Chapter 5 Chemical Bonds The Ties That Bind

Chapter 5 Chemical Bonds: The Ties That Bind Chapter 5

Chemical Bonds • Forces responsible for holding together atoms in molecules and ions in crystals • Determine shape of molecules • Predict chemical and physical properties of materials • Related to arrangement of electrons in compounds Chapter 5 2

Stable Electron Configurations • Valence electrons – outermost level with electrons • Core electrons – all other electrons in an atom • Isoelectronic – same number of valence electrons Chapter 5 3
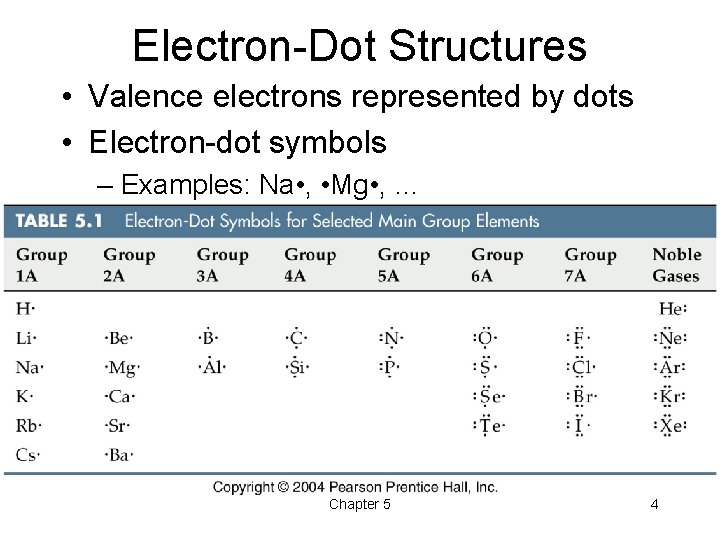
Electron-Dot Structures • Valence electrons represented by dots • Electron-dot symbols – Examples: Na • , • Mg • , … Chapter 5 4
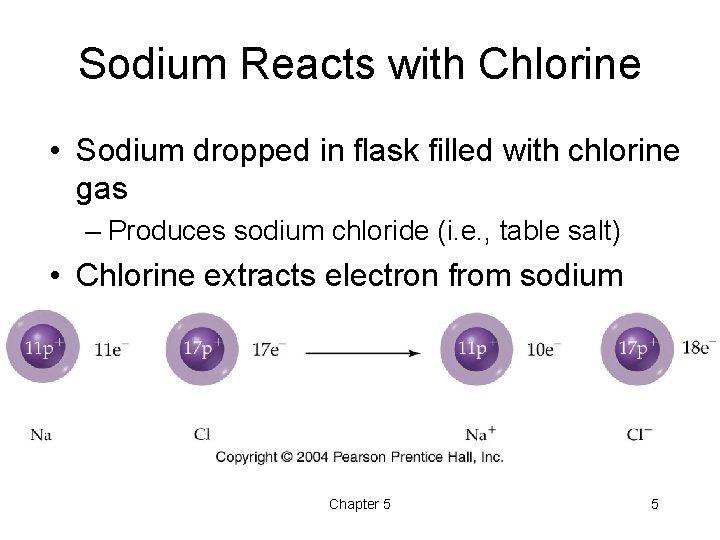
Sodium Reacts with Chlorine • Sodium dropped in flask filled with chlorine gas – Produces sodium chloride (i. e. , table salt) • Chlorine extracts electron from sodium Chapter 5 5

Ionic Bonds • Na+ and Cl– • Opposite charges attract • Ions organize themselves in orderly manner – Crystal of Na. Cl Chapter 5 6

More Ionic Compounds Chapter 5 7

Octet Rule • Atoms tend to gain or lose electrons to have eight valence electrons – Same as noble gases – He and H are exceptions, get only two valence electrons Chapter 5 8

Naming Ions • For cations, simple positive ions – Add the word ion – Examples: Na+ – sodium ion Al 3+ – aluminum ion • For anions, simple negative ions – Change the usual ending to -ide – Examples: Cl– – chloride S 2– – sulfide Chapter 5 9
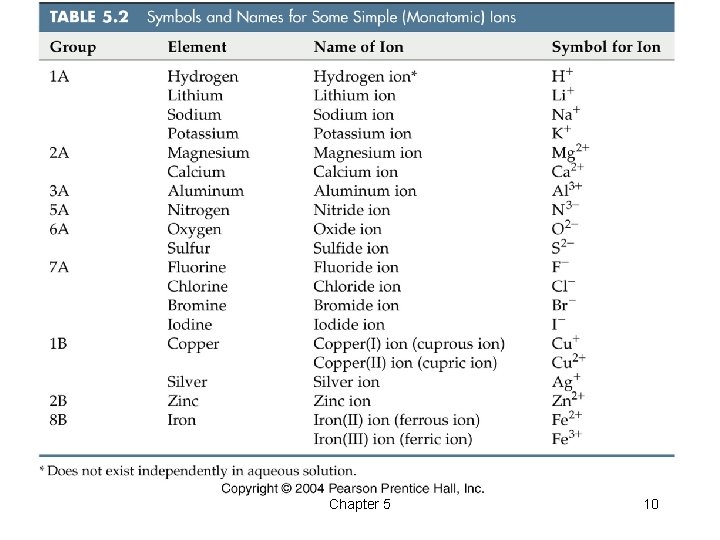
Chapter 5 10

Naming Binary Ionic Compounds • Two components in compound Chapter 5 11

Covalent Bonds • Bond formed by a shared pair of electrons • Gives atom an octet of electrons – Shared pair of electrons – bonding pair – Other electrons not involved in bonding – nonbonding pairs Chapter 5 12

Multiple Bonds • Sometimes atoms need to share more than one pair of electrons • Share two pairs – double bond • Share three pairs – triple bond Chapter 5 13

Naming Covalent Compounds Use prefixes to indicate the number of each kind of atom Chapter 5 14

Electronegativity • Measure of the attraction of an atom in a molecule for a pair of shared electrons – Fluorine most electronegative element Chapter 5 15

Polar Covalent Bonds • If elements do not have the same electronegativity, they get unequal sharing of electrons Chapter 5 16
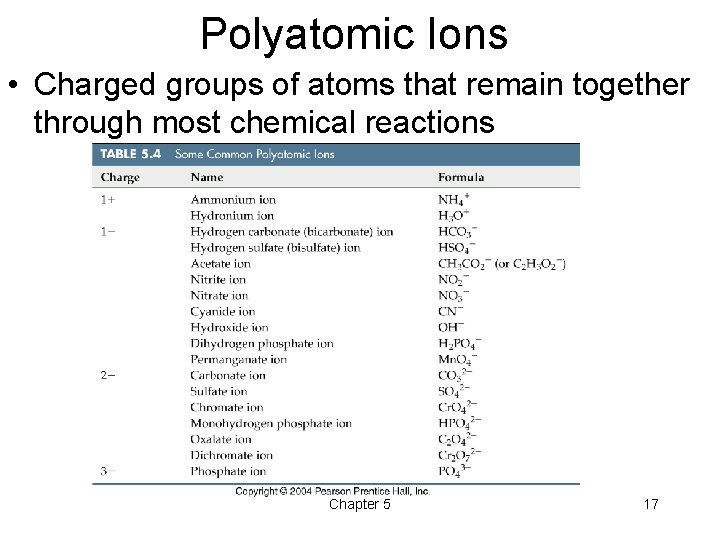
Polyatomic Ions • Charged groups of atoms that remain together through most chemical reactions Chapter 5 17

Draw Electron-Dot Formula 1. Determine total number of valence electrons 2. Write skeletal structure and connect atoms by dashed line 3. Give outer atoms an octet of electrons 4. Subtract number of electrons assigned 5. Add remaining electrons to central atom Chapter 5 18

6. If central atom does not have an octet, form multiple bonds using nonbonding electrons from outer atoms Chapter 5 19
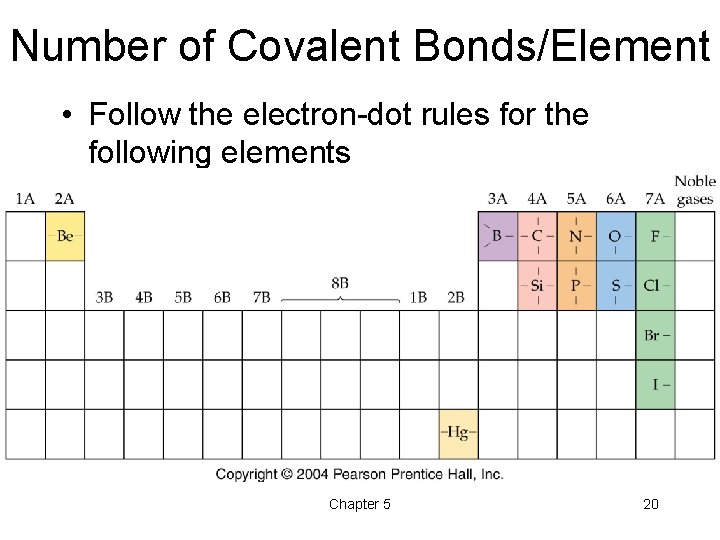
Number of Covalent Bonds/Element • Follow the electron-dot rules for the following elements Chapter 5 20

Free Radicals • Have odd number of electrons – One unpaired electron – Tend to be very reactive • Nitrogen oxides (such as NO, NO 2) common radical – Key component in smog Chapter 5 21

Molecular Shape • Determine reactive, chemical and physical properties – Very important in biological systems • Must draw electron-dot diagram to predict shape of molecule • Look at central atom Chapter 5 22
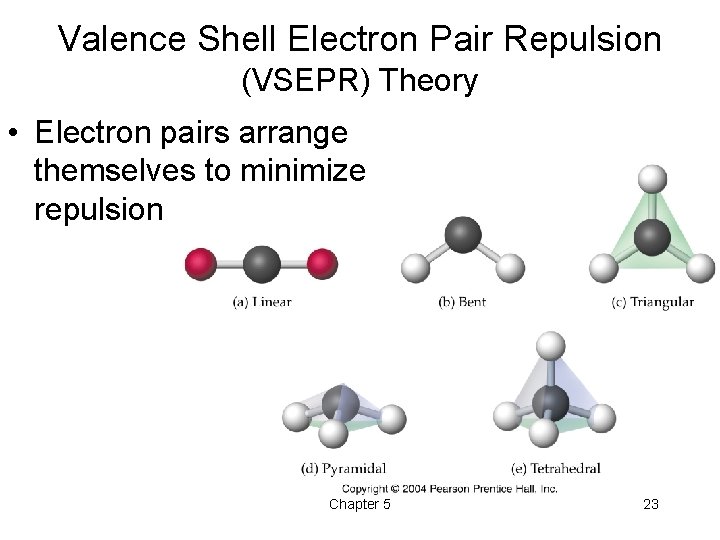
Valence Shell Electron Pair Repulsion (VSEPR) Theory • Electron pairs arrange themselves to minimize repulsion Chapter 5 23
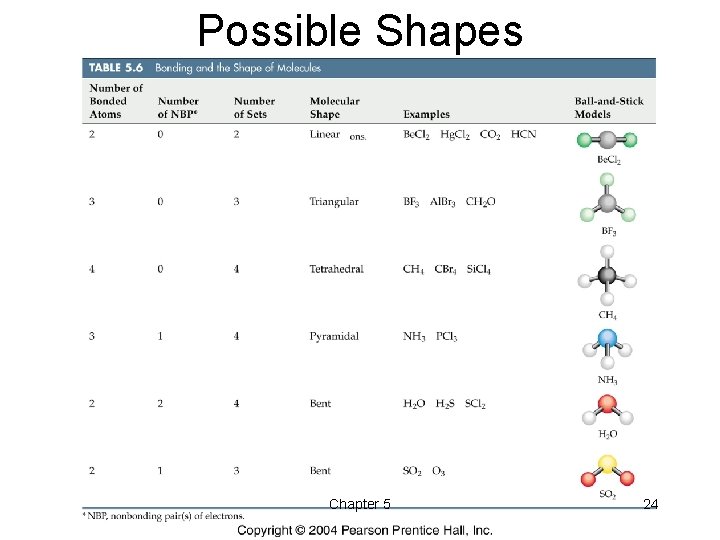
Possible Shapes Chapter 5 24
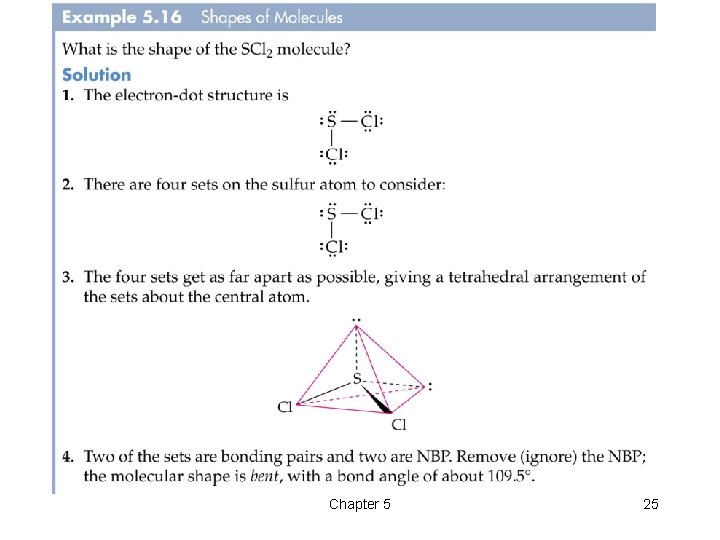
Chapter 5 25
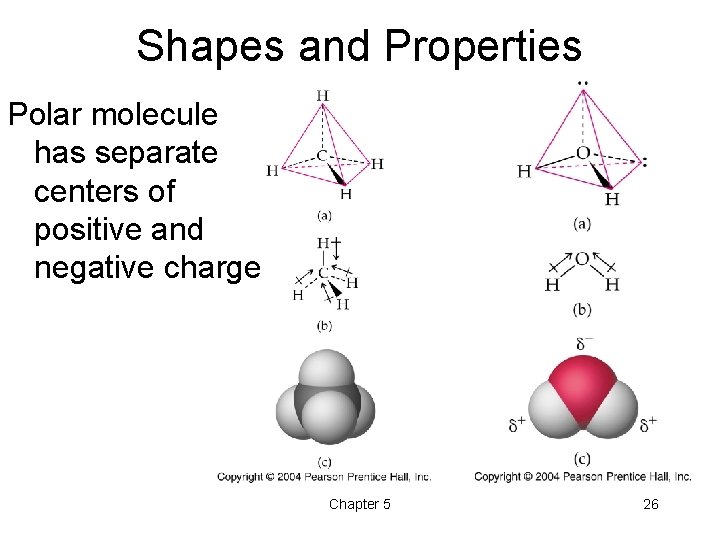
Shapes and Properties Polar molecule has separate centers of positive and negative charge Chapter 5 26
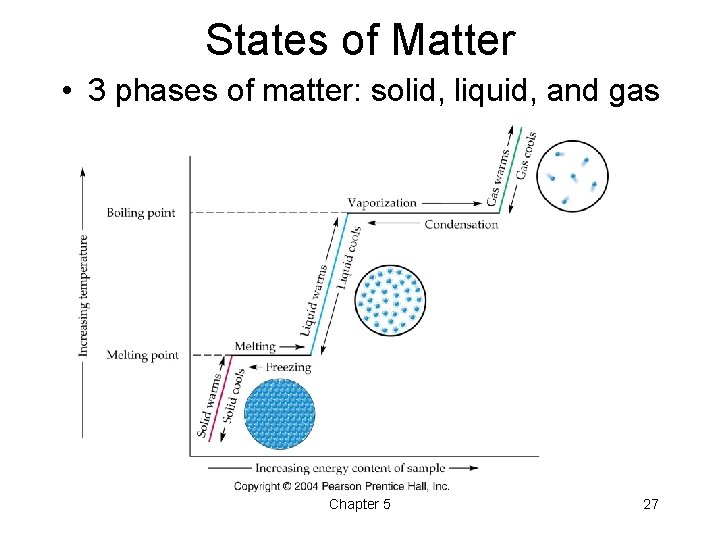
States of Matter • 3 phases of matter: solid, liquid, and gas Chapter 5 27

Intermolecular Forces • Glue that holds matter together • Melting and boiling points measure the relative strength • Ionic forces – strongest – Found in salts – Na. Cl melts at 800°C Chapter 5 28
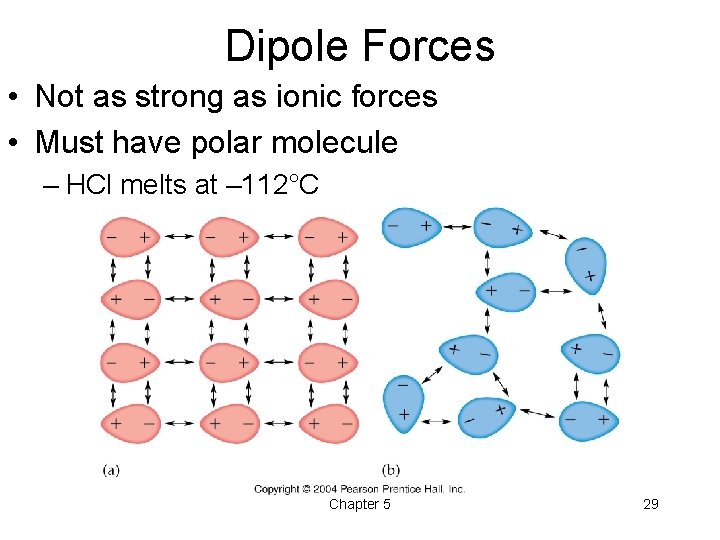
Dipole Forces • Not as strong as ionic forces • Must have polar molecule – HCl melts at – 112°C Chapter 5 29

Hydrogen Bonding • Hydrogen must be attached to electronegative atom – N, O, F • Plays important role in biological systems Chapter 5 30

Dispersion Forces • Present in all molecules • Weak momentary attractive forces – Arise for electrons moving about in molecules and atoms – Strong in larger molecules – Important in nonpolar compounds Chapter 5 31
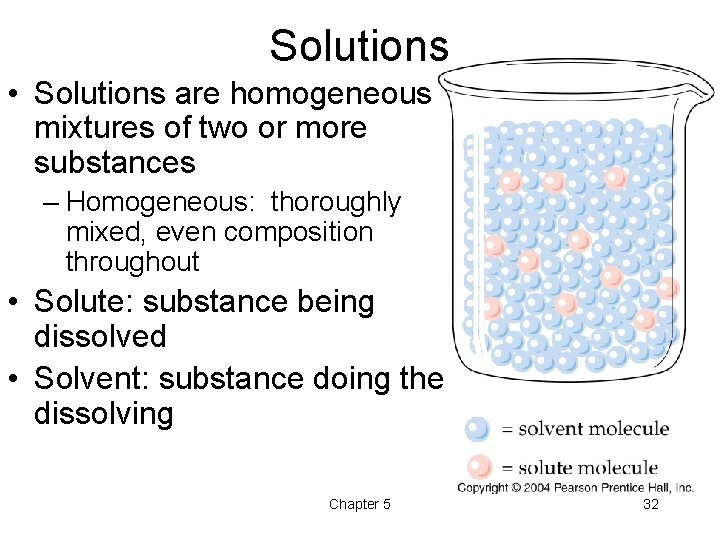
Solutions • Solutions are homogeneous mixtures of two or more substances – Homogeneous: thoroughly mixed, even composition throughout • Solute: substance being dissolved • Solvent: substance doing the dissolving Chapter 5 32

Forces in Solution • Like dissolves like • Nonpolar compounds dissolve best in nonpolar solvents – Oil and gasoline mix • Polar compounds dissolve best in polar solvents – Water and HCl mix Chapter 5 33
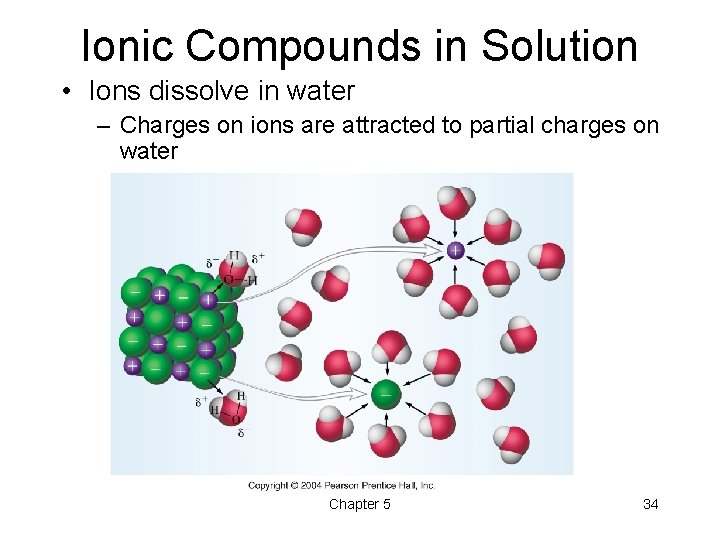
Ionic Compounds in Solution • Ions dissolve in water – Charges on ions are attracted to partial charges on water Chapter 5 34

End Of Chapter 5 35
- Slides: 35