Chapter 5 Compounds and Their Bonds 5 1

Chapter 5 Compounds and Their Bonds 5. 1 Octet Rule and Ions General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 1

Octet Rule An octet § is 8 valence electrons § is associated with the stability of the noble gases § does not occur with He; He is stable with 2 valence electrons (duet) Valence Electrons He 1 s 2 2 Ne 1 s 22 p 6 Ar 1 s 22 p 63 s 23 p 6 8 Kr 1 s 22 p 63 s 23 p 64 s 23 d 104 p 6 8 8 General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 2
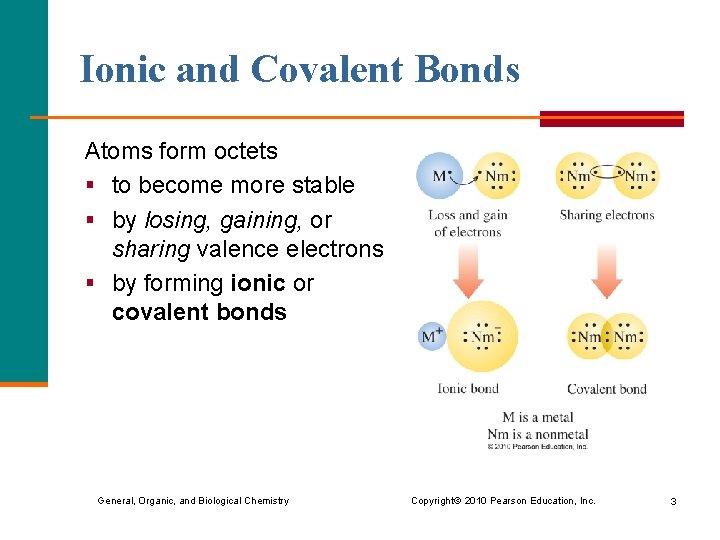
Ionic and Covalent Bonds Atoms form octets § to become more stable § by losing, gaining, or sharing valence electrons § by forming ionic or covalent bonds General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 3

Metals Form Positive Ions Metals form positive ions § by a loss of their valence electrons § with the electron configuration of the nearest noble gas § that have fewer electrons than protons Group 1 A(1) metals ion 1+ Group 2 A(2) metals ion 2+ Group 3 A(3) metals ion 3+ General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 4

Formation of a Sodium Ion, Na+ Sodium achieves an octet by losing its one valence electron. General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 5

Charge of Sodium Ion, Na+ With the loss of its valence electron, a sodium ion has a 1+ charge. Sodium atom Sodium ion 11 p+ 11 e– 10 e– 0 1 + General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 6
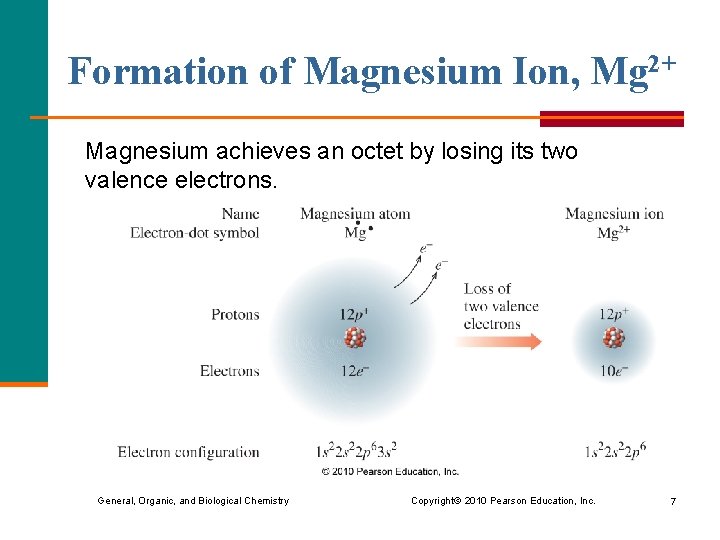
Formation of Magnesium Ion, Mg 2+ Magnesium achieves an octet by losing its two valence electrons. General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 7

Charge of Magnesium Ion, Mg 2+ With the loss of two valence electrons magnesium forms a positive ion with a 2+ charge. Mg atom Mg 2+ ion 12 p+ 12 e– 10 e– 0 2+ General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 8

Learning Check A. The number of valence electrons in aluminum is 1) 1 e– 2) 2 e– 3) 3 e– B. To acquire an octet of electrons in aluminum requires 1) a loss of 3 e– 2) a gain of 3 e– 3) a gain of 5 e– C. The ionic charge of aluminum is 1) 3– 2) 5– 3) 3+ D. The symbol for the aluminum ion is 1) Al 3+ 2) Al 3– 3) Al+ General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 9

Solution A. The number of valence electrons in aluminum is 3) 3 e– B. To acquire an octet of electrons in aluminum requires 1) a loss of 3 e– C. The ionic charge of aluminum is 3) 3+ D. The symbol for the aluminum ion is 1) Al 3+ General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 10

Formation of Negative Ions In ionic compounds, nonmetals § achieve an octet arrangement § gain electrons § form negatively charged ions with 3–, 2–, or 1– charges General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 11
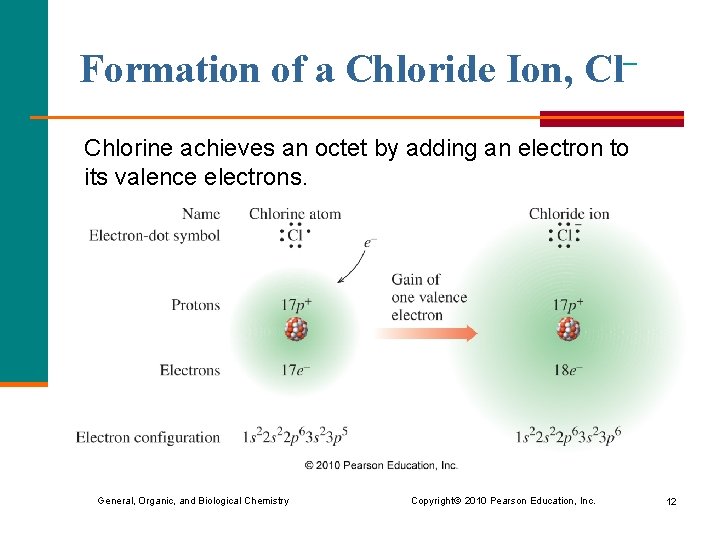
Formation of a Chloride Ion, Cl– Chlorine achieves an octet by adding an electron to its valence electrons. General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 12

Charge of a Chloride Ion, Cl– § By gaining one electron, the chloride ion has a – 1 charge. Chlorine atom Chloride ion 17 p+ 17 e– 18 e– 0 1– General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 13

Ionic Charge from Group Numbers § The charge of a positive ion is equal to its Group number. Group 1 A(1) = 1+ Group 2 A(2) = 2+ Group 3 A(3) = 3+ § The charge of a negative ion is obtained by subtracting 8 or 18 from its Group number. Group 6 A(16) = 6 – 8 = 2– or 16 – 18 = 2– General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 14
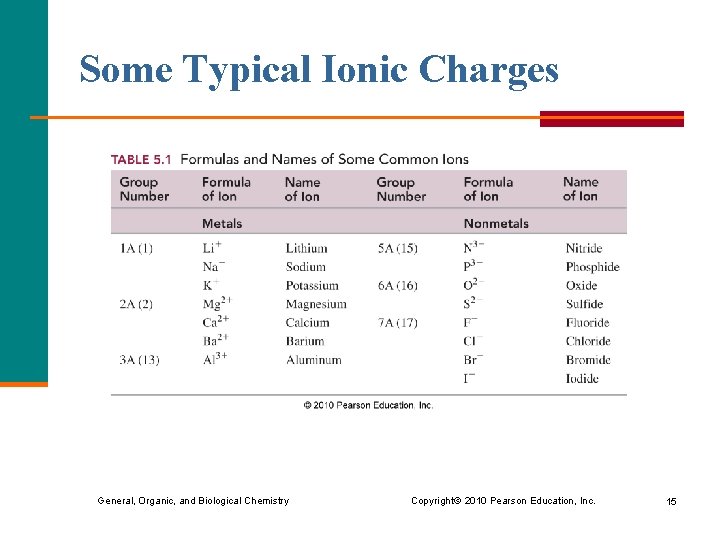
Some Typical Ionic Charges General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 15

Group Number and Ionic Charge Ions § achieve the electron configuration of their nearest noble gas § of metals in Groups 1 A(1), 2 A(2), or 3 A(13) have positive 1+, 2+, or 3+ charge. § Of nonmetals in Groups 5 A(15), 6 A(16), or 7 A(17) have negative 3–, 2–, or 1– charge. General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 16

Groups Numbers for Some Positive and Negative Ions General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 17

Learning Check A. The Group number for sulfur is 1) 4 A(14) 2) 8 A(18) 3) 6 A(16) B. The number of valence electrons in sulfur is 1) 4 e 2) 6 e 3) 8 e C. The change in electrons for an octet requires a 1) gain of 2 e 2) loss of 2 e 3) a gain of 4 e D. The ionic charge of sulfur is 1) 2+ 2) 2 3) 4 General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 18

Solution A. The Group number for sulfur is 3) 6 A(16) B. The number of valence electrons in sulfur is 2) 6 e C. The change in electrons for octet requires a 1) gain of 2 e D. The ionic charge of sulfur is 2) 2 General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 19

Sizes of Atoms and Their Ions Metals § lose their valence electrons § form ions that are smaller than their atoms Nonmetals (in ionic compounds) § gain valence electrons § form ions that are larger than their atoms General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 20

Metal Ions are Smaller In Group 1 A(1), the ions are smaller than their corresponding atoms. General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 21

Nonmetal Ions are Larger In Group 7 A(17), the ions are larger than their corresponding atoms. General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 22

Learning Check Select the larger atom or ion in the following: 1. Mg or Mg 2+ 2. S or S 2 3. Br or Br General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 23

Solution Select the larger atom or ion in the following: 1. Mg or Mg 2+ Mg 2. S or S 2 3. Br or Br General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 24
- Slides: 24