Chapter 5 Fluids and Electrolytes Acids and Bases

Chapter 5 Fluids and Electrolytes, Acids and Bases Copyright © 2017, Elsevier Inc. All rights reserved.

Distribution of Body Fluids Total body water (TBW) Intracellular fluid Ø Extracellular fluid • Interstitial fluid • Intravascular fluid • Lymph, synovial, intestinal, CSF, sweat, urine, pleural, Ø peritoneal, pericardial, and intraocular fluids Copyright © 2017, Elsevier Inc. All rights reserved. 2

Distribution of Body Fluids (Cont. ) Pediatrics 75% to 80% of body weight Ø Susceptible to significant changes in body fluids • Dehydration in newborns Ø Aging Ø Decreased percent of total body water • Decreased free fat mass and decreased muscle mass • Renal decline • Diminished thirst perception Copyright © 2017, Elsevier Inc. All rights reserved. 3

Water Movement Between Fluid Compartments Osmolality Osmotic forces Aquaporins Starling forces Ø Net filtration = forces favoring filtration minus forces opposing filtration Copyright © 2017, Elsevier Inc. All rights reserved. 4
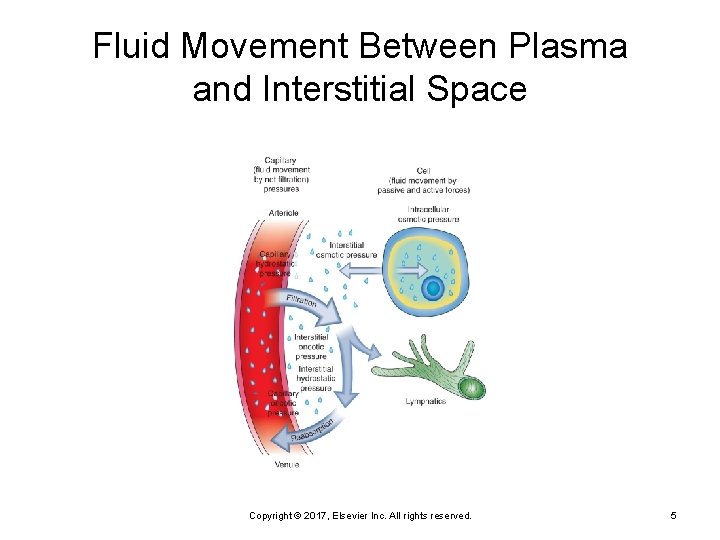
Fluid Movement Between Plasma and Interstitial Space Copyright © 2017, Elsevier Inc. All rights reserved. 5

Net Filtration Forces favoring filtration: Capillary hydrostatic pressure (blood pressure) Ø Interstitial oncotic pressure (water-pulling) Ø Forces favoring reabsorption: Plasma (capillary) oncotic pressure (water-pulling) Ø Interstitial hydrostatic pressure Ø Copyright © 2017, Elsevier Inc. All rights reserved. 6

Edema Accumulation of fluid within the interstitial spaces Causes: Increase in capillary hydrostatic pressure Ø Decrease in plasma oncotic pressure Ø Increase in capillary permeability Ø Lymph obstruction (lymphedema) Ø Localized vs. generalized Pitting edema Copyright © 2017, Elsevier Inc. All rights reserved. 7
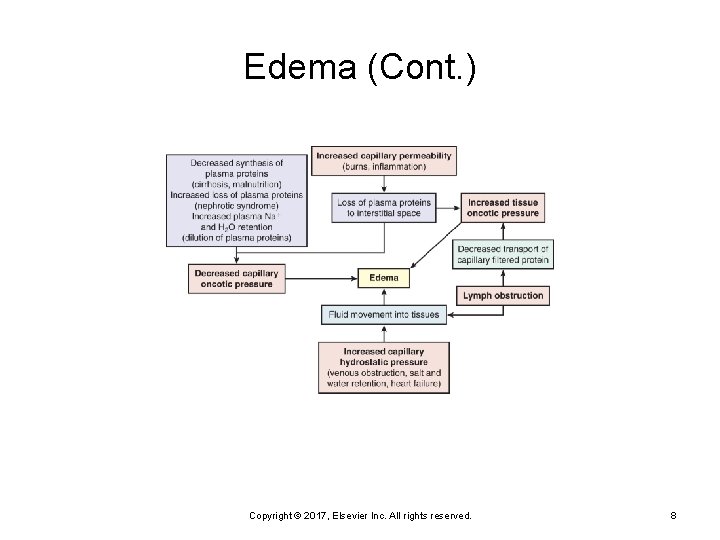
Edema (Cont. ) Copyright © 2017, Elsevier Inc. All rights reserved. 8

Sodium and Chloride Balance Sodium Primary ECF cation Ø Regulates osmotic forces, thus water Ø Roles • Neuromuscular irritability, acid-base balance, and cellular Ø chemical reactions and membrane transport Chloride Primary ECF anion Ø Provides electroneutrality Ø Copyright © 2017, Elsevier Inc. All rights reserved. 9

Sodium and Chloride Balance (Cont. ) Renin-angiotensin-aldosterone system Aldosterone—leads to sodium and water reabsorption back into the circulation and excretion of potassium Ø Natriuretic peptides Ø Causes sodium and water excretion Ø Copyright © 2017, Elsevier Inc. All rights reserved. 10

Water Balance ADH secretion Ø Increases water reabsorption into the plasma Thirst perception Osmolality receptors • Hyperosmolality and plasma volume depletion Ø Volume receptors Ø Baroreceptors Ø Copyright © 2017, Elsevier Inc. All rights reserved. 11
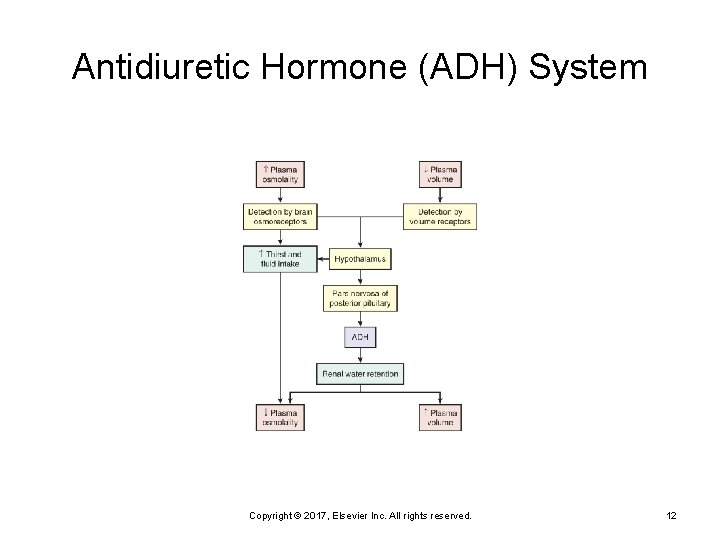
Antidiuretic Hormone (ADH) System Copyright © 2017, Elsevier Inc. All rights reserved. 12

Alterations in Na+, Cl , and Water Balance Isotonic alterations Total body water change with proportional electrolyte and water change (no change in concentration) Ø Isotonic fluid loss Ø Isotonic fluid excess Ø Copyright © 2017, Elsevier Inc. All rights reserved. 13

Alterations in Na+, Cl–, and Water Balance (Cont. ) Hypertonic alterations Ø Hypernatremia • Serum sodium >145 m. Eq/L • Related to sodium gain or water loss • Water movement from the ICF to the ECF Intracellular dehydration • Manifestations Clinical – Thirst, weight gain, bounding pulse, and increased blood pressure Central nervous system – Muscle twitching and hyperreflexia (hyperactive reflexes), confusion, coma, convulsions, and cerebral hemorrhage Copyright © 2017, Elsevier Inc. All rights reserved. 14

Alterations in Na+, Cl–, and Water Balance (Cont. ) Hypertonic alterations (Cont. ) Ø Hyperchloremia • Occurs with hypernatremia or a bicarbonate deficit Copyright © 2017, Elsevier Inc. All rights reserved. 15

Alterations in Na+, Cl–, and Water Balance (Cont. ) Hypotonic alterations Decreased osmolality Ø Hyponatremia or free water excess • Hyponatremia decreases the ECF osmotic pressure, and Ø water moves into the cell via osmosis • Cells expand Copyright © 2017, Elsevier Inc. All rights reserved. 16

Hyponatremia Serum sodium level <135 m. Eq/L Sodium deficits cause plasma hypoosmolality and cellular swelling Causes: Pure sodium loss Ø Low intake Ø Dilutional hyponatremia Ø Copyright © 2017, Elsevier Inc. All rights reserved. 17

Hyponatremia (Cont. ) Manifestations: Most life-threatening: cerebral edema and increased intracranial pressure Ø Lethargy, confusion, decreased reflexes, seizures, and coma Ø If leads to loss of ECF and hypovolemia, see hypotension, tachycardia, decreased urine output Ø If dilutional from excess water (hypervolemic hyponatremia), see weight gain, edema, ascites, jugular vein distention Ø Copyright © 2017, Elsevier Inc. All rights reserved. 18

Water Excess Compulsive water drinking Decreased urine formation Syndrome of inappropriate ADH (SIADH) ADH secretion in the absence of hypovolemia or hyperosmolality Ø Hyponatremia with hypervolemia Ø Copyright © 2017, Elsevier Inc. All rights reserved. 19

Water Excess (Cont. ) Manifestations: cerebral edema (with confusion and convulsions), weakness, muscle twitching, nausea, headache, and weight gain Copyright © 2017, Elsevier Inc. All rights reserved. 20

Hypochloremia Usually the result of hyponatremia or elevated bicarbonate concentration Develops as a result of vomiting and the loss of HCl Occurs in cystic fibrosis Treatment of underlying cause is required Copyright © 2017, Elsevier Inc. All rights reserved. 21

Potassium Major intracellular cation Concentration maintained by Na+/K+ pump Regulates intracellular electrical neutrality in relation to Na+ and H+ Essential for transmission and conduction of nerve impulses, normal cardiac rhythms, and skeletal and smooth muscle contraction Copyright © 2017, Elsevier Inc. All rights reserved. 22

Potassium Levels Changes in p. H affect K+ balance Ø Hydrogen ions accumulate in the ICF during states of acidosis; K+ shifts out to maintain a balance of cations across the membrane; result is hyperkalemia Aldosterone, insulin, and epinephrine influence serum potassium levels Kidney is most efficient regulator Potassium adaptation Ø Slow changes tolerated better than acute Copyright © 2017, Elsevier Inc. All rights reserved. 23

Hypokalemia Potassium level <3. 5 m. Eq/L Potassium balance is described by changes in plasma potassium levels Causes: reduced intake of potassium, increased entry of potassium into cells, and increased loss of potassium Copyright © 2017, Elsevier Inc. All rights reserved. 24

Hypokalemia (Cont. ) Manifestations (depend on rate and severity) Ø Membrane hyperpolarization causes a decrease in neuromuscular excitability, skeletal muscle weakness, smooth muscle atony, and cardiac dysrhythmias Copyright © 2017, Elsevier Inc. All rights reserved. 25

Hyperkalemia Potassium level >5. 5 m. Eq/L Hyperkalemia is rare because of efficient renal excretion Caused by increased intake, shift of K+ from ICF into ECF, decreased renal excretion, insulin deficiency, or cell trauma Copyright © 2017, Elsevier Inc. All rights reserved. 26

Hyperkalemia (Cont. ) Mild attacks Ø Increased neuromuscular irritability • Restlessness, intestinal cramping, and diarrhea Severe attacks Ø Decreases the resting membrane potential • Muscle weakness, loss of muscle tone, and paralysis Copyright © 2017, Elsevier Inc. All rights reserved. 27
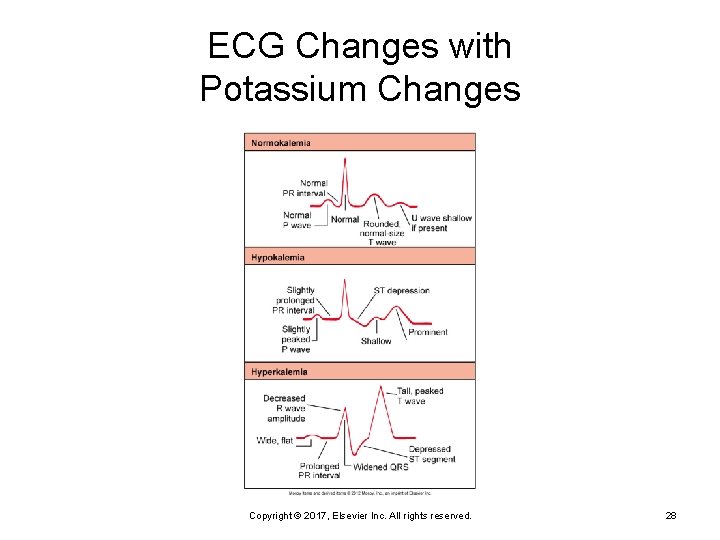
ECG Changes with Potassium Changes Copyright © 2017, Elsevier Inc. All rights reserved. 28

Calcium and Phosphate Calcium and phosphate concentrations are rigidly controlled by parathyroid hormone (PTH), vitamin D, and calcitonin Copyright © 2017, Elsevier Inc. All rights reserved. 29

Calcium 99% of calcium is located in the bone as hydroxyapatite Necessary for structure of bones and teeth, blood clotting, hormone secretion, cell receptor function, plasma membrane stability, transmission of nerve impulses, muscle contraction Serum concentration 8. 8 to 10. 5 mg/dl Copyright © 2017, Elsevier Inc. All rights reserved. 30

Hypocalcemia Causes: Inadequate intestinal absorption, deposition of ionized calcium into bone or soft tissue, blood administration Ø Decreases in PTH and vitamin D Ø Nutritional deficiencies occur with inadequate sources of dairy products or green, leafy vegetables Ø Effects: Ø Increased neuromuscular excitability • Tingling, muscle spasm (particularly in hands, feet, and facial muscles), intestinal cramping, hyperactive bowel sounds Severe cases show convulsions and tetany Ø Prolonged QT interval, cardiac arrest Ø Copyright © 2017, Elsevier Inc. All rights reserved. 31

Hypercalcemia Causes: Ø Ø Ø Hyperparathyroidism Bone metastases with calcium resorption from breast, prostate, renal, and cervical cancer Sarcoidosis Excess vitamin D Many tumors that produce PTH Effects: Many nonspecific: fatigue, weakness, lethargy, anorexia, nausea, constipation Ø Impaired renal function, kidney stones Ø Dysrhythmias, bradycardia, cardiac arrest Ø Bone pain, osteoporosis Ø Copyright © 2017, Elsevier Inc. All rights reserved. 32

Phosphate Like calcium, most phosphate is also located in the bone Provides energy for muscle contraction Parathyroid hormone, vitamin D 3, and calcitonin act together to control phosphate absorption and excretion Normal value = 2. 5 -5. 0 mg/dl Copyright © 2017, Elsevier Inc. All rights reserved. 33

Hypophosphatemia Causes: Intestinal malabsorption (vitamin D deficiency, use of magnesium- and aluminumcontaining antacids, longterm alcohol abuse) Ø Malabsorption syndromes Ø Respiratory alkalosis Ø Increased renal excretion of phosphate associated with hyperparathyroidism Ø Effects: Reduced capacity for oxygen transport by red blood cells, thus disturbed energy metabolism Ø Leukocyte and platelet dysfunction Ø Deranged nerve and muscle function Ø In severe cases, irritability, confusion, numbness, coma, convulsions, possibly respiratory failure, cardiomyopathies, bone resorption Ø Copyright © 2017, Elsevier Inc. All rights reserved. 34

Hyperphosphatemia Causes: Acute or chronic renal failure with significant loss of glomerular filtration Ø Treatment of metastatic tumors with chemotherapy that releases large amounts of phosphate into serum Ø Long-term use of laxatives or enemas containing phosphates Ø Hypoparathyroidism Ø Effects: Symptoms primarily related to low serum calcium levels (caused by high phosphate levels) similar to the results of hypocalcemia Ø When prolonged, calcification of soft tissues in lungs, kidneys, joints Ø Copyright © 2017, Elsevier Inc. All rights reserved. 35

Magnesium Intracellular cation Serum concentration 1. 8 to 3. 0 m. Eq/L Acts as a cofactor in intracellular enzymatic reactions Increases neuromuscular excitability Copyright © 2017, Elsevier Inc. All rights reserved. 36

Hypomagnesemia Causes: Malnutrition Ø Malabsorption syndromes Ø Alcoholism Ø Urinary losses (renal tubular dysfunction, loop diuretics) Ø Effects: Behavioral changes Ø Irritability Ø Increased reflexes Ø Muscle cramps Ø Ataxia Ø Nystagmus Ø Tetany Ø Convulsions Ø Tachycardia Ø Hypotension Ø Copyright © 2017, Elsevier Inc. All rights reserved. 37

Hypermagnesemia Causes: Usually renal insufficiency or failure Ø Excessive intake of magnesium-containing antacids Ø Adrenal insufficiency Ø Effects: Ø Ø Ø Ø Skeletal smooth muscle contraction Excess nerve function Loss of deep tendon reflexes Nausea and vomiting Muscle weakness Hypotension Bradycardia Respiratory distress Copyright © 2017, Elsevier Inc. All rights reserved. 38

1. Which is the initial treatment for hypernatremia? A. B. C. D. Restriction of fluids Administration of a diuretic Isotonic 0. 9% normal saline fluid Isotonic salt-free fluid (5% dextrose in water) Copyright © 2017, Elsevier Inc. All rights reserved. 39

Acid-Base Balance Acid-base balance is carefully regulated to maintain a normal p. H via multiple mechanisms Copyright © 2017, Elsevier Inc. All rights reserved. 40

p. H Negative logarithm of the H+ concentration If the H+ are high in number, the p. H is low (acidic); if the H+ are low in number, the p. H is high (alkaline) Copyright © 2017, Elsevier Inc. All rights reserved. 41

p. H (Cont. ) Acids are formed as end products of protein, carbohydrate, and fat metabolism To maintain the body’s normal p. H (7. 35 -7. 45), the H+ must be neutralized or excreted The bones, lungs, and kidneys are the major organs involved in the regulation of acid-base balance Copyright © 2017, Elsevier Inc. All rights reserved. 42

p. H (Cont. ) Body acids exist in two forms: Volatile • H 2 CO 3 (can be eliminated as CO 2 gas) Ø Nonvolatile • Sulfuric, phosphoric, and other organic acids • Eliminated by the renal tubules with the regulation of Ø HCO 3– Copyright © 2017, Elsevier Inc. All rights reserved. 43

Buffering Systems A buffer is a chemical that can bind excessive H+ or OH– without a significant change in p. H The most important plasma-buffering systems are the carbonic acid–bicarbonate pair Copyright © 2017, Elsevier Inc. All rights reserved. 44

Carbonic Acid–Bicarbonate Pair Operates in the lung and the kidney The greater the partial pressure of carbon dioxide, the more carbonic acid is formed At a p. H of 7. 4, the ratio of bicarbonate to carbonic acid is 20: 1 Ø Bicarbonate and carbonic acid can increase or decrease, but the ratio must be maintained Ø Copyright © 2017, Elsevier Inc. All rights reserved. 45

Carbonic Acid–Bicarbonate Pair (Cont. ) The respiratory system compensates by increasing ventilation to expire carbon dioxide or by decreasing ventilation to retain carbon dioxide The renal system compensates by producing acidic or alkaline urine Copyright © 2017, Elsevier Inc. All rights reserved. 46

Other Buffering Systems Protein buffering (hemoglobin) Ø Proteins have negative charges, so they can serve as buffers for H+ Renal buffering Ø Secretion of H+ in the urine and reabsorption of HCO 3– Copyright © 2017, Elsevier Inc. All rights reserved. 47

Acid-Base Imbalances Normal arterial blood p. H 7. 35 to 7. 45 Ø Obtained by arterial blood gas (ABG) sampling Ø Acidosis Ø Systemic increase in H+ concentration or decrease in bicarbonate (base) Alkalosis Ø Systemic decrease in H+ concentration or increase in bicarbonate Copyright © 2017, Elsevier Inc. All rights reserved. 48

Acidosis and Alkalosis Four categories of acid-base imbalances: Respiratory acidosis—elevation of p. CO 2 as a result of ventilation depression Ø Respiratory alkalosis—depression of p. CO 2 as a result of alveolar hyperventilation Ø Metabolic acidosis—depression of HCO 3– or an increase in noncarbonic acids Ø Metabolic alkalosis—elevation of HCO 3– usually caused by an excessive loss of metabolic acids Ø Copyright © 2017, Elsevier Inc. All rights reserved. 49
- Slides: 49