Chapter 5 Chemical Bonding the Covalent Bond Model
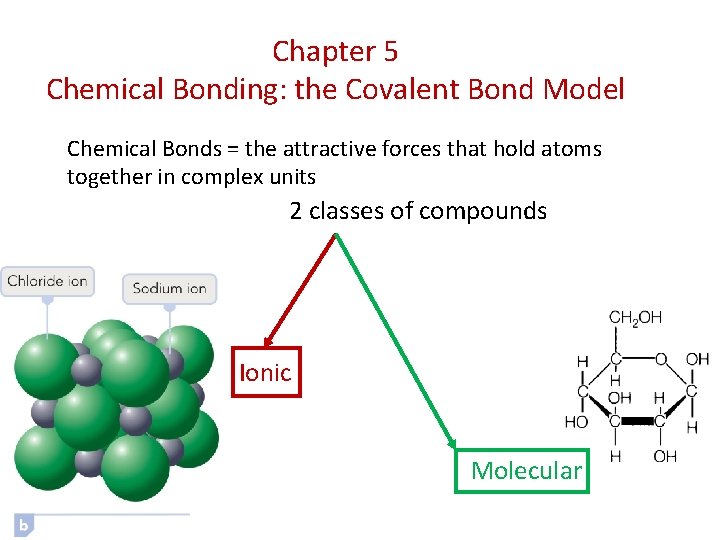
Chapter 5 Chemical Bonding: the Covalent Bond Model Chemical Bonds = the attractive forces that hold atoms together in complex units 2 classes of compounds Ionic Molecular

Names & Formulas of Binary Covalent/Molecular Compounds Formula Name CBr 4 phosphorus pentachloride S 2 O 4 SF 6

2 classes of compounds Ionic character: properties: examples: naming: Molecular (covalent)
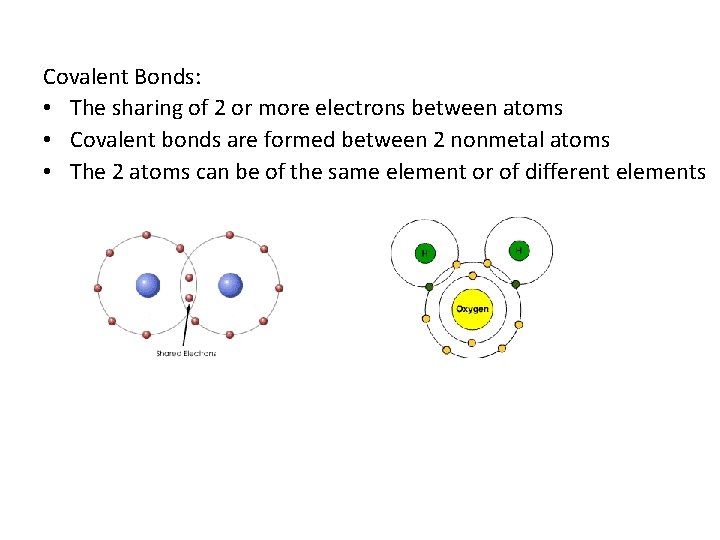
Covalent Bonds: • The sharing of 2 or more electrons between atoms • Covalent bonds are formed between 2 nonmetal atoms • The 2 atoms can be of the same element or of different elements

Diatomic Elements: H 2 F 2 O 2 N 2
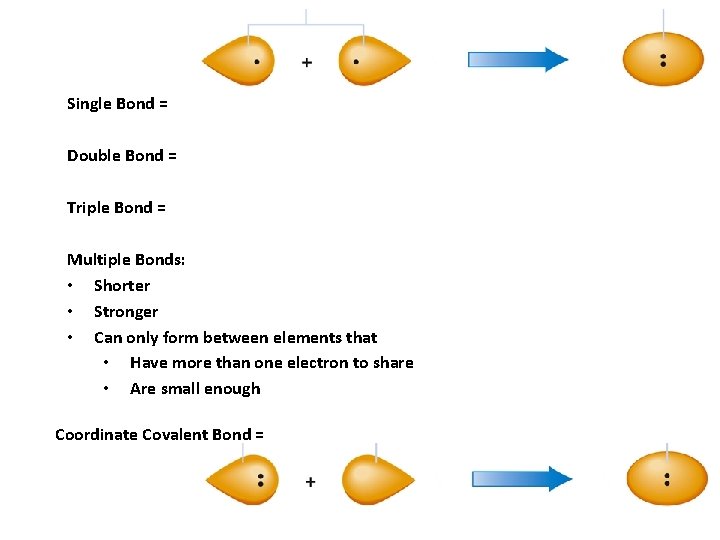
Single Bond = Double Bond = Triple Bond = Multiple Bonds: • Shorter • Stronger • Can only form between elements that • Have more than one electron to share • Are small enough Coordinate Covalent Bond =

Sharing Electrons between Atoms of Different Elements

Drawing Lewis Dot Structures for Molecules (H + 2 nd period element): 1. Find the total valence electrons. HF H 2 O NH 3 CH 4 2. Find the central atom—H is always “terminal”. 3. Write the symbols of the atoms in a skeletal arrangement & place a single bond between adjacent atoms – subtract the electrons used. 4. Place lone pairs of electrons around outermost atoms to satisfy octet rule (H is satisfied with 2 electrons). 5. If electrons remain, assign them to the central atom.

Drawing Lewis Dot Structures for Molecules: 1. NI 3 Find the total valence electrons. PF 3 HCN CO 2 2. Find the central atom: —H & F are always “terminal” — the central atom is the one that appears once — for H, O & something else, the something else is central — C is central in most C-containing compounds. 3. Arrange the atoms & connect with single bonds 4. Place lone pairs of electrons around outermost atoms to satisfy octet rule. 5. If electrons remain, assign them to the central atom. 6. If the central atom still does not have 4 pair, convert one or more terminal atom lone pairs to double or triple bonds (C, O, N, S, P only).

Drawing Lewis Dot Structures for Molecules: Cl 2 O HOCl COCl 2 CH 3 OH

Drawing Lewis Dot Structures for Molecules: SO 2 SO 3 Resonance: a molecule or ion can be represented by 2 or more equivalent Lewis structures—the true structure is a hybrid of the resonance forms BH 3 Exceptions to the Octet Rule?

Drawing Lewis Dot Structures for Polyatomic Ions: 1. Find the total valence electrons—for ions, add 1 electron for each -1 charge, subtract 1 electron for each +1 charge. NO 3 - SO 42 - NH 4+
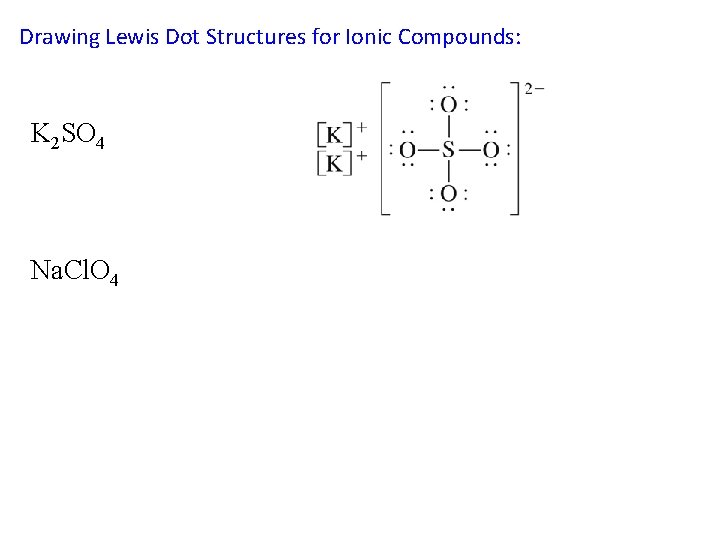
Drawing Lewis Dot Structures for Ionic Compounds: K 2 SO 4 Na. Cl. O 4
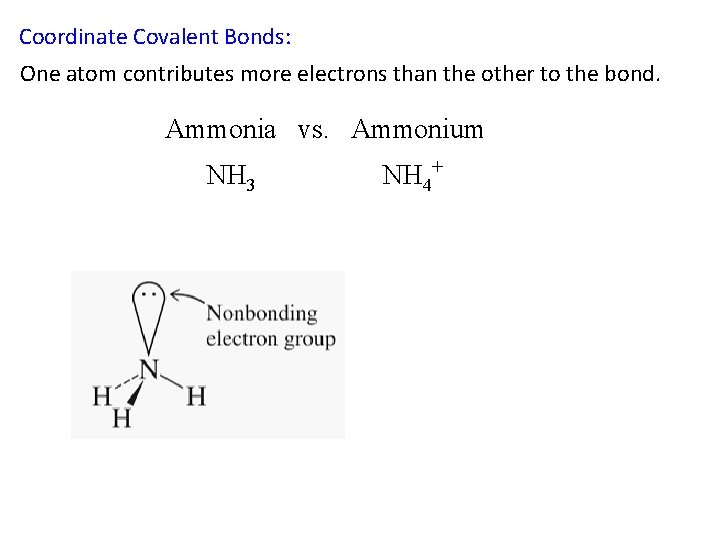
Coordinate Covalent Bonds: One atom contributes more electrons than the other to the bond. Ammonia vs. Ammonium NH 3 NH 4+
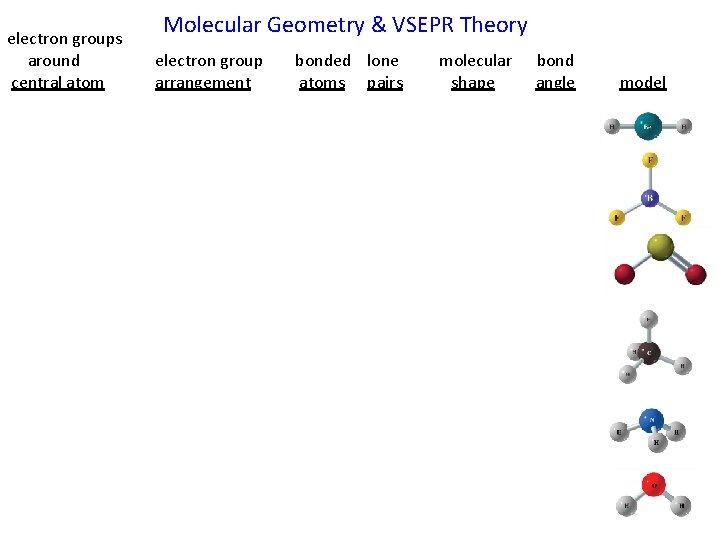
electron groups around central atom Molecular Geometry & VSEPR Theory electron group arrangement bonded lone atoms pairs molecular shape bond angle model

formula valence e SF 2 As. Br 3 SO 2 CCl 4 BF 3 NO 3 - electron dot structure electron pairs/ lone pairs shape angles

Molecules with more than one central atom: show all bond angles Acetylene hydrogen peroxide hydrogen azide HCCH HOOH HNNN
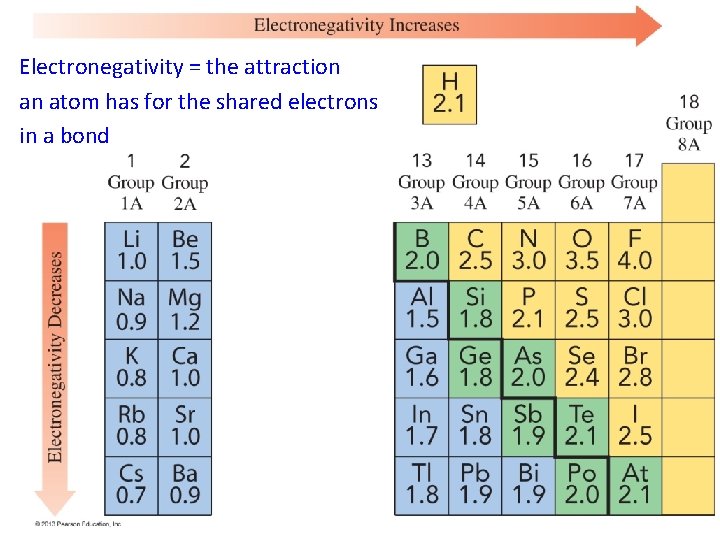
Electronegativity = the attraction an atom has for the shared electrons in a bond
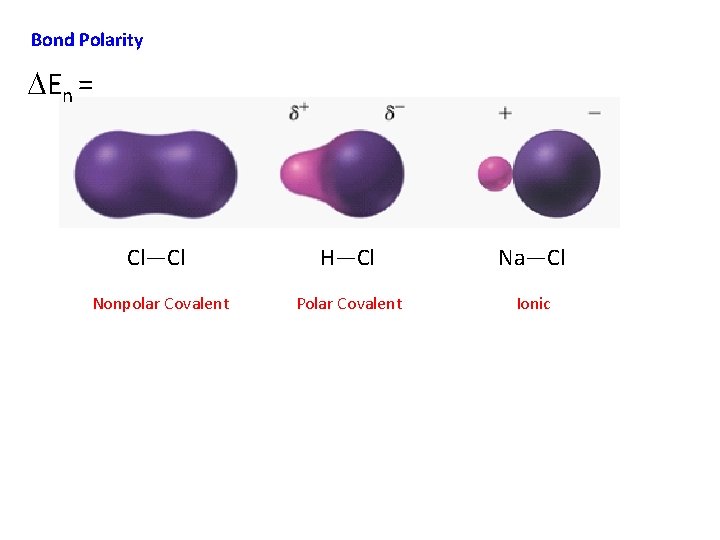
Bond Polarity DEn = Cl—Cl H—Cl Na—Cl Nonpolar Covalent Polar Covalent Ionic
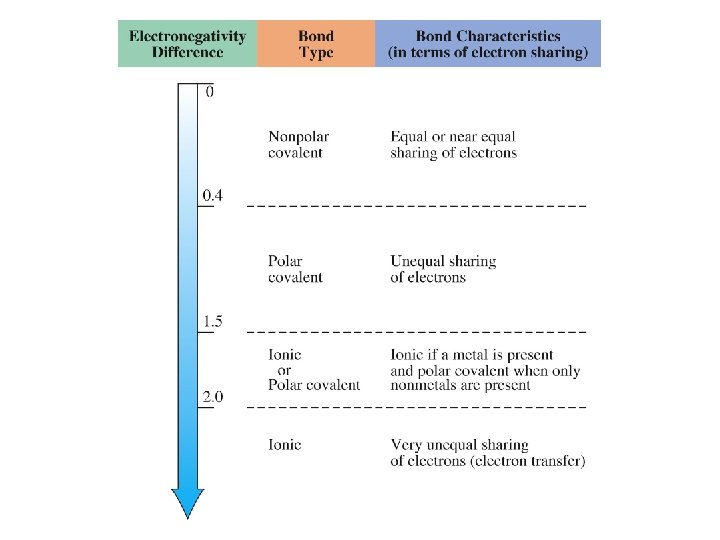

Bond Polarity Label each bond as Nonpolar, Polar Covalent or Ionic -if polar, idicate the direction of the polarity H—H C—O N—S N—O F—Cl N—Cl K—Cl Cl—Si
Molecular Polarity How can you tell if a molecule is polar?
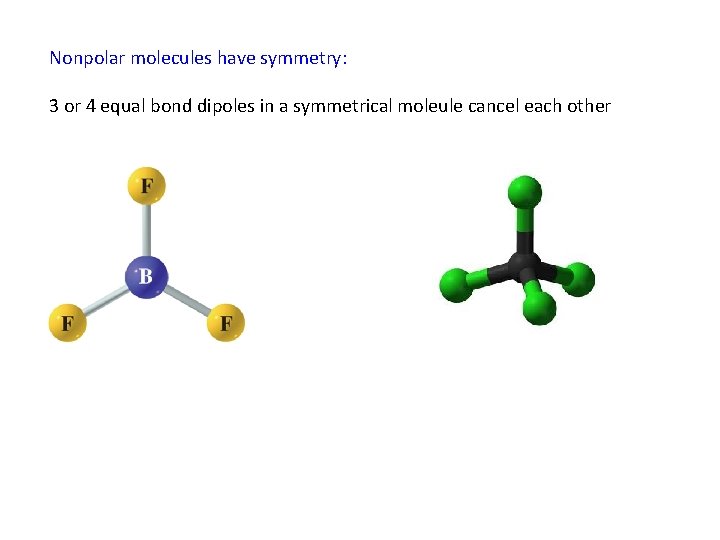
Nonpolar molecules have symmetry: 3 or 4 equal bond dipoles in a symmetrical moleule cancel each other
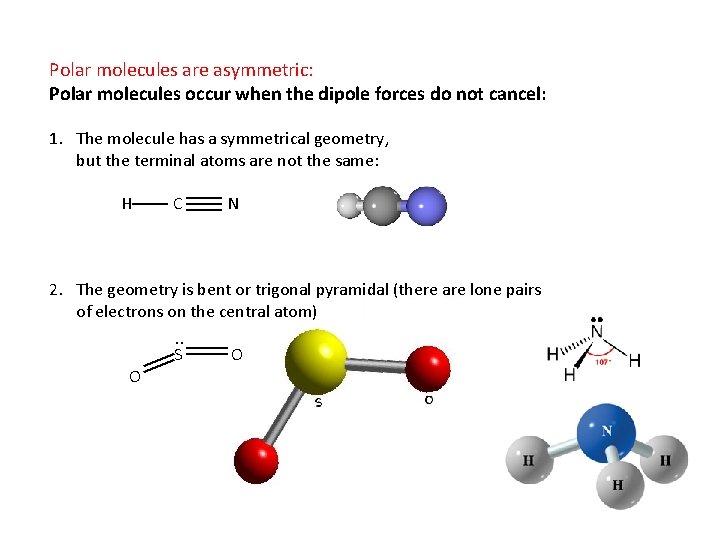
Polar molecules are asymmetric: Polar molecules occur when the dipole forces do not cancel: 1. The molecule has a symmetrical geometry, but the terminal atoms are not the same: H C N 2. The geometry is bent or trigonal pyramidal (there are lone pairs of electrons on the central atom). . S O O
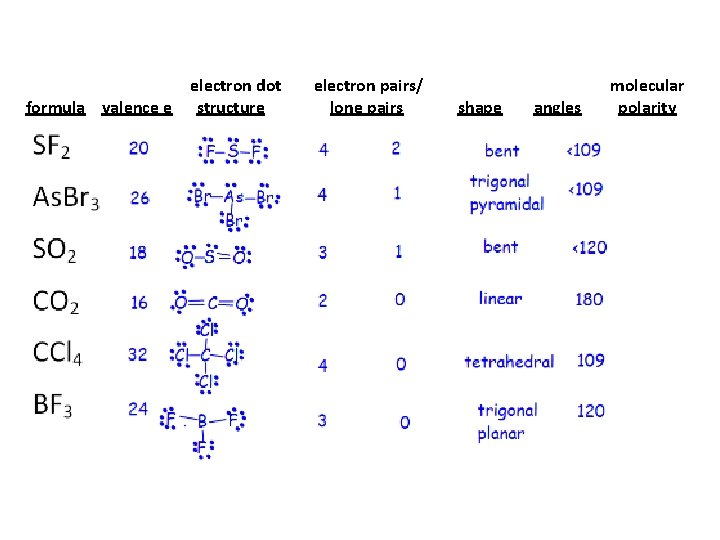
formula valence e electron dot structure electron pairs/ lone pairs shape angles molecular polarity

Attractive Forces in Liquids & Solids or Why is polarity important? 1. 2. 3. 4. Solid & Liquid particles have kinetic energy. The kinetic energy of all particles is related to the Absolute Temperature. Particles in condensed phases are close together. Particles in condensed phases are attracted to each other.
- Slides: 26