Differentiation of ALL from AML cont’d PAS POSITIVE L 1 ALL PAS POSITIVE PEROXIDASE NEGATIVE POSITIVE L 1 ALL L 2 ALL 58
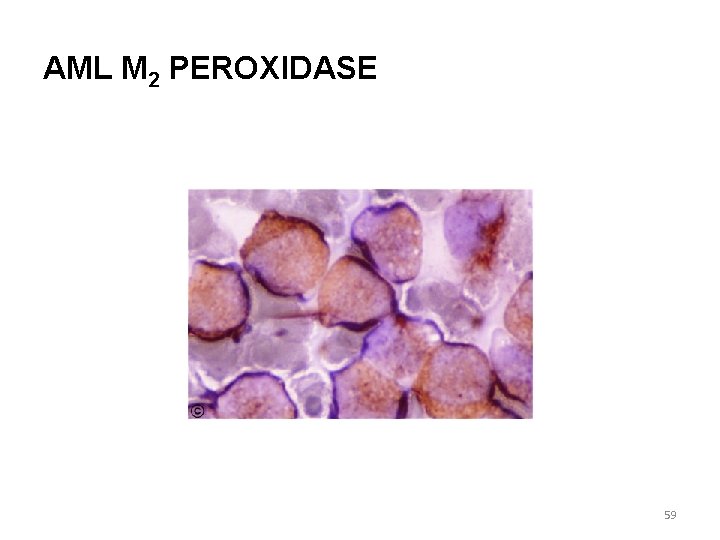
AML M 2 PEROXIDASE 59

The Chronic Leukemias Chronic Myeloid Leukemia (CML) q One of the Myeloproliferative disorders q Comprises <20% of all the leukemias and is seen most frequently in middle age q In over 95% of patients there is a replacement of normal bone marrow by cells with an abnormal chromosome- the Philadelphia or Ph chromosome q. This is an abnormal chromosome 22 due to the translocation of part of a long (q) arm of chromosome 22 to another chromosome, usually 9, with translocation of part of chromosome 9 to chromosome 22 60
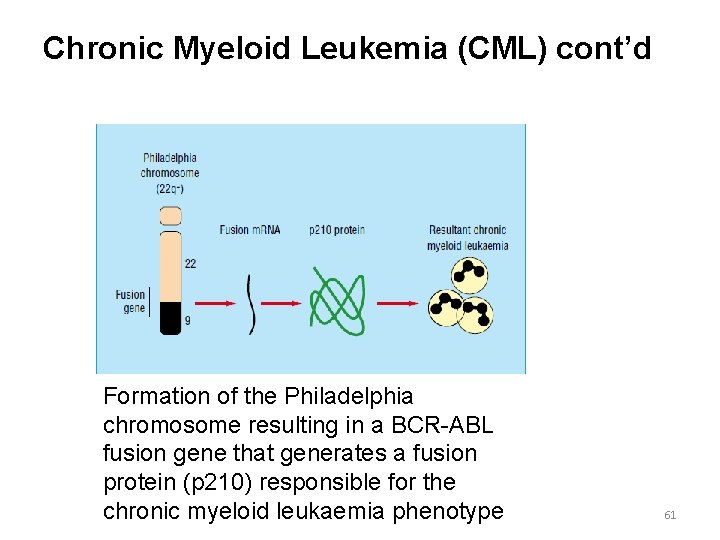
Chronic Myeloid Leukemia (CML) cont’d Formation of the Philadelphia chromosome resulting in a BCR-ABL fusion gene that generates a fusion protein (p 210) responsible for the chronic myeloid leukaemia phenotype 61

Chronic Myeloid Leukemia (CML) cont’d q It is an acquired abnormality of hemopoietic stem cells that is present in all dividing: q. Granulocytic cells q. Erythyroid cells q. Megakaryocytic cells in the marrow q. And also in some B-cell lymphocytes and probably a minority of T lymphocytes q A great increase in total body granulocyte mass is responsible for most of the clinical features q In at least 70% of patients there is a terminal metamorphosis to acute leukemia (myeloblastic or lymphoblastic) with an increase of blast cells (blast crisis) in the marrow to 50% or more 62

Chronic Myeloid Leukemia (CML) cont’d q It occurs in either gender (male: female = 1. 4: 1), most frequently between the ages of 40 and 60 years q It may occur in children and neonates and in the elderly q In most cases there are no predisposing factors but the incidence was increased in survivors of the atom bomb exposures in Japan Laboratory findings in CML q Leukocytosis is usually >50 x 109/l and sometimes >500 x 109/l q. A complete spectrum of myeloid cells is seen in the peripheral blood q. The levels of neutrophils and myelocytes exceed 63 those of blast cells and promyelocytes

Laboratory findings in CML cont’d q Philadelphia (Ph) chromosome on cytogenetic analysis of blood or bone marrow q Hypercellular bone marrow with granulopoietic predominance q Leukocyte alkaline phosphatase score is invariably low q Increased circulating basophils q Normochromic, normocytic anemia is usual q Platelet count may be increased (most frequently), normal or decreased q Serum vitamin B 12 and vitamin B 12 -binding capacity are increased q Serum uric acid is usually raised 64
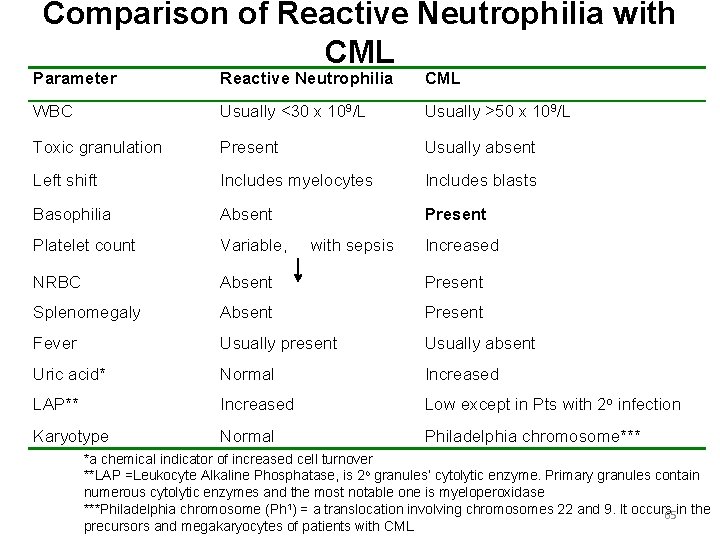
Comparison of Reactive Neutrophilia with CML Parameter Reactive Neutrophilia CML WBC Usually <30 x 109/L Usually >50 x 109/L Toxic granulation Present Usually absent Left shift Includes myelocytes Includes blasts Basophilia Absent Present Platelet count Variable, NRBC Absent Present Splenomegaly Absent Present Fever Usually present Usually absent Uric acid* Normal Increased LAP** Increased Low except in Pts with 2 o infection Karyotype Normal Philadelphia chromosome*** with sepsis Increased *a chemical indicator of increased cell turnover **LAP =Leukocyte Alkaline Phosphatase, is 2 o granules’ cytolytic enzyme. Primary granules contain numerous cytolytic enzymes and the most notable one is myeloperoxidase ***Philadelphia chromosome (Ph 1) = a translocation involving chromosomes 22 and 9. It occurs 65 in the precursors and megakaryocytes of patients with CML

Chronic lymphocytic leukemia q It accounts for 25% or more of the leukemias seen in clinical practice q It occurs in older subjects and is rare before 40 years q The male to female ratio is 2: 1 q The accumulation of large numbers of lymphocytes to 50 -100 times the normal lymphoid mass in the blood, bone marrow, spleen, lymph nodes and liver may be related to immunological non-reactivity and excessive lifespan q. The cells are a monoclonal population of B lymphocytes q With advanced CLL: q. Bone marrow failure q. A tumorous syndrome with generalized discrete lymphadenopathy 66 q. Soft tissue lymphoid masses

Chronic lymphocytic leukemia cont’d q Immunological failure results from reduced humoral and cellular immune processes with a tendency to infection Laboratory findings in CLL q Lymphocytosis q. The absolute lymphocyte count is >5 x 109/l and may be up to 300 x 109/l or more q. Between 70% and 99% of white cells in the blood film appear as small lymphocytes q. Smudge or smear cells are also present 67

Laboratory findings in CLL q Normocytic normocytic anemia is present in later states due to marrow infiltration or hypersplenism q. Autoimmune hemolysis may also occur q Thrombocytopenia occurs in many patients q Bone marrow aspiration shows lymphocytic replacement of normal marrow elements q. Lymphocytes comprise 25 -95% of all the cells q Reduced concentrations of serum immunoglobulins q. More marked with advanced disease q. Rarely a paraprotien is present 68

Hairy Cell Leukemia q Also known as leukemic reticuloendotheliosis q It is a slow growing leukemia q It is most common in older white males q It is an unusual disease of peak age 40 -60 years q Men are affected nearly four times as frequently as women q It is a type of chronic lymphoid leukemia q The disease is characterized clinically by features of Pancytopenia q The spleen may be moderately enlarged 69
Hairy Cell Leukemia cont’d q There is a monoclonal proliferation of cells with an irregular cytoplasmic outline (‘hairy’ cells, a type of B lymphocyte) in: q. The peripheral blood q. Bone marrow q. Liver and other organs q The number of hairy cells in the peripheral blood is variable q The bone marrow trephine shows a characteristic appearance of mild fibrosis and a diffuse cellular infiltrate q A serum paraprotein may be present and patients may have arthritis, serositis or vasculitis 70
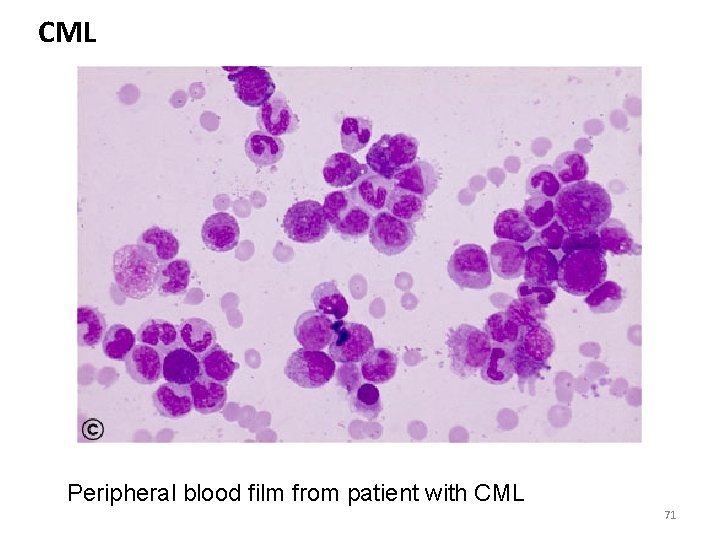
CML Peripheral blood film from patient with CML 71
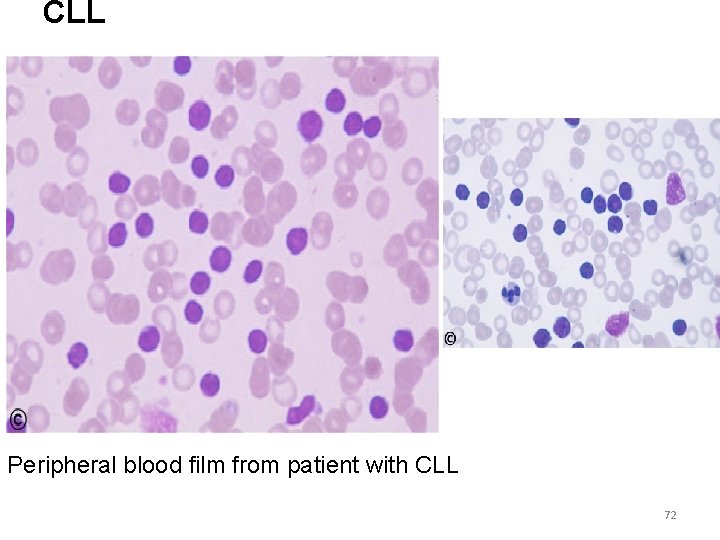
CLL Peripheral blood film from patient with CLL 72
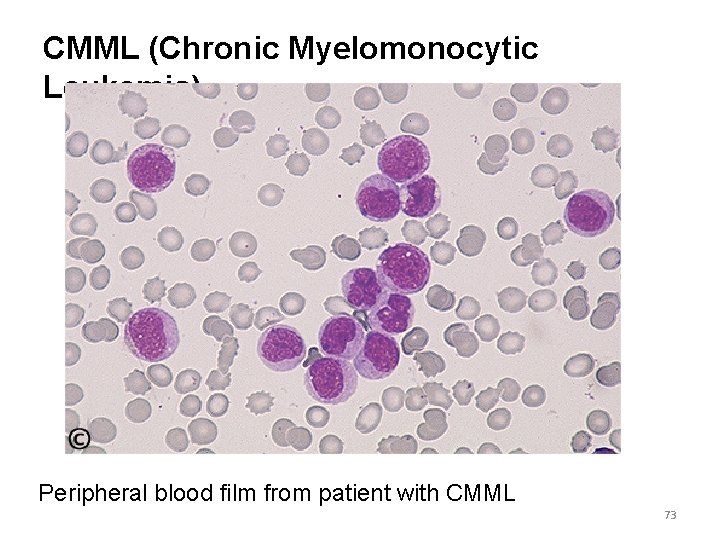
CMML (Chronic Myelomonocytic Leukemia) Peripheral blood film from patient with CMML 73
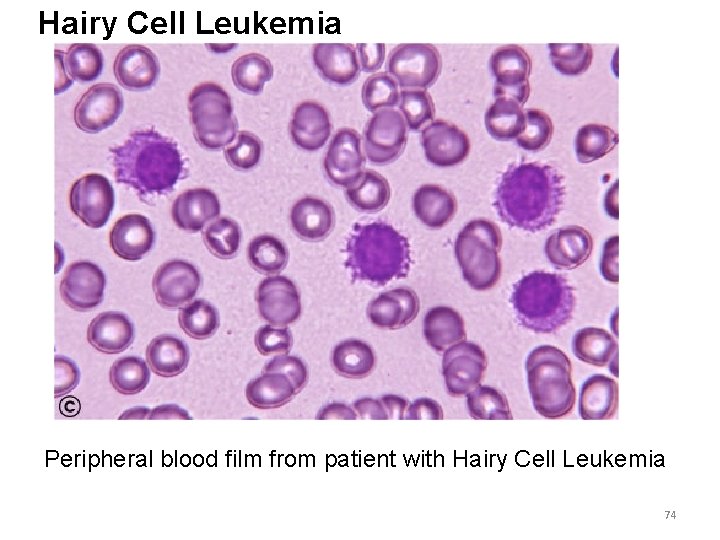
Hairy Cell Leukemia Peripheral blood film from patient with Hairy Cell Leukemia 74
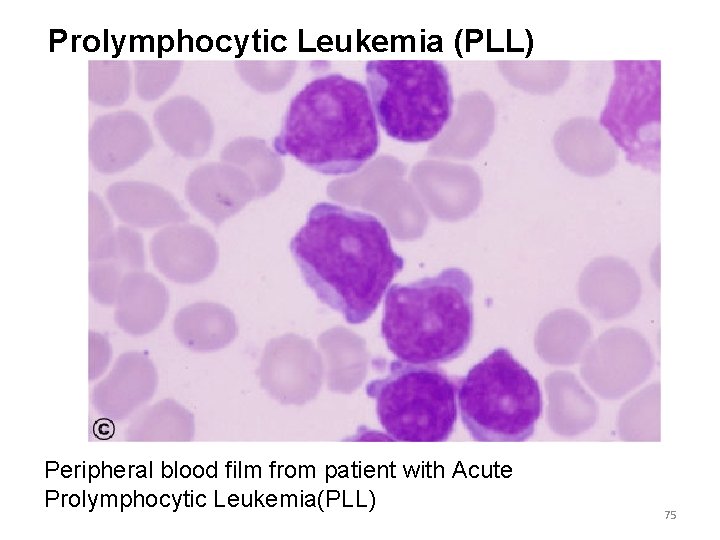
Prolymphocytic Leukemia (PLL) Peripheral blood film from patient with Acute Prolymphocytic Leukemia(PLL) 75

Myelodysplastic Syndromes (MDS)/Myelodysplasia q A heterogeneous group of disease states that usually present as peripheral blood cytopenias with a hypercellular bone marrow q Most common in the elderly and males are more commonly affected q There is a tendency to progress to acute myeloid leukemia, although death often occurs before this develops q The fundamental disorder is the clonal proliferation of stem cells that produce progeny that fail to mature normally q. The maturation defect is more subtle; mature forms develop but they are often morphologically atypical 76

Myelodysplastic Syndromes (Myelodysplasia) cont’d q The MDS are classified into five subgroups: q. Refractory anemia (RA) q. RA with ring sideroblasts (RARS) q. RA with excess blasts (RAEB) q. RAEB in transformation (RAEB-t) q. Chronic myelomonocytic leukemia (CMML) 77

WHO classification • RA with or without RS • • • only Erythroid dysplasia (5% blast) Refractory cytopenia 2 or 3 lineage dysplasia with multilineage dysplasia (<5% blast) RAEB 1 Blasts <5% RAEB 2 Blasts 10 -19% 5 q-syndrome Blasts <5% MDS- unclassified dysplasia, not meeting above criteria 78

Myelodysplastic Syndromes (Myelodysplasia) cont’d Laboratory features of MDS q Peripheral blood q. Pancytopenia is a frequent finding q. The red cells are usually macrocytic or dimorphic but occasionally hypochromic; normoblasts may be present q. The reticulocyte count is low (CRC<1% q. Granulocytes are often reduced q. Show lack of granulation q. Their chemotactic, phagocytic and adhesive functions are impaired 79

Laboratory features cont’d q The Pelger Huet-type abnormality (single or bilobed nucleus in neutrophils)) is often present q In CMML, monocytes are >1. 0 x 109/l in the blood q The total white blood count may be >100 x 109/l with left shift q The platelets may be unduly large or small and are usually decreased in number but in 10% of cases are elevated q Leukoerythroblastic blood picture q Variable numbers of myeloblasts in blood indicate poor prognosis 80

Laboratory features cont’d Bone marrow q The cellularity is usually increased q Ring sideroblasts may occur in all five FAB types q Multinucleate normoblasts and other dyserythropoietic features are seen q The granulocyte precursors q. Show defective primary and secondary granulation, and q. Cells which are difficult to identify as either agranular myelocytes, monocytes or promonocytes are frequent q Megakaryocytes are abnormal with micro, small binuclear or polynuclear forms q Bone marrow biopsy shows fibrosis in 10% of cases 81

Malignant Lymphomas q This group of diseases is divided into: q. Hodgkin’s disease q. Non-Hodgkin’s lymphomas q In both, there is replacement of normal lymphoid structure by collections of abnormal cells q. Hodgkin’s lymphoma is characterized by the presence of Reed-Sternberg (RS) cells q. The non-Hodgkin’s lymphomas are characterized by diffuse or nodular collections of abnormal lymphocytes or, rarely, histiocytes 82

Hodgkin’s Disease q It is a type of lymphoma characterized by the presence of Reed-Sternberg cells (see Figure) q In many patients, the disease is localized initially to a single peripheral lymph node region and its subsequent progression is by contiguity within the lymphatic system q RS cells and the associated abnormal and smaller mononuclear cells are neoplastic and the associated inflammatory cells represent a hypersensitivity response by the host q After a variable period of containment within the lymph nodes, the natural progression of the disease is to disseminate to involve non-lymphatic tissue 83

Hodgkin’s disease cont’d q The disease can present at any age but is rare in children q It has bimodal age incidence q. First peak in young adults (age 20 -30 years) q. A second after the age of 50 q In developed counties the ratio of young adults to child cases and of nodular sclerosing disease to other types is increased q There is an almost 2: 1 male predominance 84

Laboratory findings in Hodgkin’s disease q Normochromic, normocytic anemias is most common q One-third of patients have a leucocytosis due to a neutrophil increase q Eosinophilia is frequent q Advanced disease is associated with lymphopenia q The platelet count is normal or increased during early disease, and reduced in later stages q The ESR is usually raised and is useful in monitoring disease progress 85

Laboratory findings in Hodgkin’s disease cont’d q Bone marrow involvement is unusual in early disease q. It may be demonstrated by trephine biopsy, usually in patients with disease at many sites q There is progressive loss of immunologically competent T lymphocytes with reduced cell-mediated immune reactions q Infections are common, particularly: q. Herpes zoster q. Cytomegalovirus q. Fungal, e. g. , Cryptococcus and Candida q. Tuberculosis may occur 86

Laboratory findings in Hodgkin’s disease cont’d q Patients with bone disease may show: q. Hypercalcemia q. Hypophosphatemia q. Increased levels of serum alkaline phosphatase q Serum lactate dehydrogenase (LDH) is raised initially in 30 -40% of cases an indicates a poor prognosis q Elevated levels of serum transaminase may indicate liver involvement q Serum bilirubin may be raised due to biliary obstruction caused by large lymph nodes at the bile duct q Hyperuricemia may occur 87

Non-Hodgkin’s lymphomas q The clinical presentation and natural history of these malignant lymphomas are more variable than in Hodgkin’s disease q Pattern of spread is not regular q A greater proportion of patients present with extra nodal disease or leukemic manifestations Laboratory findings in Non-Hodgkin’s lymphoma q A normochromic normocytic anemia is usual but autoimmune hemolytic anemia may also occur q In advance disease with marrow involvement there may be neutropenia, thrombocytopenia especially if the spleen is enlarged or leuko-erythroblastic features 88
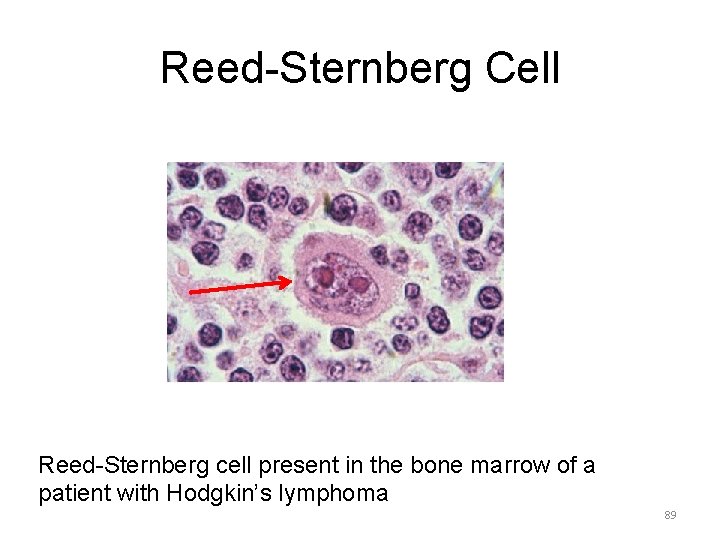
Reed-Sternberg Cell Reed-Sternberg cell present in the bone marrow of a patient with Hodgkin’s lymphoma 89

Laboratory findings in non-Hodgkin’s lymphomas cont’d q Lymphoma cells (also referred to as cleaved follicular lymphoma or blast cells) with variable nuclear abnormalities may be found in the peripheral blood in some patients q Trephine biopsy of marrow shows focal involvement, usually paratrabecular, in 20% of cases q Elevation of serum uric acid may occur q. Abnormal liver function tests suggest disseminate disease q The serum LDH level is raised in more rapidly proliferating and extensive disease and may be used as a prognostic marker 90
Multiple Myeloma q A neoplastic monoclonal proliferation of bone marrow plasma cells characterized by: q. Lytic bone lesions q. Plasma cell accumulation in the bone marrow, and q. The presence of monoclonal protein in the serum and of urine or both q 98% of cases occur over the age of 40 with a peak incidence in the seventh decade of life 91

Laboratory finding in multiple myeloma q In 98% of patients monoclonal protein occurs in the serum or urine or both q The serum paraprotein is Ig. G in two-thirds q Ig. A in one-third q Rare Ig. M or Ig. D or mixed cases q Normal serum immunoglobulins (Ig. G, Ig. A and Ig. M) are depressed q The urine contains Bence-Jones protein in two-thirds of cases q The bone marrow shows increased plasma cells often with abnormal forms referred to as myeloma cells q Immunological testing shows these cells to be monoclonal B cells q Express the same immunoglobulin heavy and light chains as the serum monoclonal protein 92

Laboratory finding in multiple myeloma cont’d q Usually a normochromic, normocytic or macrocytic anemia q Rouleaux formation is marked in most cases q Neutropenia and thrombocytopenia occur in advanced disease q Abnormal plasma cells appear in the blood film in 15% of patients q Leuko-erythroblastic changes are occasionally seen q High ESR q Serum calcium elevation occurs in 45% of patients 93

Laboratory finding in multiple myeloma cont’d q The blood urea is raised above 14 mmol/l and serum creatinine raised in 20% of cases q Proteinaceous deposits from heavy Bence-Jones proteinuria, hypercalcaemia, uric acid, amyloid and pyelonephritis may all contribute to renal failure q A low serum albumin occurs with advance disease q Serum β 2 -microglobulin (the light chain of the HLA class 1 antigens) is a useful indicator of prognosis q. It partly reflects renal function q. Levels less than 4 mg/l imply a relatively good prognosis 94

Myeloproliferative Disorders q A group of conditions characterized by clonal proliferation of one or more hemopoietic components in the bone marrow and, in many cases, the liver and spleen q. Polycythemia vera (PV) q. Essential thrombocythemia q. Myelofibrosis with myeloid metaplasia q. Chronic Myeloid Leukemia 95

Polycythemia vera (PV) q Stem cell disorder, a clonal malignancy with excessive production of mature hematopoietic cells, especially RBCs q Refers to a pattern of blood cell changes that includes: q. An increase in hemoglobin above 17. 5 g/dl in adult males and 15. 5 g/dl in females q. An accompanying rise in red cell count (above 6. 0 x 1012/l in males and 5. 5 x 1012/l in females) q. Hematocrit (above 55% in males and 47% in females) q The increase in red cell volume is caused by endogenous myeloproliferation q The stem cell origin of the defect is shown in many patients by an over-production of granulocytes and platelets as well as of red cells q This is a disease of older subjects with equal incidence in 96 males and females

Laboratory findings in PV q The hemoglobin, hematocrit and red cell count are increased q A neutrophil leukocytosis is seen in over half the patients, and some have increased circulating basophils q Increased platelet count is present in about half the patients q The leukocyte alkaline phosphatase (LAP) score is usually increased above normal q Increased serum vitamin B 12 and vitamin B 12 -binding capacity due to an increase in transcobalamin I 97

Laboratory findings in PV cont’d q The bone marrow is hypercellular with increased presence of megakaryocytes q. Best assessed by a trephine biopsy q. Clonal cytogenetic abnormalities may occur, but there is no single characteristic change q Circulating erythroid progenitors are increased and grow in vitro independently of added erythropoietin q Blood viscosity is increased q Plasma urea is often increased 98

Essential Thrombocythemia q Megakaryocyte proliferation and overproduction of platelets is the dominant feature of this condition q There is sustained increase in platelet count above normal (400 x 109/l) q Recurrent hemorrhage and thrombosis are the principal clinical features q Splenic enlargement is frequent in the early phase but splenic atrophy due to platelets blocking the splenic mirocirculation is seen in some patients q There may be anemia due to: q. Iron deficiency from chronic gastrointestinal or uterine hemorrhage q. The marrow disorder itself 99
Laboratory findings in Essential thrombocythemia q Abnormal large platelets and megakaryocyte fragments may be seen in the blood film q The bone marrow picture is similar to that of Polycythemia Vera q Platelet function tests are consistently abnormal 100

Myelofibrosis with Myeloid Metaplasia q There is the gradual replacement of the bone marrow by connective tissue q A prime feature is extramedullary hematopoieis q Patients will typically have an enlarged spleen and liver q Typically affects patients more than 50 years old Laboratory findings in myelofibrosis q Anemia is usual but a normal or increased hemoglobin level may be found in some patients q The white cell and platelet counts are frequently high at the time of presentation q. Later in the disease leukopenia and thrombocytopenia are common 101

Laboratory findings in myelofibrosis cont’d q A leuko-erythroblastic blood film is found q The red cells show characteristic tear-drop poikilocytes q Bone marrow is usually unobtainable by aspiration q Trephine biopsy may show a hypercellular marrow with an increase in reticulin-fibre pattern q Low serum and red cell folate levels, raised serum vitamin B 12 and vitamin B 12 -binding capacity, and an increased leukocyte alkaline phosphatase (LAP) score are usual q High serum uric acid, LDH and hydroxybutyrate dehydrogenase levels reflect the increased but largely ineffective turnover of hemopoietic cells q Transformation to acute myeloid leukemia occurs in 10 -20% of patients 102

Review Questions 1. Briefly describe the classification of leukemia 2. Explain the laboratory diagnosis of different types of leukemia 3. Define myelodysplastic syndrome (MDS) and indicate the hematological findings associated with MDS 4. What is the distinguishing morphological characteristic of Hodgkin’s lymphoma? 5. What are the laboratory findings of patients with multiple myeloma? 6. Define myeloproliferative disorders 103

Bibliography • MA Lichtman, E Beutler, U Seligsohn, K Kaushansky, TO Kipps (Editors). William’s Hematology. 7 th Ed. Mc. Graw. Hill Co. Inc. 2008. • Dacie, John V and Lewis, S. M. Practical Hematology 10 th Edition Churchill-Livingstone 2006. • Wintrobe, Maxwell M. Clinical Hematology 11 th Edition Lea and Febiger, Philadelphia 2003. 104
- Slides: 104