Chapter 5 Gases and Gas Laws Gases indefinite
















- Slides: 20

Chapter 5 - Gases and Gas Laws Gases indefinite volume and shape, low density. Liquids definite volume, indefinite shape, and high density. Solids definite volume and shape, high density Solids and liquids have high densities because their molecules are close together.

Kinetic Theory u. Kinetic theory says that molecules are in constant motion. u. Perfume molecules moving across the room are evidence of this.

The Kinetic Theory of Gases Makes three assumptions about gases Ê A Gas is composed of particles Husually molecules or atoms HConsidered to be hard spheres far enough apart that we can ignore their volume. HBetween the molecules is empty space.

Ë The particles are in constant random motion. H Move in straight lines until they bounce off each other or the walls. Ì All collisions are perfectly elastic Ì http: //www. chemistry. ohiostate. edu/betha/neal. Gas. Law/fr 2. 2. html

The Average speed of an oxygen molecule is 1656 km/hr at 20ºC The molecules don’t travel very far without hitting each other so they move in random directions.

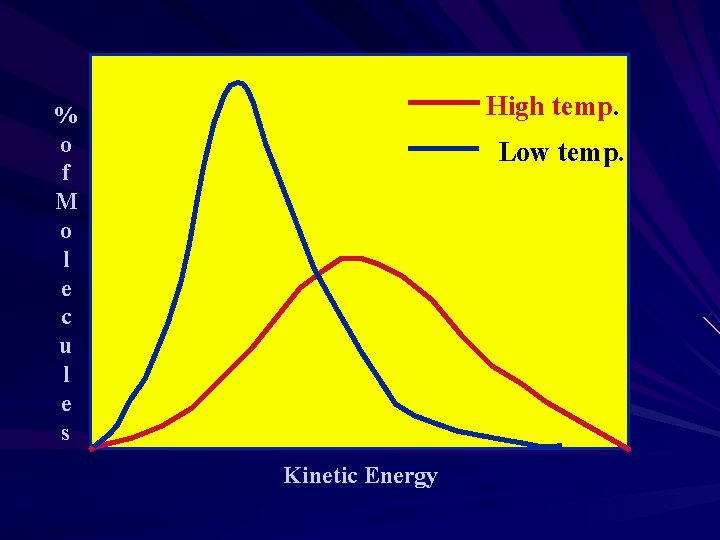
Kinetic Energy and Temperature is a measure of the Average kinetic energy of the molecules of a substance. Higher temperature faster molecules. At absolute zero (0 K) all molecular motion would stop. http: //intro. chem. okstate. edu/1314 F 00/Lab oratory/GLP. htm http: //www 2. biglobe. ne. jp/~norimari/scienc e/Java. App/Mole/e-gas. html

High temp. % o f M o l e c u l e s Low temp. Kinetic Energy

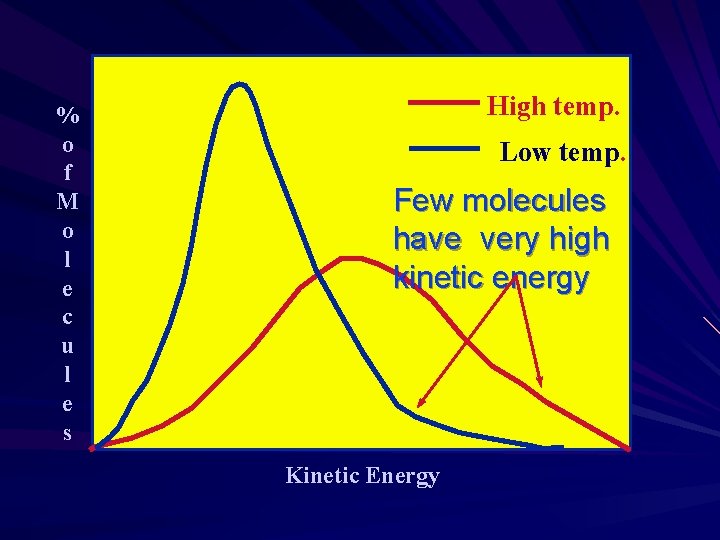
% o f M o l e c u l e s High temp. Low temp. Few molecules have very high kinetic energy Kinetic Energy

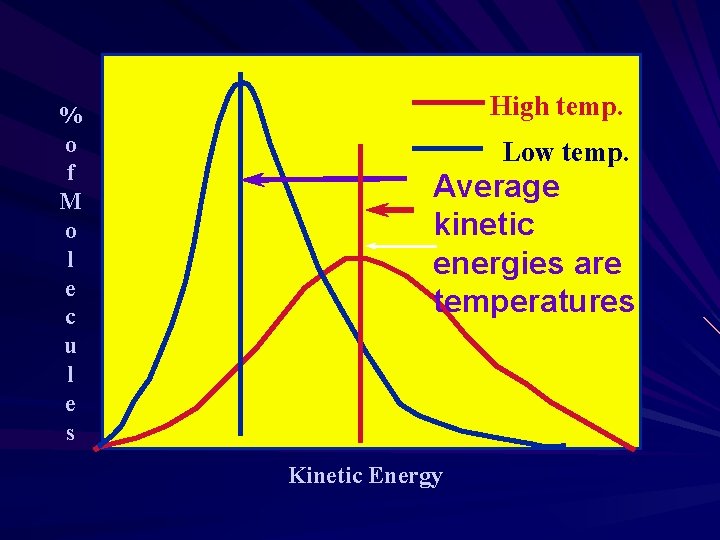
% o f M o l e c u l e s High temp. Low temp. Average kinetic energies are temperatures Kinetic Energy

Temperature The average kinetic energy is directly proportional to the temperature in Kelvin If you double the temperature (in Kelvin) you double the average kinetic energy. If you change the temperature from 300 K to 600 K the kinetic energy doubles.

Temperature If you change the temperature from 300ºC to 600ºC the Kinetic energy doesn’t double. 873 K is not twice 573 K

Pressure is the result of collisions of the molecules with the sides of a container. A vacuum is completely empty space it has no pressure. Pressure is measured in units of atmospheres (atm). It is measured with a device called a barometer.

Graham’s Law of Diffusion http: //chem. salve. edu/chemistry/diffusion. a sp

Real Gases Real molecules do take up space and they do interact with each other (especially polar molecules). Need to add correction factors to the ideal gas law to account for these.

Volume Correction The actual volume free to move in is less because of particle size. More molecules will have more effect. Corrected volume V’ = V - nb b is a constant that differs for each gas. P’ = n. RT (V-nb)

Pressure correction Because the molecules are attracted to each other, the pressure on the container will be less than ideal depends on the number of molecules per liter. since two molecules interact, the effect must be squared.

Attractive Forces in Real Gases

Pressure correction Because the molecules are attracted to each other, the pressure on the container will be less than ideal n depends on the number of molecules per liter. n since two molecules interact, the effect must be squared. 2 n Pobserved = P’ - a () n V

Altogether () Pobs= n. RT - a n 2 V-nb V Called the Van der Wall’s equation if rearranged Corrected Pressure Corrected Volume

Where does it come from a and b are determined by experiment. Different for each gas. Bigger molecules have larger b. a depends on both size and polarity. once given, plug and chug.