CHAPTER 5 DIFFUSION IN SOLIDS ISSUES TO ADDRESS





![CONCENTRATION PROFILES & FLUX • Concentration Profile, C(x): [kg/m 3] Adapted from Fig. 5. CONCENTRATION PROFILES & FLUX • Concentration Profile, C(x): [kg/m 3] Adapted from Fig. 5.](https://slidetodoc.com/presentation_image/618126da7a21d484aace35e8dcb09c22/image-9.jpg)


- Slides: 11

CHAPTER 5: DIFFUSION IN SOLIDS ISSUES TO ADDRESS. . . • How does diffusion occur? • Why is it an important part of processing? • How can the rate of diffusion be predicted for some simple cases? • How does diffusion depend on structure and temperature?

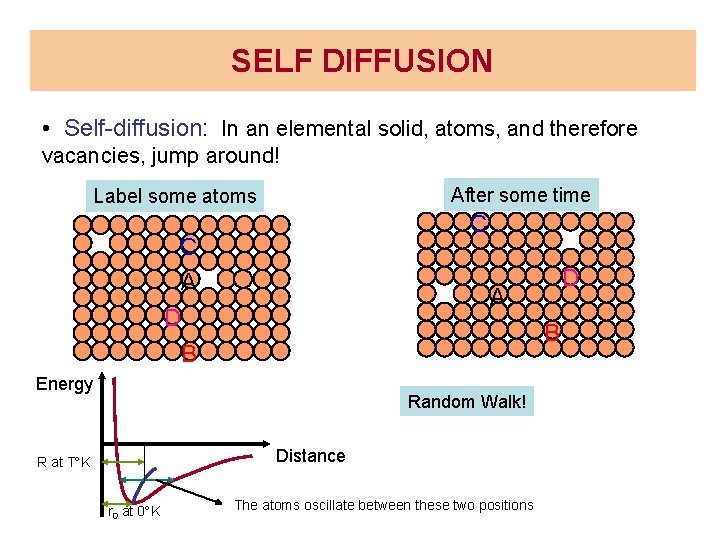
SELF DIFFUSION • Self-diffusion: In an elemental solid, atoms, and therefore vacancies, jump around! After some time Label some atoms C C A D B A B Energy Random Walk! Distance R at T K r 0 at 0 K D The atoms oscillate between these two positions

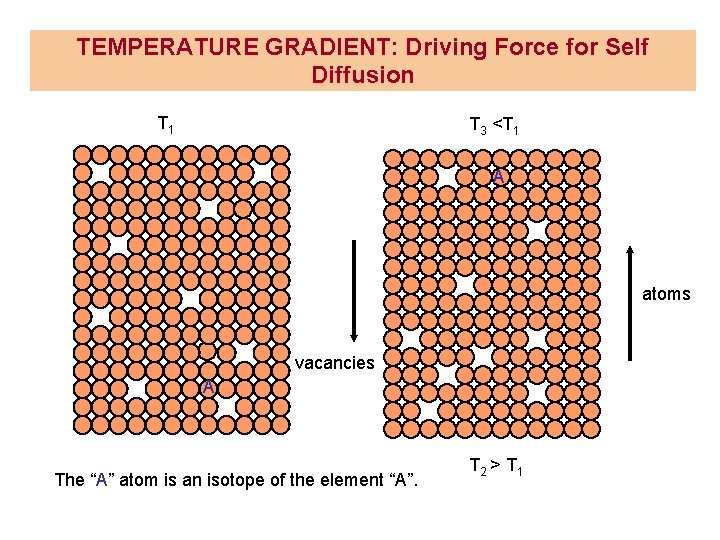
TEMPERATURE GRADIENT: Driving Force for Self Diffusion T 1 T 3 <T 1 A atoms vacancies A The “A” atom is an isotope of the element “A”. T 2 > T 1

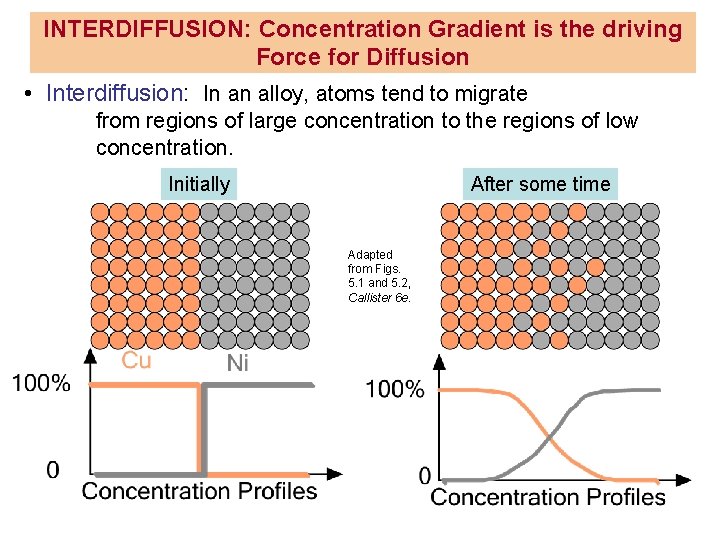
INTERDIFFUSION: Concentration Gradient is the driving Force for Diffusion • Interdiffusion: In an alloy, atoms tend to migrate from regions of large concentration to the regions of low concentration. Initially After some time Adapted from Figs. 5. 1 and 5. 2, Callister 6 e.

DIFFUSION MECHANISM Substitutional Diffusion: • applies to substitutional impurities • atoms exchange with vacancies • rate depends on: --number of vacancies --activation energy to exchange.

DIFFUSION SIMULATION • Simulation of interdiffusion across an interface: • Rate of substitutional diffusion depends on: --vacancy concentration --frequency of jumping. (Courtesy P. M. Anderson)

DIFFUSION MECHANISM Interstitial Diffusion: Ø Applies to interstitial impurities. Ø More rapid than the diffusion of substitutional atoms, because it does not require the presence of vacancies. Ø Simulation: shows the jumping of a smaller atom (gray) from one interstitial site to another in a BCC structure. The interstitial sites considered here at midpoints along the unit cell edges. (Courtesy P. M. Anderson)

MODELING DIFFUSION: FLUX • Flux: • Directional Quantity • Flux can be measured for: --vacancies --host (A) atoms --impurity (B) atoms
![CONCENTRATION PROFILES FLUX Concentration Profile Cx kgm 3 Adapted from Fig 5 CONCENTRATION PROFILES & FLUX • Concentration Profile, C(x): [kg/m 3] Adapted from Fig. 5.](https://slidetodoc.com/presentation_image/618126da7a21d484aace35e8dcb09c22/image-9.jpg)
CONCENTRATION PROFILES & FLUX • Concentration Profile, C(x): [kg/m 3] Adapted from Fig. 5. 2(c), Callister 6 e. • Fick's First Law: flux in x-dir. [kg/m 2 -s] Diffusion coefficient [m 2 /s] d. C = Jx D dx concentration gradient [kg/m 4 ] • The steeper the concentration profile, the greater the flux!

STEADY STATE DIFFUSION • Steady State: the concentration profile doesn't change with time. • Apply Fick's First Law: • If Jx)left = Jx)right , then • Result: the slope, d. C/dx, must be constant (i. e. , slope doesn't vary with position)!

EXAMPLE: STEADY STATE DIFFUSION • Steel plate at 700 C with geometry shown: • Q: How much carbon transfers from the rich to the deficient side? Adapted from Fig. 5. 4, Callister 6 e.