Chapter 5 Lecture Basic Chemistry Fourth Edition Chapter

Chapter 5 Lecture Basic Chemistry Fourth Edition Chapter 5 Electronic Structure and Periodic Trends 5. 1 Electromagnetic Radiation Learning Goal Compare the wavelength of radiation with its frequency. © 2014 Pearson Education, Inc.

Wave Equation The wave equation expresses the relationship of the speed of light (m/s) to wavelength (m) and frequency (s− 1). c = λν Speed of light (c) = 3. 00 × 108 m/s = wavelength (λ) × frequency (ν) = wavelength (m) × frequency (s− 1) © 2014 Pearson Education, Inc.
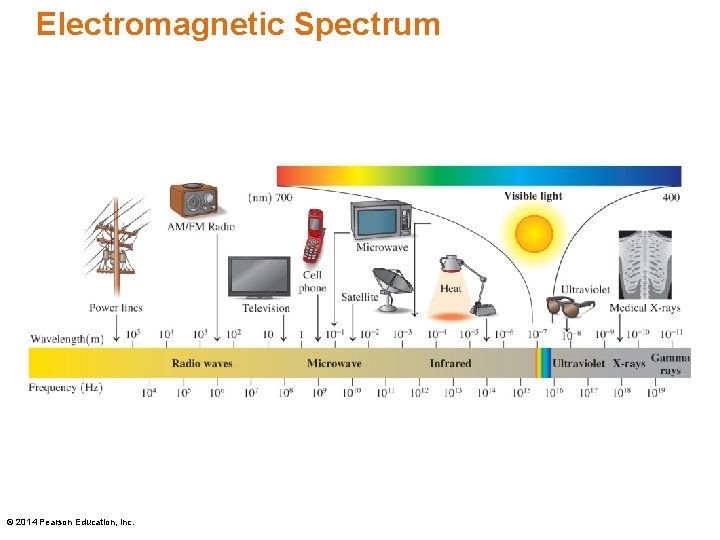
Electromagnetic Spectrum © 2014 Pearson Education, Inc.

Learning Check If the frequency of blue light is 6. 3 × 1014 Hz, what is its frequency in kilohertz (k. Hz) and in megahertz (MHz)? © 2014 Pearson Education, Inc.

Solution If the frequency of blue light is 6. 3 × 1014 Hz, what is its frequency in kilohertz (k. Hz) and in megahertz (MHz)? © 2014 Pearson Education, Inc.

Chapter 5 Lecture Basic Chemistry Fourth Edition Chapter 5 Electronic Structure and Periodic Trends 5. 3 Sublevels and Orbitals Learning Goal Describe the sublevels and orbitals in atoms. © 2014 Pearson Education, Inc.

Energy Levels Energy levels • are assigned quantum numbers n = 1, 2, 3. . . • increase in energy as the value of n increases • have a maximum number of electrons equal to 2 n 2 © 2014 Pearson Education, Inc.

Number of Sublevels Energy levels • contain one or more sublevels identified by the letters, s, p, d, and f • contain the number of sublevels equal to the principal quantum number (n) © 2014 Pearson Education, Inc.

Orbital Capacity and Spin Electrons in the same orbital have opposite spins, and therefore their magnetic fields cancel. We use arrows to represent electron spin. © 2014 Pearson Education, Inc.

Number of Electrons in Sublevels There is a maximum number of electrons that can occupy each sublevel. • An s sublevel can hold one or two electrons. • Because each p orbital can hold two electrons, the three p orbitals in a p sublevel can hold a maximum of six electrons. © 2014 Pearson Education, Inc.

Number of Electrons in Sublevels • A d sublevel with five d orbitals can hold a maximum of 10 electrons. • An f sublevel with seven f orbitals can hold a maximum of 14 electrons. © 2014 Pearson Education, Inc.

Learning Check The number of A. electrons that can occupy a p orbital is (1) 1 (2) 2(3) 3 B. p orbitals in the 2 p sublevel is (1) 1 (2) 2 (3) 3 C. d orbitals in the n = 4 energy level is (1) 1 © 2014 Pearson Education, Inc. (2) 3 (3) 5

Solution The number of A. electrons that can occupy a p orbital is (2) 2 B. p orbitals in the 2 p sublevel is (3) 3 C. d orbitals in the n = 4 energy level is (3) 5 © 2014 Pearson Education, Inc.

Chapter 5 Lecture Basic Chemistry Fourth Edition Chapter 5 Electronic Structure and Periodic Trends 5. 4 Orbital Diagrams and Electron Configurations Learning Goal Draw the orbital diagram and write the electron configuration for an element. © 2014 Pearson Education, Inc.
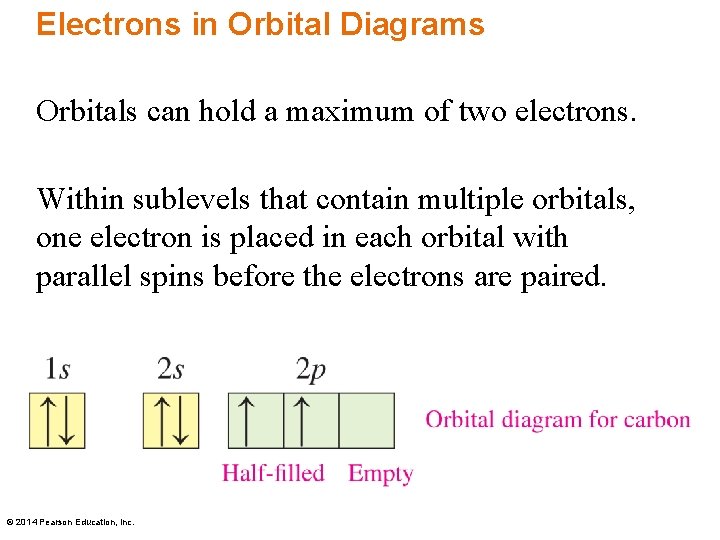
Electrons in Orbital Diagrams Orbitals can hold a maximum of two electrons. Within sublevels that contain multiple orbitals, one electron is placed in each orbital with parallel spins before the electrons are paired. © 2014 Pearson Education, Inc.

Electron Configurations Chemists use a notation called the electron configuration to indicate placement of electrons in an atom. The lowest energy sublevel is written first, then sublevels with increasing energies. The number of electrons in each sublevel is written as a superscript. © 2014 Pearson Education, Inc.
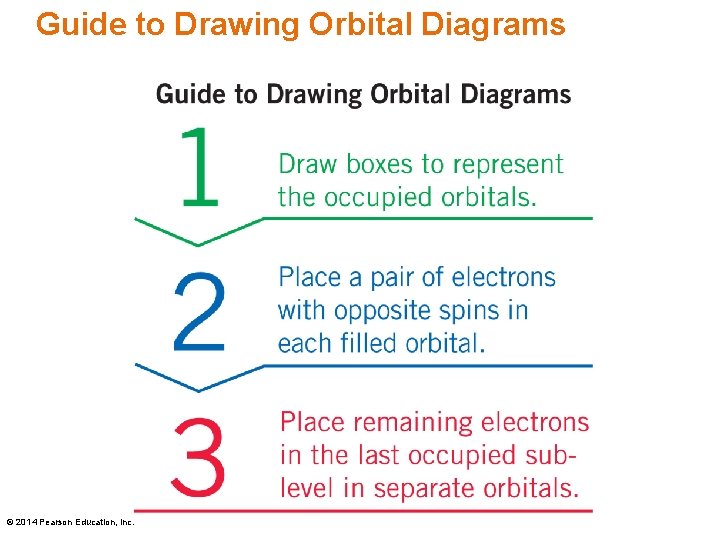
Guide to Drawing Orbital Diagrams © 2014 Pearson Education, Inc.
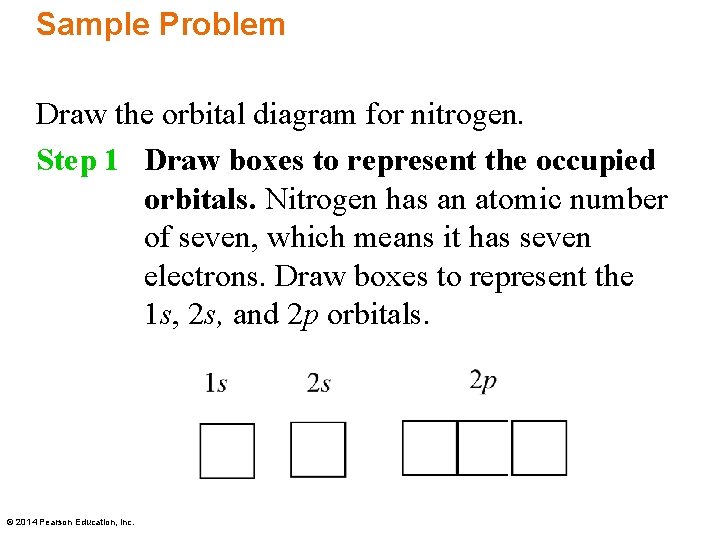
Sample Problem Draw the orbital diagram for nitrogen. Step 1 Draw boxes to represent the occupied orbitals. Nitrogen has an atomic number of seven, which means it has seven electrons. Draw boxes to represent the 1 s, 2 s, and 2 p orbitals. © 2014 Pearson Education, Inc.
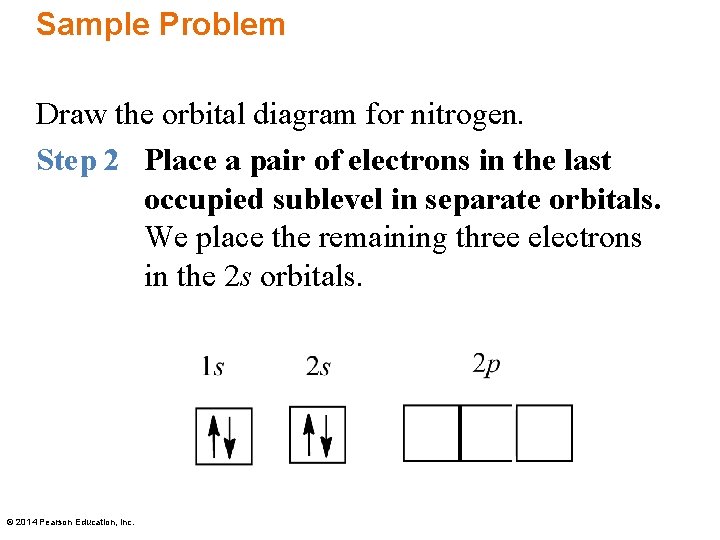
Sample Problem Draw the orbital diagram for nitrogen. Step 2 Place a pair of electrons in the last occupied sublevel in separate orbitals. We place the remaining three electrons in the 2 s orbitals. © 2014 Pearson Education, Inc.

Sample Problem Draw the orbital diagram for nitrogen. Step 3 Place remaining electrons with opposite spins in each filled orbital. First we place a pair of electrons with opposite spins in the 2 p orbitals, with arrows in the same direction. © 2014 Pearson Education, Inc.
![Learning Check Draw the abbreviated orbital diagram for aluminum, [Ne] 3 s 2 3 Learning Check Draw the abbreviated orbital diagram for aluminum, [Ne] 3 s 2 3](http://slidetodoc.com/presentation_image_h2/4e5a8780e105076ccaf6a34602c87900/image-21.jpg)
Learning Check Draw the abbreviated orbital diagram for aluminum, [Ne] 3 s 2 3 p 1. © 2014 Pearson Education, Inc.
![Solution Draw the abbreviated orbital diagram for aluminum, [Ne] 3 s 2 3 p Solution Draw the abbreviated orbital diagram for aluminum, [Ne] 3 s 2 3 p](http://slidetodoc.com/presentation_image_h2/4e5a8780e105076ccaf6a34602c87900/image-22.jpg)
Solution Draw the abbreviated orbital diagram for aluminum, [Ne] 3 s 2 3 p 1. 1. The preceding noble gas is Ne; we use this to represent 1 s 2 2 p 6. 2. Fill the 3 s, and add the last electron to the 3 p sublevel. [Ne] © 2014 Pearson Education, Inc.

Chapter 5 Lecture Basic Chemistry Fourth Edition Chapter 5 Electronic Structure and Periodic Trends 5. 5 Electron Configurations and the Periodic Table Learning Goal Write the electron configuration for an atom using the sublevel blocks on the periodic table. © 2014 Pearson Education, Inc.
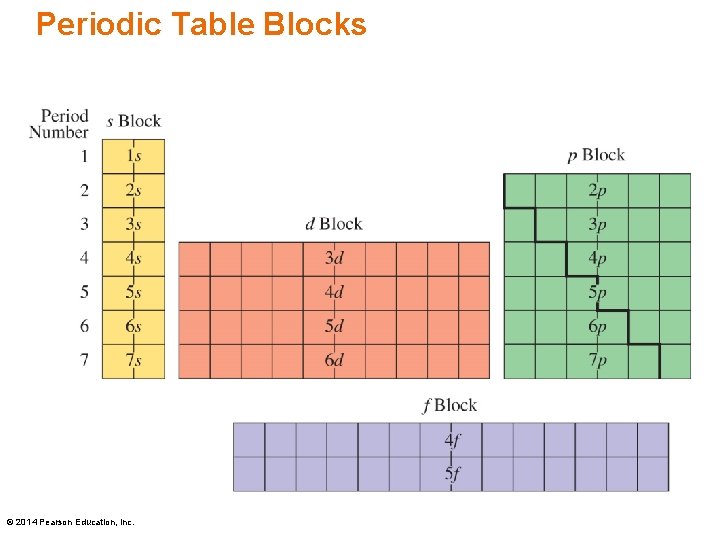
Periodic Table Blocks © 2014 Pearson Education, Inc.

Guide to Writing Electron Configurations © 2014 Pearson Education, Inc.

Sample Problem Use the sublevel blocks on the periodic table to write the electron configuration for chlorine. Step 1 Locate the element on the periodic table. Chlorine (atomic number 17) is in Group 7 A (17) and Period 3. © 2014 Pearson Education, Inc.

Sample Problem Use the sublevel blocks on the periodic table to write the electron configuration for chlorine. Step 2 Write the filled sublevels in order, going across each period. © 2014 Pearson Education, Inc.

Sample Problem Use the sublevel blocks on the periodic table to write the electron configuration for chlorine. Step 3 Complete the configuration by counting the electrons in the last occupied sublevel block. The electron configuration for chlorine (Cl) is 1 s 2 2 p 6 3 s 2 3 p 5 © 2014 Pearson Education, Inc.

Learning Check Use the periodic table to give the symbol of the element with each of the following electron configurations. A. 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 7 B. [Ar] 4 s 2 3 d 10 4 p 5 © 2014 Pearson Education, Inc.

Solution Use the periodic table to give the symbol of the element with each of the following electron configurations. A. 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 7 Element is cobalt (Co). B. [Ar] 4 s 2 3 d 10 4 p 5 Element is bromine (Br). © 2014 Pearson Education, Inc.

Chapter 5 Lecture Basic Chemistry Fourth Edition Chapter 5 Electronic Structure and Periodic Trends 5. 6 Trends in Periodic Properties Learning Goal Use the electron configurations of elements to explain the trends in periodic properties. © 2014 Pearson Education, Inc.

Valence Electrons The valence electrons, the s and p sublevels in the highest energy level, • are related to the group number of the element • determine the chemical properties of an element Example: Phosphorus has five valence electrons. P Group 5 A(15) © 2014 Pearson Education, Inc. 1 s 2 2 p 6 3 s 2 3 p 3
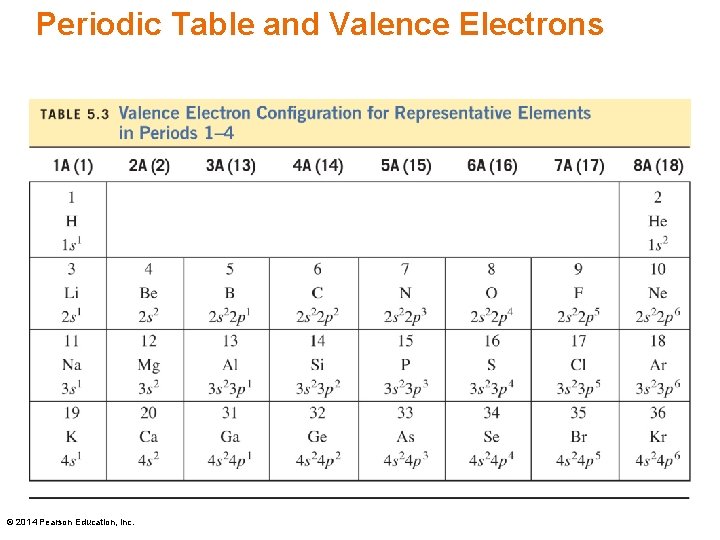
Periodic Table and Valence Electrons © 2014 Pearson Education, Inc.

Learning Check State the number of valence electrons for each: A. O (1) 4 (2) 6 (3) 8 B. Al (1) 13 (2) 3 (3) 1 C. Cl (1) 2 (2) 5 (3) 7 D. Group 6 A (16) (1) 2 (2) 4 E. Tin (1) 2 (2) 4 © 2014 Pearson Education, Inc. (3) 6 (3) 14

Solution State the number of valence electrons for each: A. O (2) 6 B. Al (2) 3 C. Cl (3) 7 D. Group 6 A (16) E. Tin © 2014 Pearson Education, Inc. (3) 6 (2) 4

Electron-Dot Symbols An electron-dot symbol • indicates the number of valence electrons as dots around the symbol of the element • for Mg shows two valence electrons placed as single dots on the sides of the symbol Mg Mg 1 s 2 2 p 6 3 s 2 Electron-Dot Symbols for Magnesium © 2014 Pearson Education, Inc.
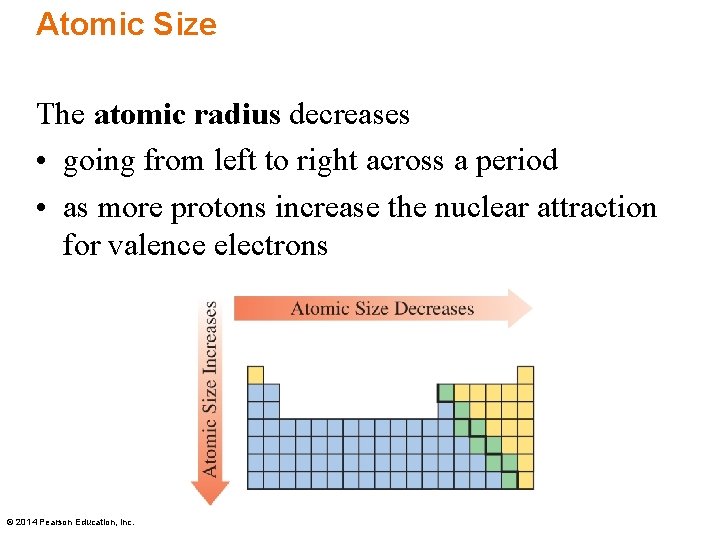
Atomic Size The atomic radius decreases • going from left to right across a period • as more protons increase the nuclear attraction for valence electrons © 2014 Pearson Education, Inc.

Ionization Energy Metals have • 1 -3 valence electrons • lower ionization energies Nonmetals have • 5 -7 valence electrons • higher ionization energies Noble gases have • complete octets (He has two valence electrons. ) • highest ionization energies in a period © 2014 Pearson Education, Inc.

Metallic Character Elements with high metallic character lose their electrons easily. Elements that are • metals lose their electrons easily • metalloids lose electrons, but not as easily as metals • nonmetals do not easily lose their electrons © 2014 Pearson Education, Inc.

Summary, Periodic Table Trends © 2014 Pearson Education, Inc.
- Slides: 40