Chapter 5 Chemical Reactions 5 1 Chemical Equations

















- Slides: 22

Chapter 5 Chemical Reactions 5. 1 Chemical Equations 1

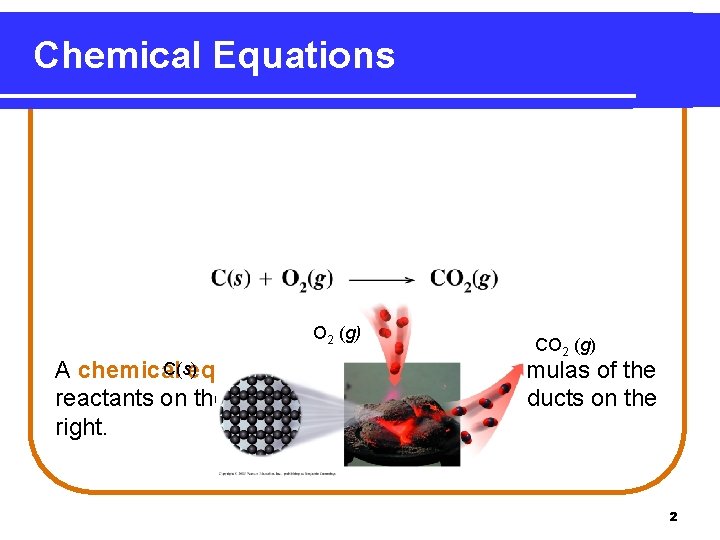
Chemical Equations O 2 (g) C(s) A chemical equation gives the chemical formulas of the reactants on the left of the arrow and the products on the right. Reactants Product 2

Symbols Used in Equations Symbols used in chemical equations show TABLE 5. 2 • the states of the reactants. • the states of the products. • the reaction conditions. 3

Chemical Equations are Balanced In a balanced chemical reaction • atoms are not gained or lost. • the number of reactant atoms is equal to the number of product atoms. 4

A Balanced Chemical Equation In a balanced chemical equation, • there must be the same number of each type of atom on the reactant side and on the product side of a balanced equation. • numbers called coefficients are used in front of one or more formulas. Al + S 2 Al + 3 S 2 Al 3 S Al 2 S 3 = 2 Al = 3 S Not Balanced 5

Learning Check State the number of atoms of each element on the reactant side and the product side for each of the following balanced equations. A. P 4(s) + 6 Br 2(l) 4 PBr 3(g) B. 2 Al(s) + Fe 2 O 3(s) 2 Fe(s) + Al 2 O 3(s) 6

Solution A. P 4(s) + 6 Br 2(l) 4 PBr 3(g) 4 P 12 Br B. 2 Al(s) + Fe 2 O 3(s) 2 Fe(s) + Al 2 O 3(s) 2 Al 2 Fe 3 O 7

Learning Check Determine if each equation is balanced or not. A. Na(s) + N 2(g) Na 3 N(s) B. C 2 H 4(g) + H 2 O(l) C 2 H 5 OH(l) 8

Solution Determine if each equation is balanced or not. A. Na(s) + N 2(g) Na 3 N(s) No. 2 N on reactant side, 1 N on product side. 1 Na on reactant side, 3 Na on product side. B. C 2 H 4(g) + H 2 O(l) C 2 H 5 OH(l) Yes. 2 C = 2 C 6 H = 6 H 1 O = 1 O 9

Guide to Balancing a Chemical Equation Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 10

Steps in Balancing an Equation To balance the following equation, Fe 3 O 4(s) + H 2(g) Fe(s) + H 2 O(l) • work on one element at a time. • use only coefficients in front of formulas. • do not change any subscripts. Fe: Fe 3 O 4(s) + H 2(g) 3 Fe(s) + H 2 O(l) O: Fe 3 O 4(s) + H 2(g) 3 Fe(s) + 4 H 2 O(l) H: Fe 3 O 4(s) + 4 H 2(g) 3 Fe(s) + 4 H 2 O(l) 11

Balancing Chemical Equations 1. Write the equation with the correct formulas. NH 3(g) + O 2(g) NO(g) + H 2 O(g) 2. Determine if the equation is balanced. No, not all atoms are balanced. 3. Balance with coefficients in front of formulas. 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) 4. Check that atoms of each element are equal in reactants and products. 4 N (4 x 1 N) = 4 N (4 x 1 N) 12 H (4 x 3 H) = 12 H (6 x 2 H) 10 O (5 x 2 O) = 10 O (4 O + 6 O) 12

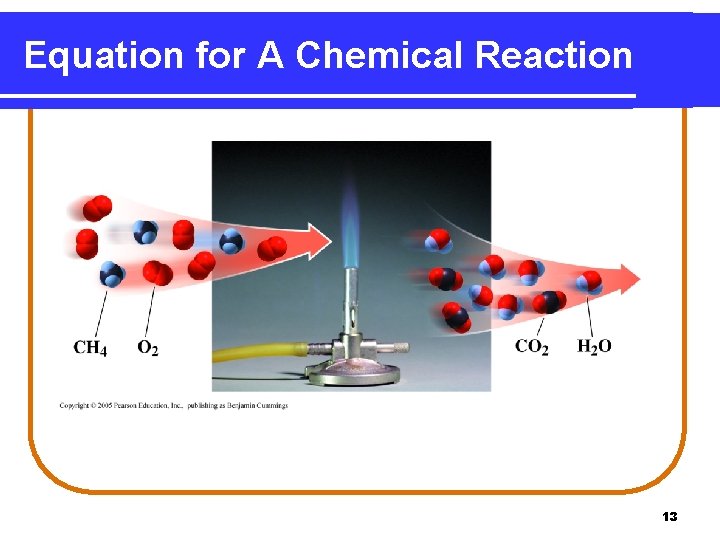
Equation for A Chemical Reaction 13

Checking a Balanced Equation Reactants 1 C atom 4 H atoms 4 O atoms = = = Products 1 C atom 4 H atoms 4 O atoms 14

Learning Check the balance of atoms in the following. Fe 3 O 4(s) + 4 H 2(g) 3 Fe(s) + 4 H 2 O(l) A. Number of H atoms in products. 1) 2 2) 4 3) 8 B. Number of O atoms in reactants. 1) 2 2) 4 3) 8 C. Number of Fe atoms in reactants. 1) 1 2) 3 3) 4 15

Solution Fe 3 O 4(s) + 4 H 2(g) 3 Fe(s) + 4 H 2 O(l) A. Number of H atoms in products. 3) 8 (4 H 2 O) B. Number of O atoms in reactants. 2) 4 (Fe 3 O 4) C. Number of Fe atoms in reactants. 2) 3 (Fe 3 O 4) 16

Learning Check Balance each equation and list the coefficients in the balanced equation going from reactants to products: A. __Mg(s) 1) 1, 3, 2 B. __Al(s) 1) 3, 3, 2 + __N 2(g) __Mg 3 N 2(s) 2) 3, 1, 2 3) 3, 1, 1 + __Cl 2(g) 2) 1, 3, 1 __Al. Cl 3(s) 3) 2, 3, 2 17

Solution A. 3) 3, 1, 1 3 Mg(s) + 1 N 2(g) 1 Mg 3 N 2(s) B. 3) 2, 3, 2 2 Al(s) + 3 Cl 2(g) 2 Al. Cl 3(s) 18

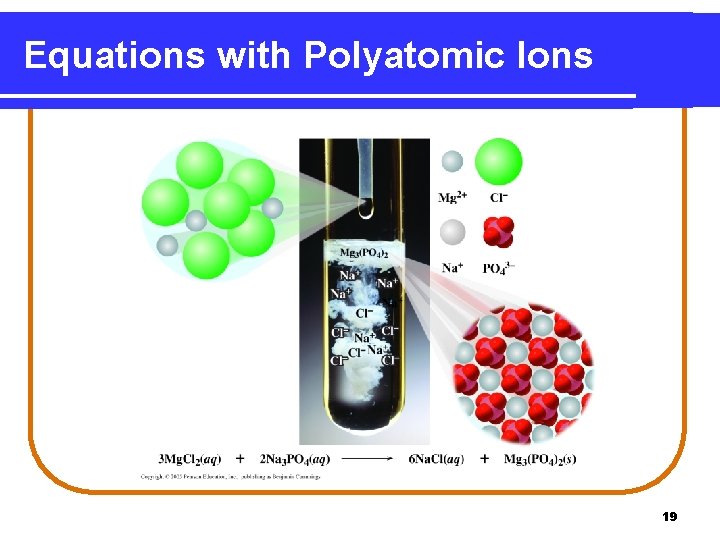
Equations with Polyatomic Ions 19

Balancing with Polyatomic Ions Mg. Cl 2(aq) + Na 3 PO 4(aq) Na. Cl(aq) + Mg 3(PO 4)2(s) Balance PO 43 - as a unit Mg. Cl 2(aq) + 2 Na 3 PO 4(aq) Na. Cl(aq) + Mg 3(PO 4)2(s) 2 PO 43= 2 PO 43 - Balance Mg and Cl 3 Mg. Cl 2(aq) + 2 Na 3 PO 4(aq) 6 Na. Cl(aq) + Mg 3(PO 4)2(s) 3 Mg 2+ = 3 Mg 2+ 6 Na+ = 6 Na+ 6 Cl= 6 Cl 20

Learning Check Balance and list the coefficients from reactants to products. A. __Fe 2 O 3(s) + __C(s) __Fe(s) + __CO 2(g) 1) 2, 3, 2, 3 2) 2, 3, 4, 3 3) 1, 1, 2, 3 B. __Al(s) + __Fe. O(s) __Fe(s) + __Al 2 O 3(s) 1) 2, 3, 3, 1 2) 2, 1, 1, 1 3) 3, 3, 3, 1 C. __Al(s) + __H 2 SO 4(aq) __Al 2(SO 4)3(aq) + __H 2(g) 1) 3, 2, 1, 2 2) 2, 3, 1, 3 3) 2, 3, 2, 3 21

Solution A. 2) 2, 3, 4, 3 2 Fe 2 O 3(s) + 3 C(s) 4 Fe(s) B. 1) 2, 3, 3, 1 2 Al(s) + 3 Fe. O(s) + 3 CO 2(g) 3 Fe(s) + 1 Al 2 O 3(s) C. 2) 2, 3, 1, 3 2 Al(s) + 3 H 2 SO 4(aq) 1 Al 2(SO 4)3(aq) + 3 H 2(g) 22