CHAPTER 5 Bonding Theories Explaining Molecular Geometry Chapter
CHAPTER 5: Bonding Theories Explaining Molecular Geometry

Chapter Outline § 5. 1 Molecular Shape § 5. 2 Valence-Shell Electron-Pair Repulsion Theory (VSEPR) § 5. 3 Polar Bonds and Polar Molecules » What Makes a Molecule Polar? » Dipole Moments § § 5. 4 5. 5 5. 6 5. 7 Valence Bond Theory Shape and Interactions with Large Molecules Chirality and Molecular Recognition Molecular Orbital Theory

Bar Code Reader App (IOS) QR Code Reader and Scanner 4+ Shop. Savvy, Inc. https: //tinyurl. com/clj 7 f 7 u

VSEPR Molecular Shapes Website https: //phet. Colorado. edu https: //tinyurl. com/pcwl 97 v

Predict the geometry of the molecule from the negative repulsions between the electron pairs (both bonding and nonbonding lone pairs). Linear 180 o Trigonal planar 120 o Tetrahedral 109. 5 o Trigonal Bipyramidal 120 and 90 o Octahedral 90 o
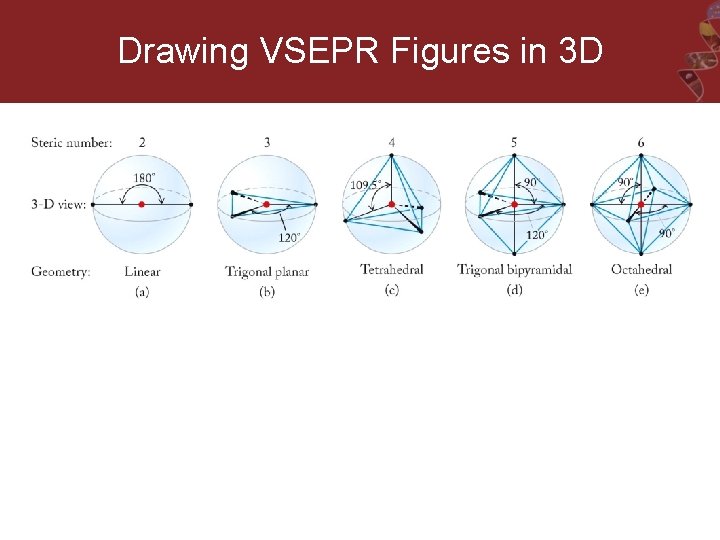
Drawing VSEPR Figures in 3 D

Steric Number (SN) = Number of atoms bonded to the central atom + Number of lone pairs bonded to central atom Used to select the correct VSEPR geometry https: //tinyurl. com/pcwl 97 v

Central Atoms with No Lone Pairs Electron Group Geometry = Molecular Geometry SN 2 = Linear Bond Angle = 180 o
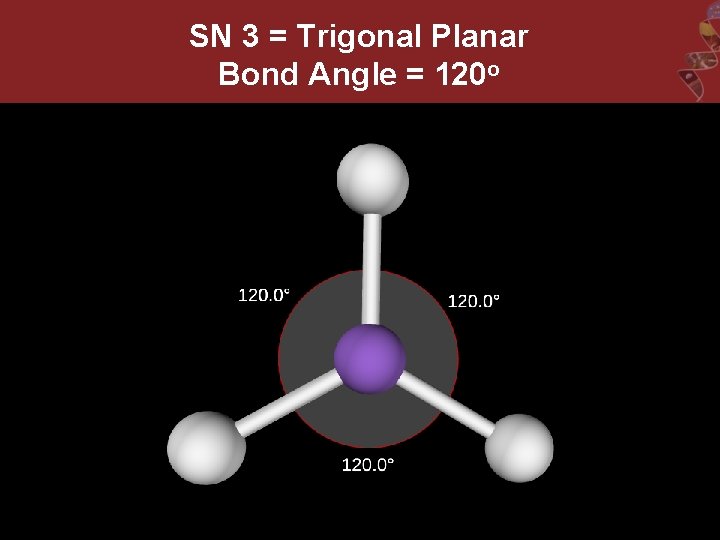
SN 3 = Trigonal Planar Bond Angle = 120 o
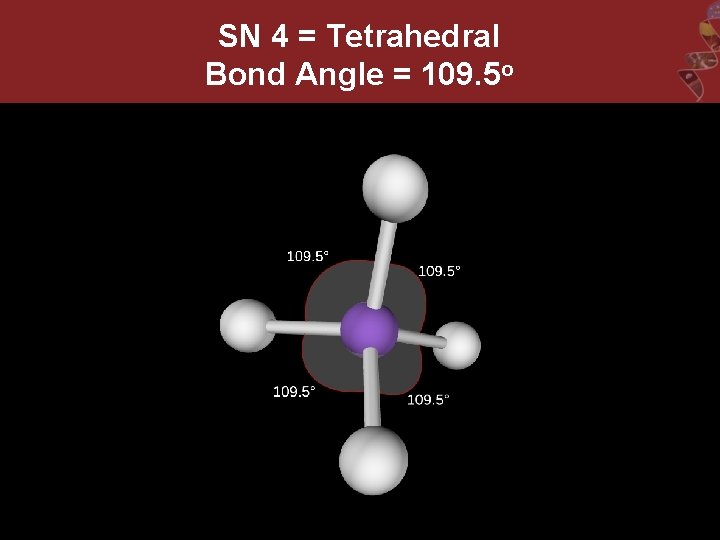
SN 4 = Tetrahedral Bond Angle = 109. 5 o
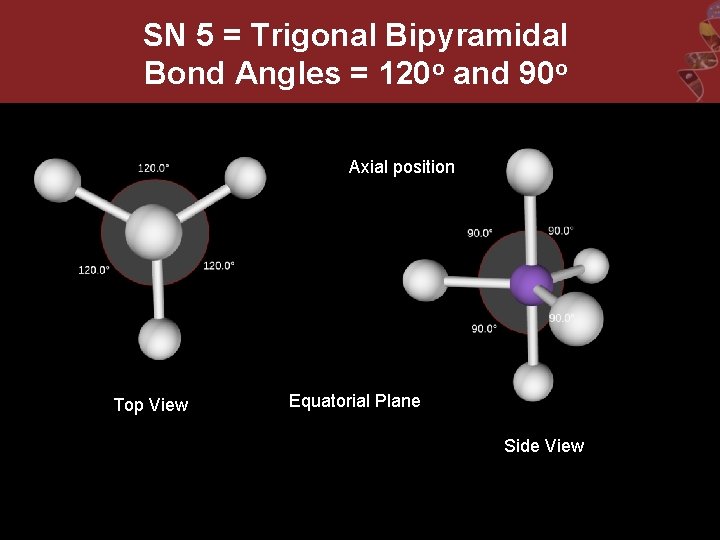
SN 5 = Trigonal Bipyramidal Bond Angles = 120 o and 90 o Axial position Top View Equatorial Plane Side View
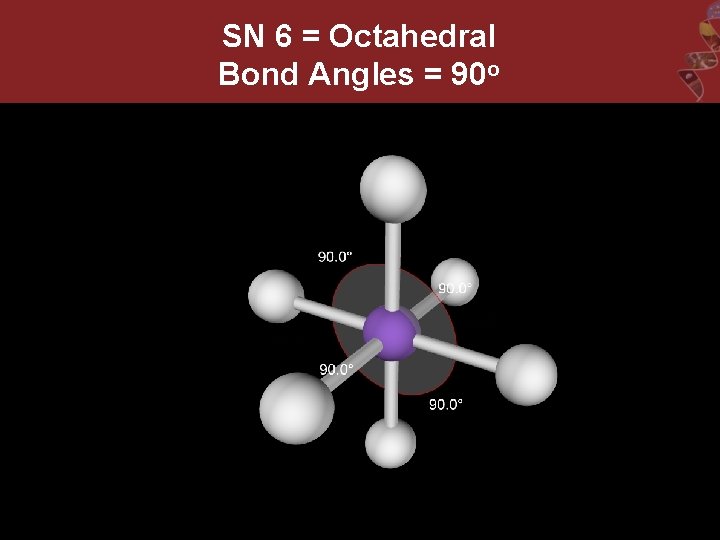
SN 6 = Octahedral Bond Angles = 90 o

Applying VSEPR Theory 1. Determine the Lewis structure 2. Determine the Steric Number around the CENTRAL ATOM 3. Multiple bonds count as one electron group 4. Find out the appropriate VSEPR geometry for the specified number of electron groups 5. Use the positions of atoms to establish the resulting molecular geometry.

Central Atoms with No Lone Pairs Example I: Carbon Dioxide, CO 2
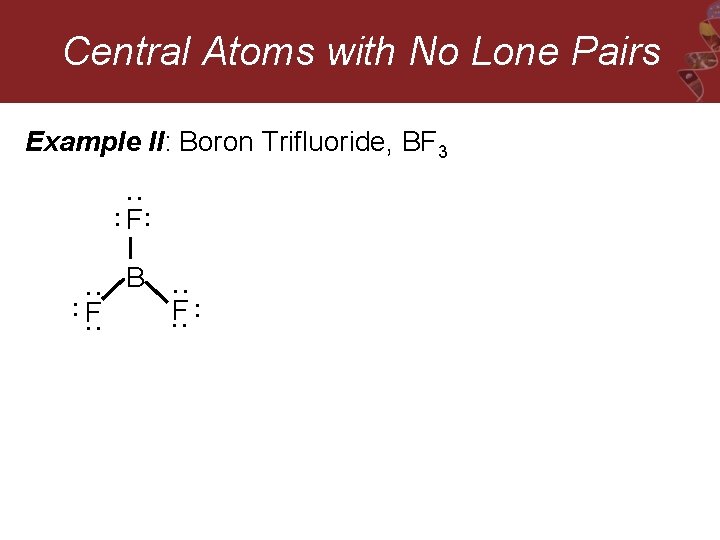
Central Atoms with No Lone Pairs Example II: Boron Trifluoride, BF 3 : : : F : : F B : : : F:
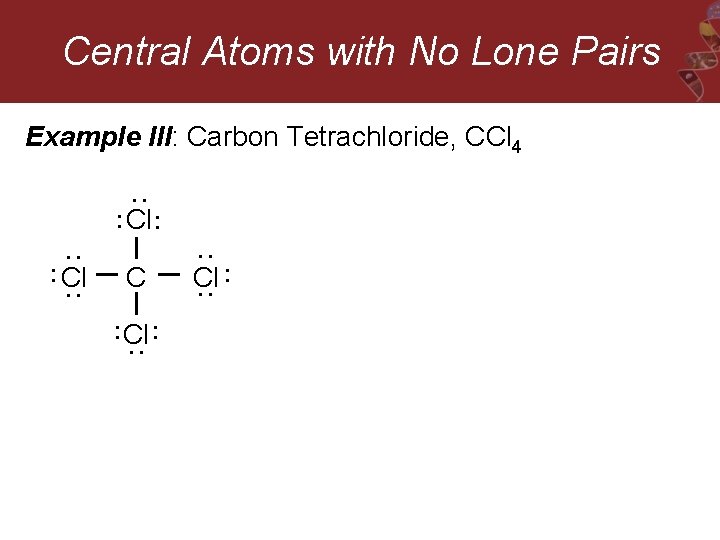
Central Atoms with No Lone Pairs Example III: Carbon Tetrachloride, CCl 4 : : Cl : : : Cl : C : : : Cl : :
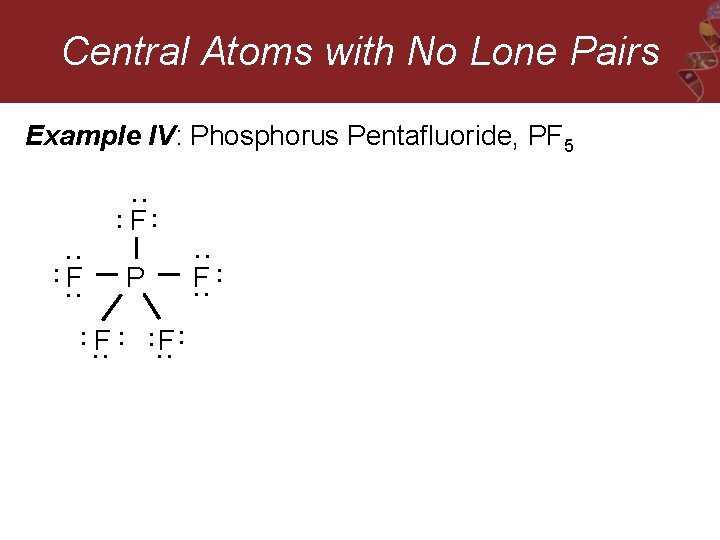
Central Atoms with No Lone Pairs Example IV: Phosphorus Pentafluoride, PF 5 : : F: F : : : : : F F : P : : : F :

Central Atoms with No Lone Pairs Example V: Sulfur hexafluoride, SF 6 : : F : F: : F F: : S : : : F : F:

Sample Exercise 9. 1 –a double bond counts as one pair Formaldehyde, CH 2 O, is a gas at room temperature and is an ingredient in solutions used to preserve biological samples. Use VSEPR to predict the molecular geometry of formaldehyde.
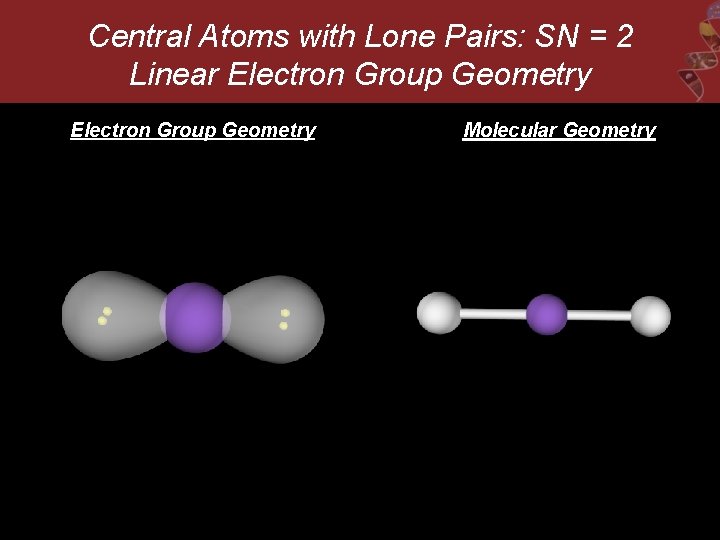
Central Atoms with Lone Pairs: SN = 2 Linear Electron Group Geometry Molecular Geometry

Central Atoms with Lone Pairs: SN = 2 Linear Electron Group Geometry e. g. HCN H C N:
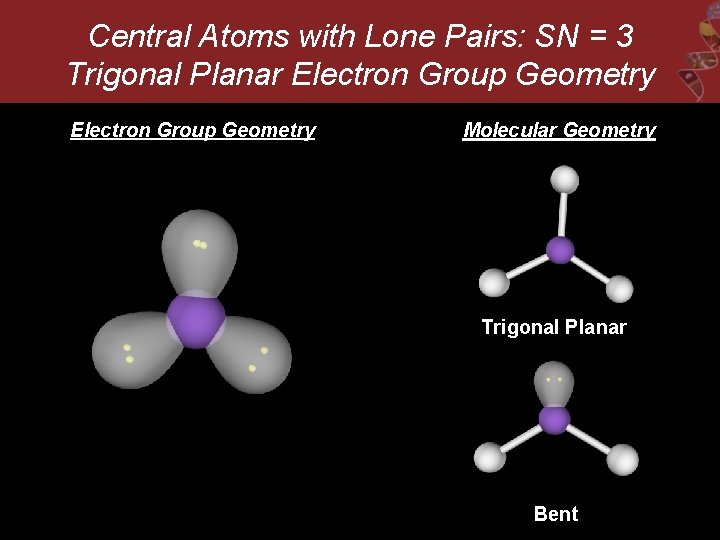
Central Atoms with Lone Pairs: SN = 3 Trigonal Planar Electron Group Geometry Molecular Geometry Trigonal Planar Bent
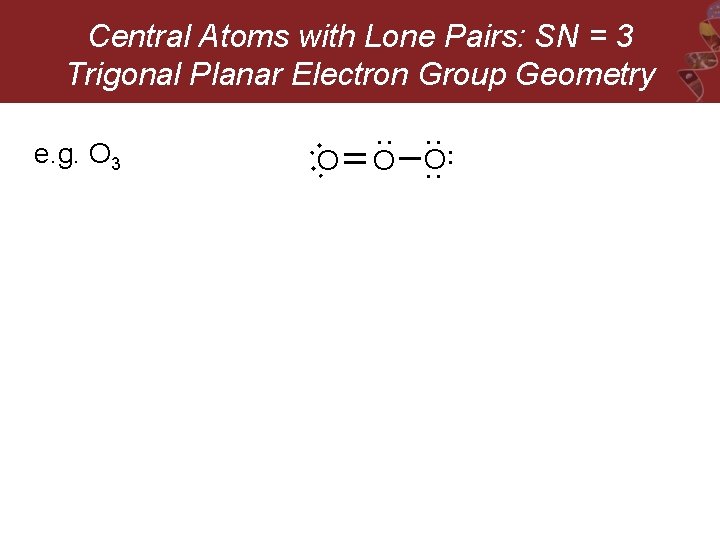
O: : O : e. g. O 3 : Central Atoms with Lone Pairs: SN = 3 Trigonal Planar Electron Group Geometry

Electron Pair Repulsions § Electron Pair Repulsion Order: • Lone pair—Lone pair = greatest repulsion. • Lone pair—Bonding pair is next. • Bonding pair—Bonding pair = least repulsion. » Double bonds exert more repulsion than single bonds. § Bond angles around central atom decrease as repulsive forces increase.
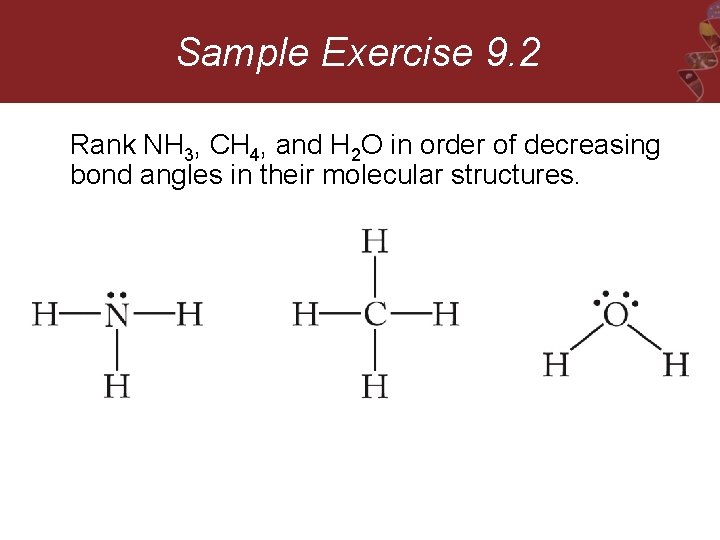
Sample Exercise 9. 2 Rank NH 3, CH 4, and H 2 O in order of decreasing bond angles in their molecular structures.
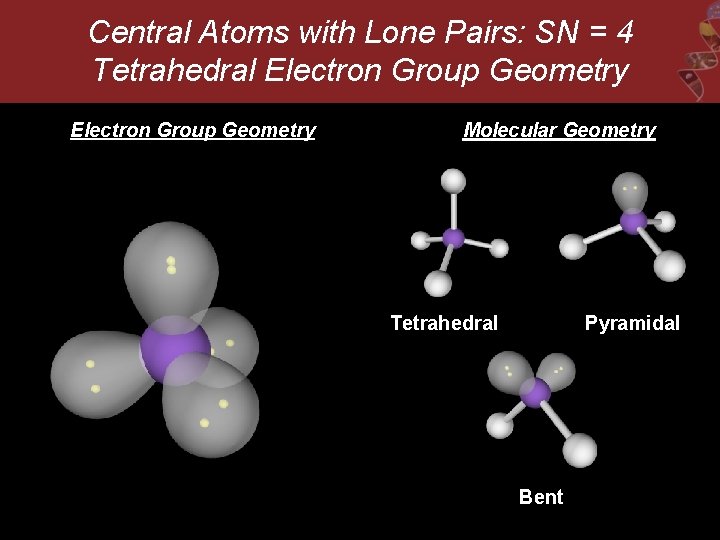
Central Atoms with Lone Pairs: SN = 4 Tetrahedral Electron Group Geometry Molecular Geometry Tetrahedral Pyramidal Bent
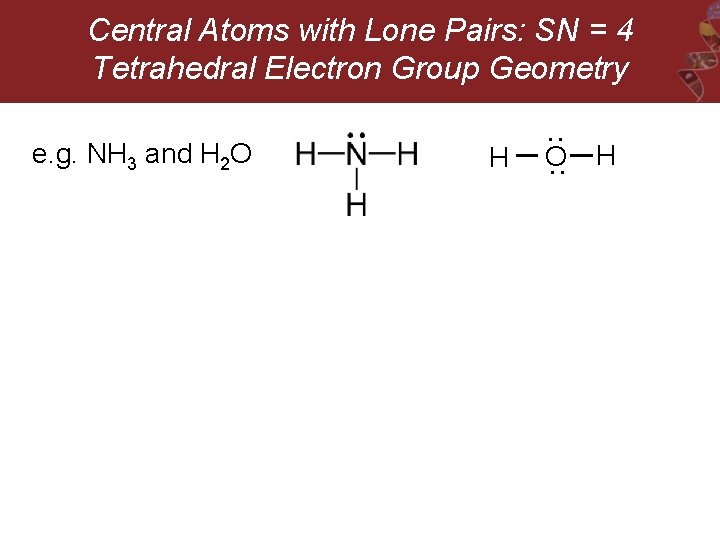
Central Atoms with Lone Pairs: SN = 4 Tetrahedral Electron Group Geometry O : H : e. g. NH 3 and H 2 O H
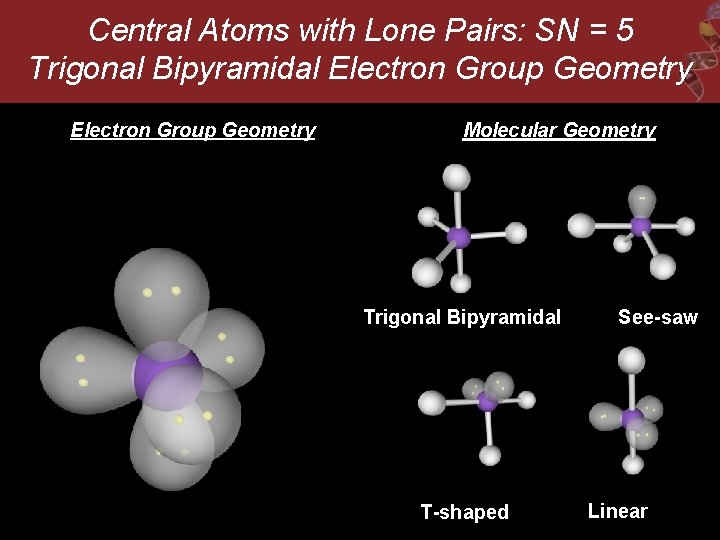
Central Atoms with Lone Pairs: SN = 5 Trigonal Bipyramidal Electron Group Geometry Molecular Geometry Trigonal Bipyramidal T-shaped See-saw Linear
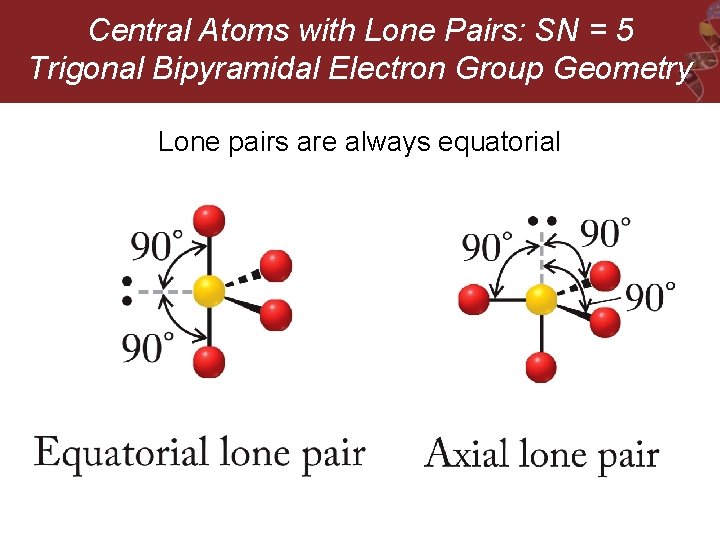
Central Atoms with Lone Pairs: SN = 5 Trigonal Bipyramidal Electron Group Geometry Lone pairs are always equatorial
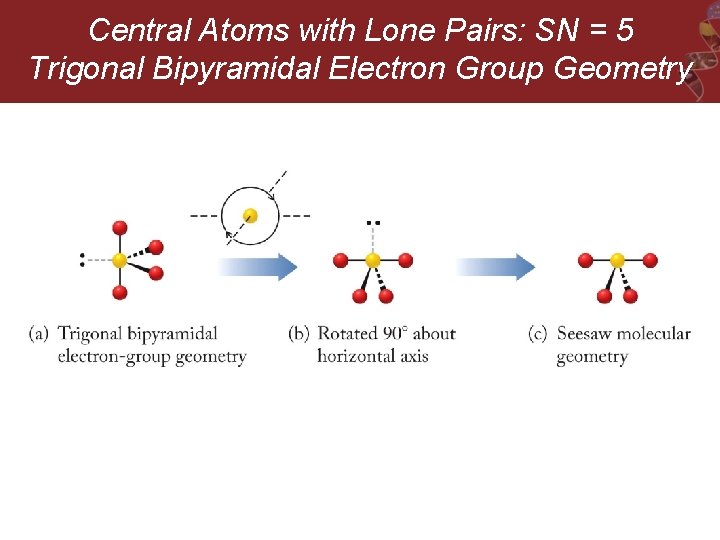
Central Atoms with Lone Pairs: SN = 5 Trigonal Bipyramidal Electron Group Geometry
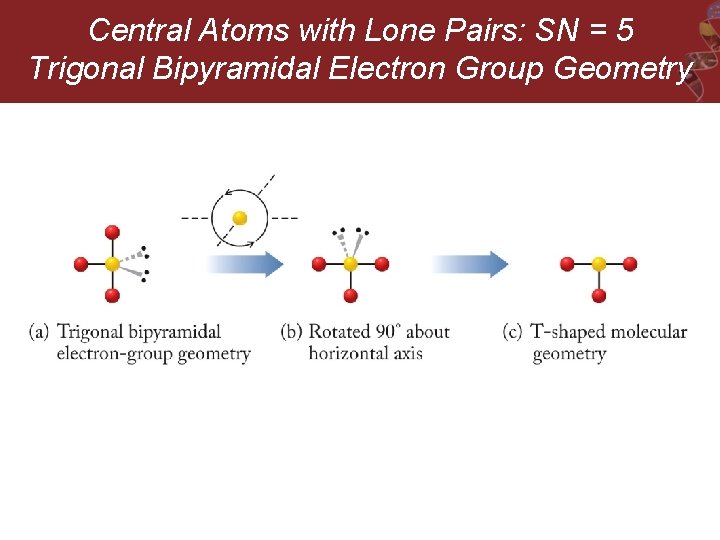
Central Atoms with Lone Pairs: SN = 5 Trigonal Bipyramidal Electron Group Geometry
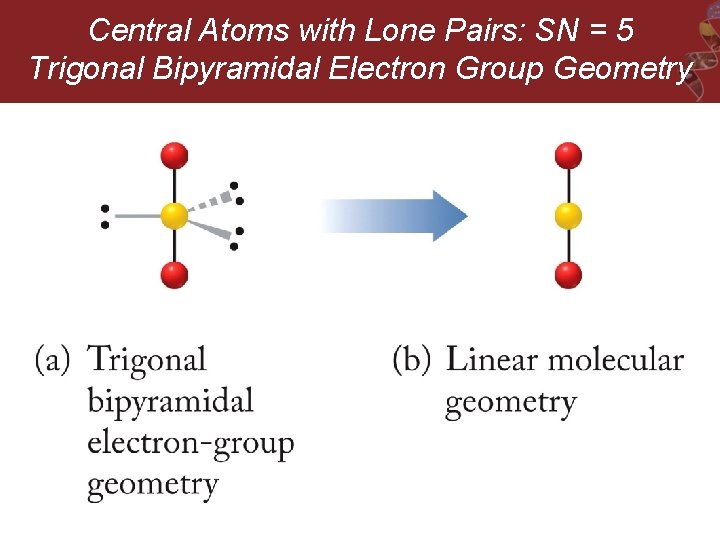
Central Atoms with Lone Pairs: SN = 5 Trigonal Bipyramidal Electron Group Geometry

Central Atoms with Lone Pairs: SN = 5 Trigonal Bipyramidal Electron Group Geometry e. g. SF 4, Cl. F 3, and I 3 -
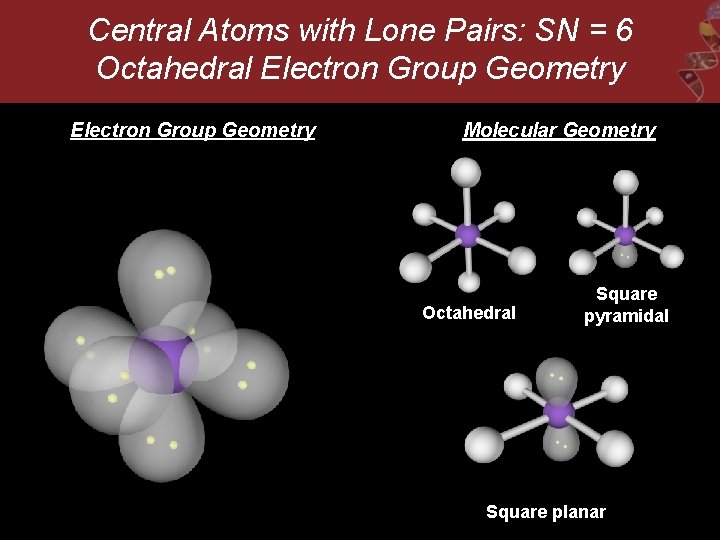
Central Atoms with Lone Pairs: SN = 6 Octahedral Electron Group Geometry Molecular Geometry Octahedral Square pyramidal Square planar
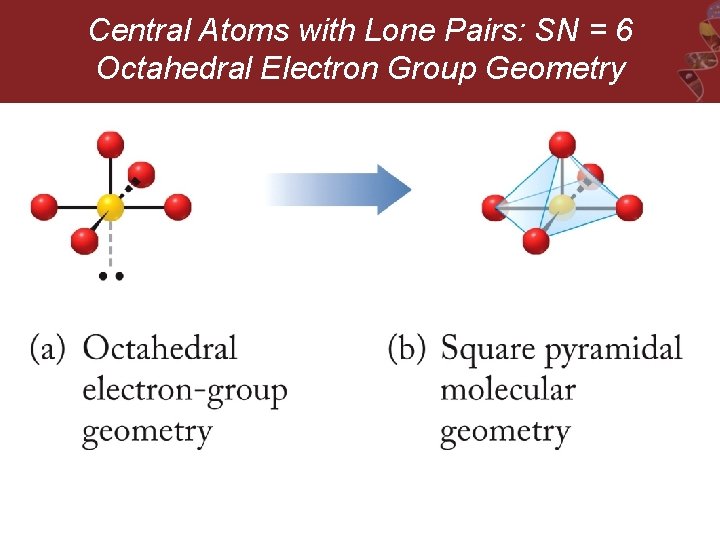
Central Atoms with Lone Pairs: SN = 6 Octahedral Electron Group Geometry
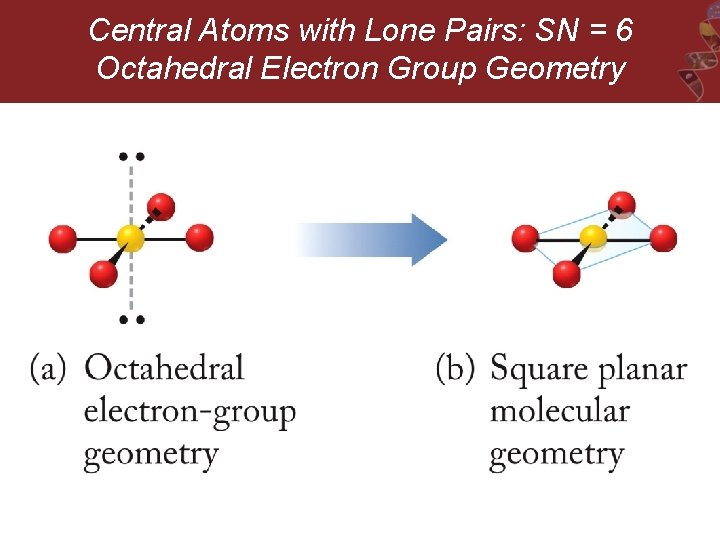
Central Atoms with Lone Pairs: SN = 6 Octahedral Electron Group Geometry

Central Atoms with Lone Pairs: SN = 6 Octahedral Electron Group Geometry e. g. SF 6, Br. F 5, and Xe. F 4

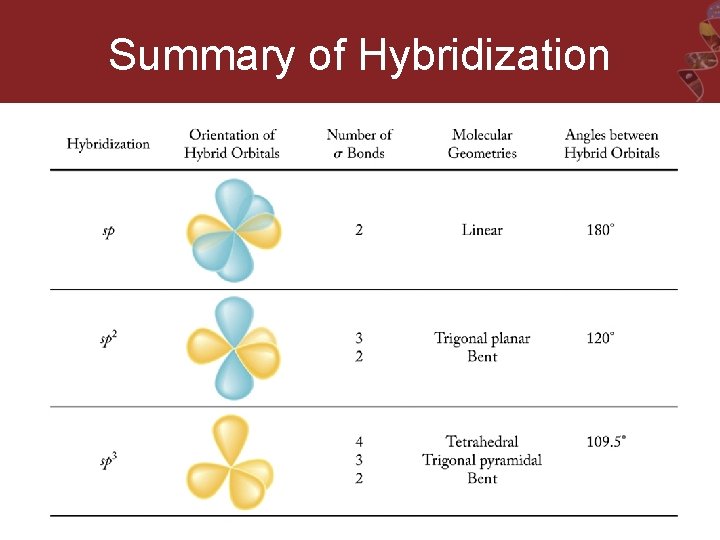
Summary of all Geometries (with and without lone pairs)

Chapter Outline § 5. 1 Molecular Shape § 5. 2 Valence-Shell Electron-Pair Repulsion Theory (VSEPR) § 5. 3 Polar Bonds and Polar Molecules » What Makes a Molecule Polar? » Dipole Moments § § 5. 4 5. 5 5. 6 5. 7 Valence Bond Theory Shape and Interactions with Large Molecules Chirality and Molecular Recognition Molecular Orbital Theory

Polar Bonds and Polar Molecules § Requirements for Polar Molecule: • 1. Molecule must contain polar bonds (i. e. , covalent bond between atoms with ΔEN). • 2. Orientation of polar bonds results in charge separation from one part of the molecule to another. Polar bonds… but linear shape results in partial charges canceling out; nonpolar!
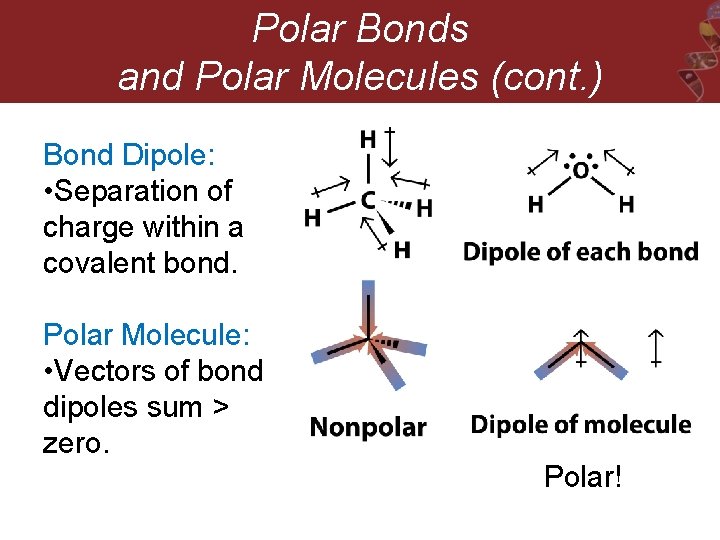
Polar Bonds and Polar Molecules (cont. ) Bond Dipole: • Separation of charge within a covalent bond. Polar Molecule: • Vectors of bond dipoles sum > zero. Polar!
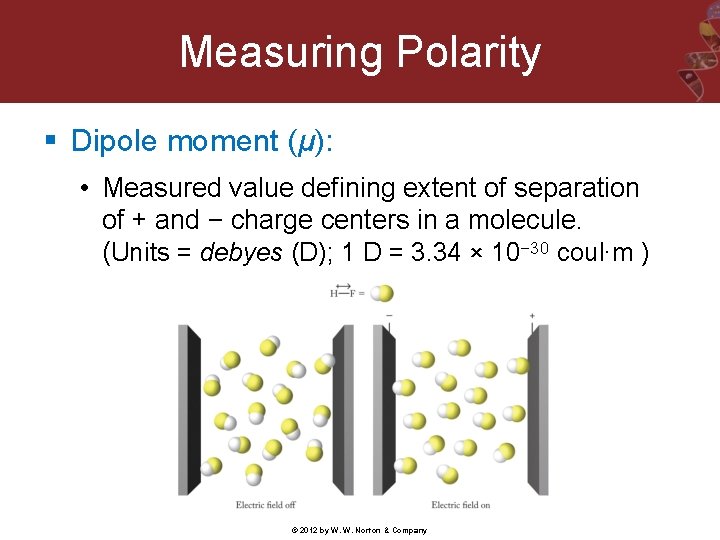
Measuring Polarity § Dipole moment (μ): • Measured value defining extent of separation of + and − charge centers in a molecule. (Units = debyes (D); 1 D = 3. 34 × 10− 30 coul∙m ) © 2012 by W. W. Norton & Company

Examples Determine whether or not the following molecules are polar - (a) CH 2 O (b) CHCl 3 (c) CCl 3 F
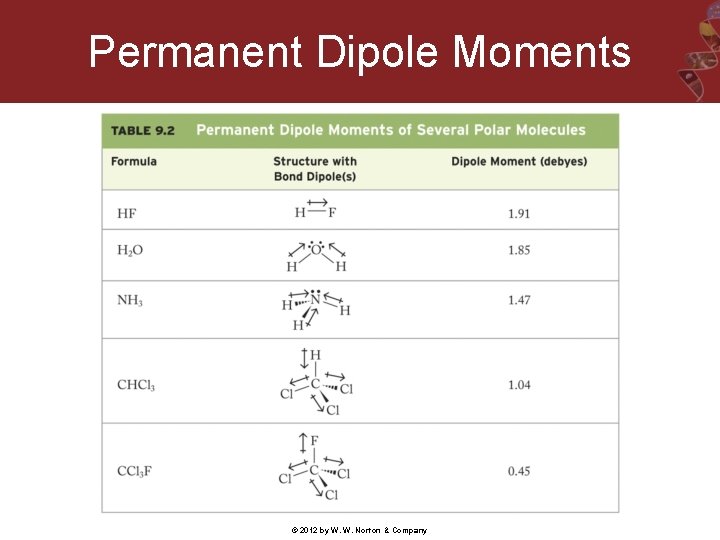
Permanent Dipole Moments © 2012 by W. W. Norton & Company

Chapter Outline § 5. 1 Molecular Shape § 5. 2 Valence-Shell Electron-Pair Repulsion Theory (VSEPR) § 5. 3 Polar Bonds and Polar Molecules § 5. 4 Valence Bond Theory » Orbital Overlap and Hybridization. » Hybridization and Molecular Geometries § 5. 5 Shape and Interactions with Large Molecules § 5. 6 Chirality and Molecular Recognition § 5. 7 Molecular Orbital Theory
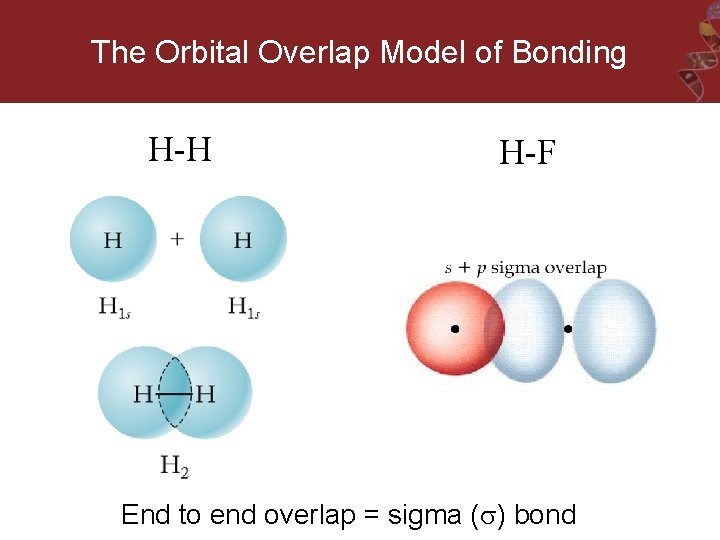
The Orbital Overlap Model of Bonding H-H H-F End to end overlap = sigma ( ) bond
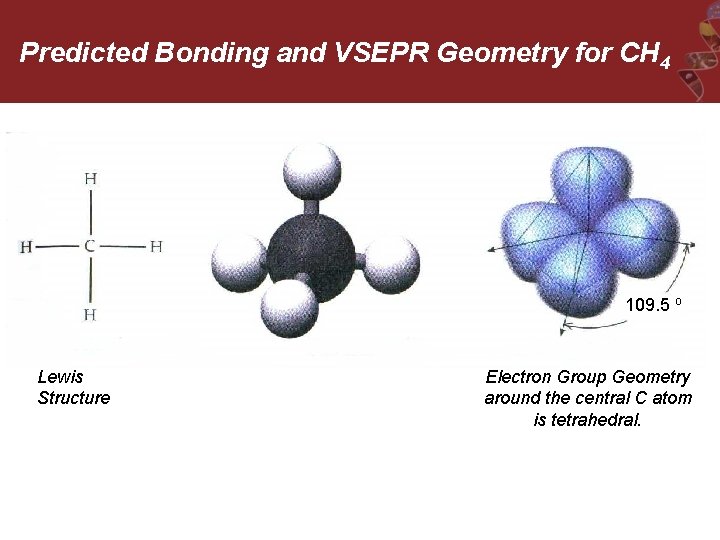
Predicted Bonding and VSEPR Geometry for CH 4 109. 5 o Lewis Structure Electron Group Geometry around the central C atom is tetrahedral.
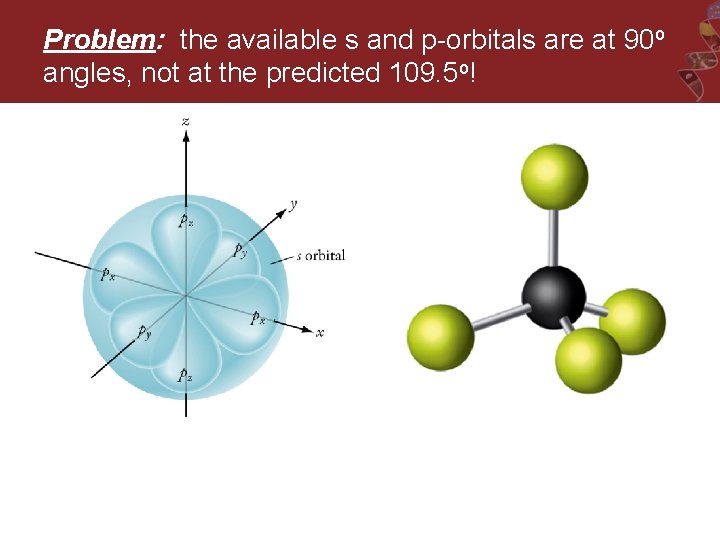
Problem: the available s and p-orbitals are at 90 o angles, not at the predicted 109. 5 o!
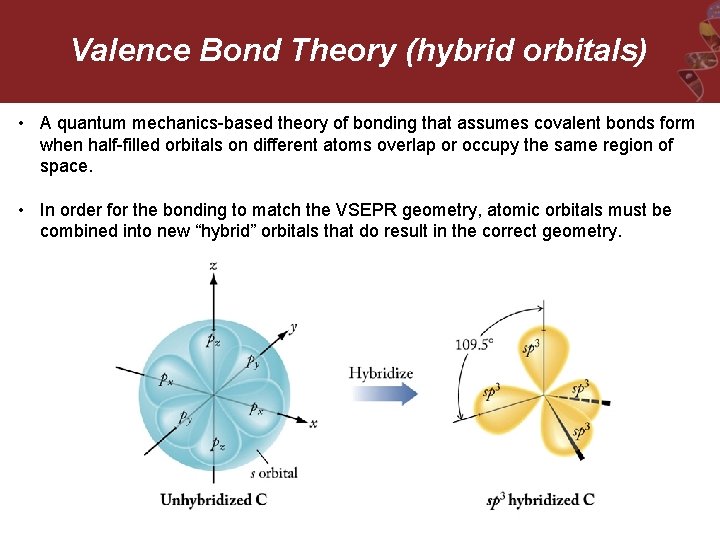
Valence Bond Theory (hybrid orbitals) • A quantum mechanics-based theory of bonding that assumes covalent bonds form when half-filled orbitals on different atoms overlap or occupy the same region of space. • In order for the bonding to match the VSEPR geometry, atomic orbitals must be combined into new “hybrid” orbitals that do result in the correct geometry.

Hybridization Rules – will be upgraded as we go New orbitals are constructed from pre-existing s, p, and d-orbitals = hybrid orbitals 1. Hybridize the CENTRAL ATOM ONLY (others as needed) 2. Only use valence shell electrons 3. The number of hybrid orbitals formed = number of atomic orbitals used
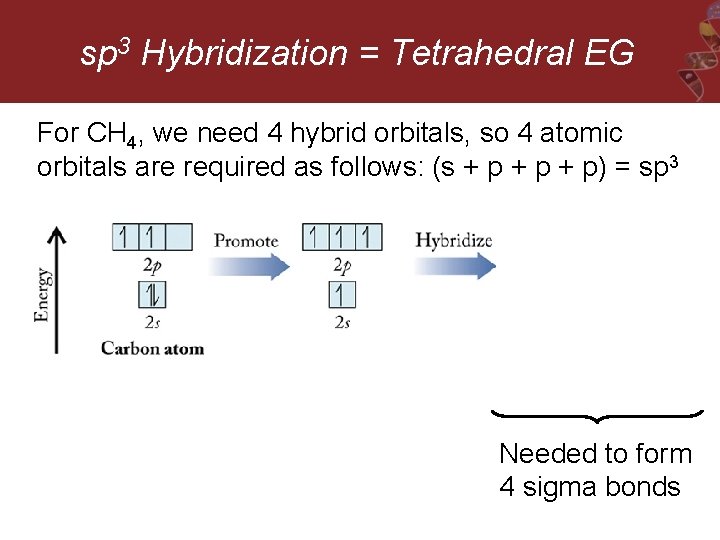
sp 3 Hybridization = Tetrahedral EG For CH 4, we need 4 hybrid orbitals, so 4 atomic orbitals are required as follows: (s + p + p) = sp 3 Needed to form 4 sigma bonds


Hybridization Rules – cont’d 1. Hybridize the CENTRAL ATOM ONLY (others as needed) 2. Only use valence shell electrons 3. The number of hybrid orbitals formed = number of atomic orbitals used 4. Hybrid orbitals get 1 electron for a -bond, 2 electrons for a lone pair.
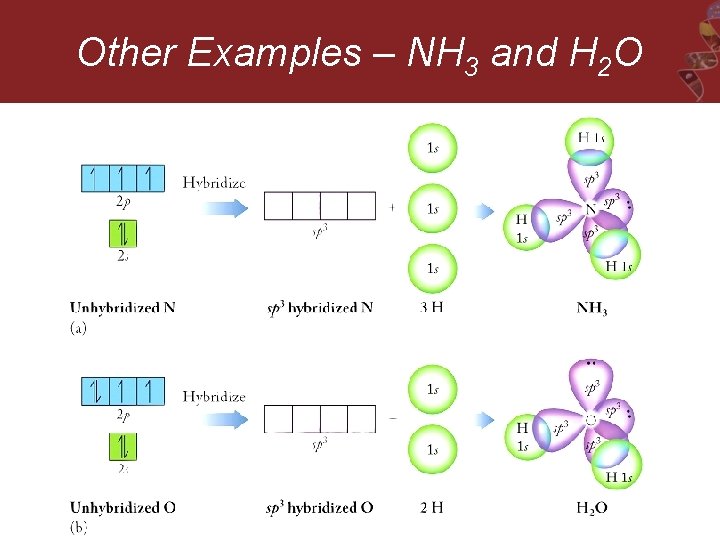
Other Examples – NH 3 and H 2 O
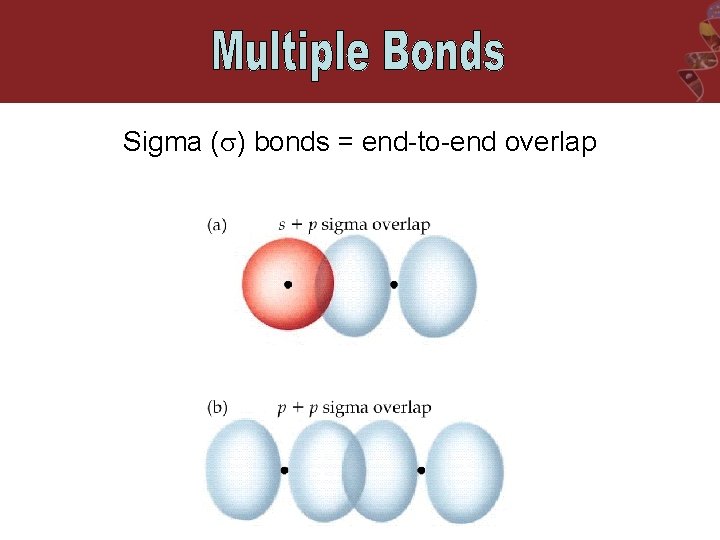
Sigma ( ) bonds = end-to-end overlap
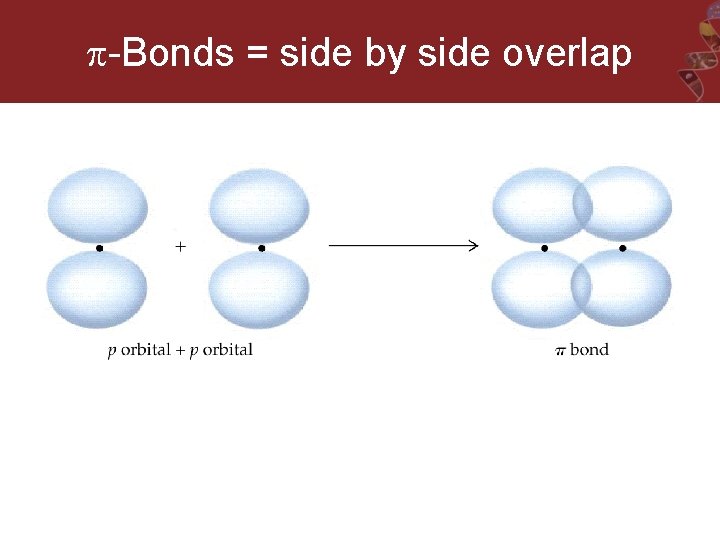
-Bonds = side by side overlap
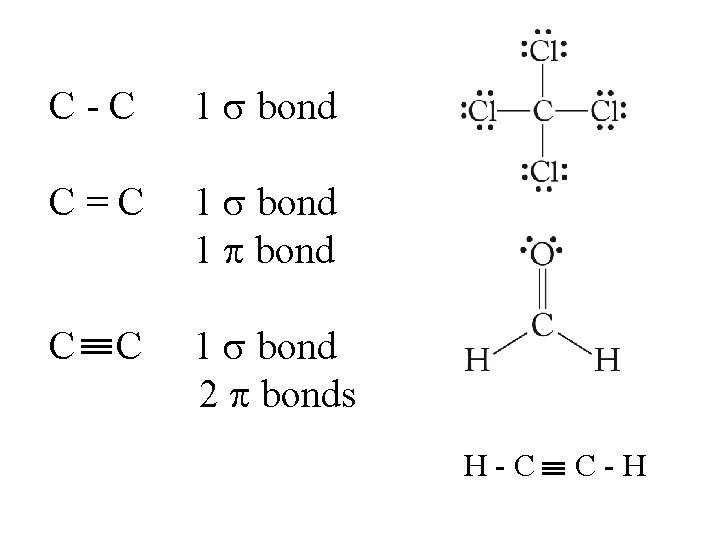
C-C 1 bond C=C 1 bond 2 bonds C H-C C-H

(upgraded – more will be added) 1. Hybrid orbitals get 1 electron for a -bond, 2 electrons for a lone pair. 2. Remaining electrons go into unhybridized orbitals = bonds
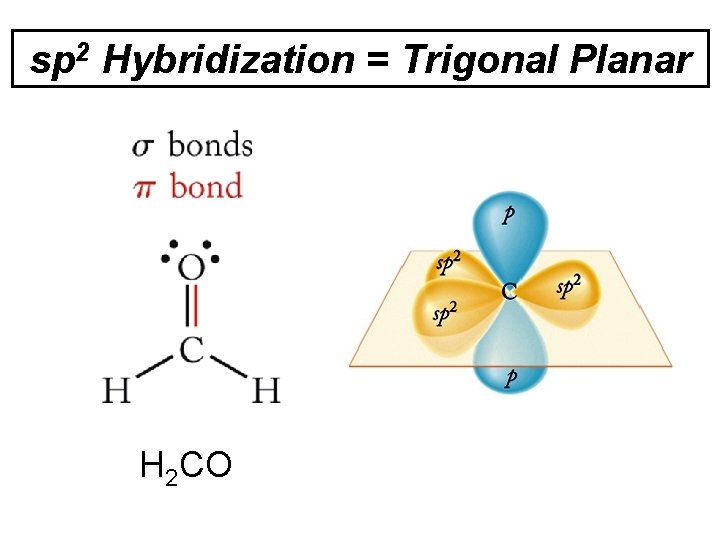
sp 2 Hybridization = Trigonal Planar H 2 CO



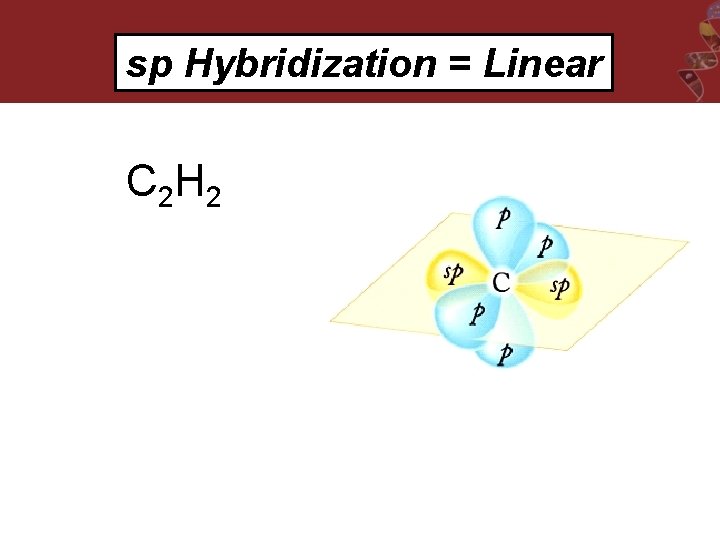
sp Hybridization = Linear C 2 H 2
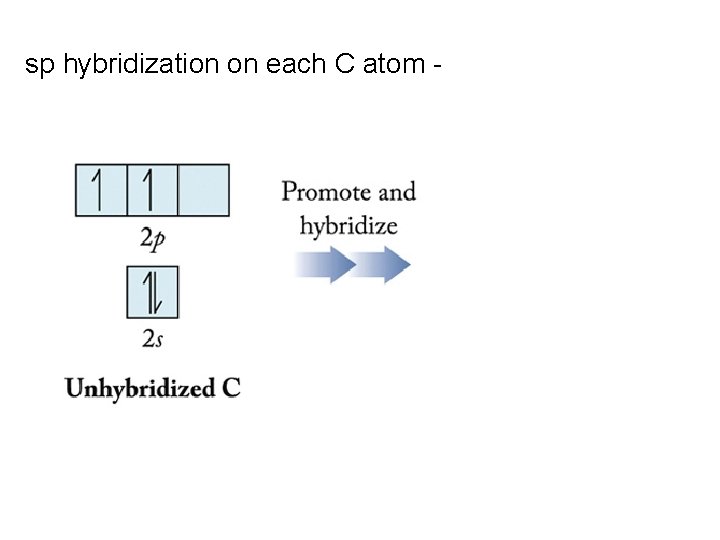
sp hybridization on each C atom -


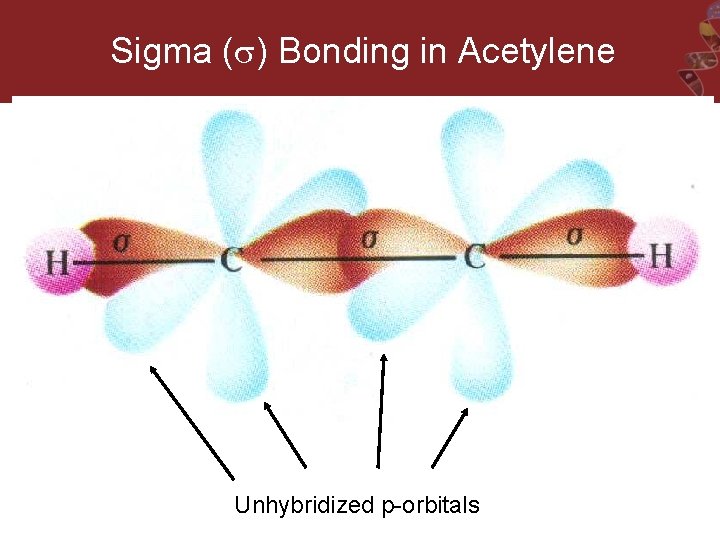
Sigma ( ) Bonding in Acetylene Unhybridized p-orbitals
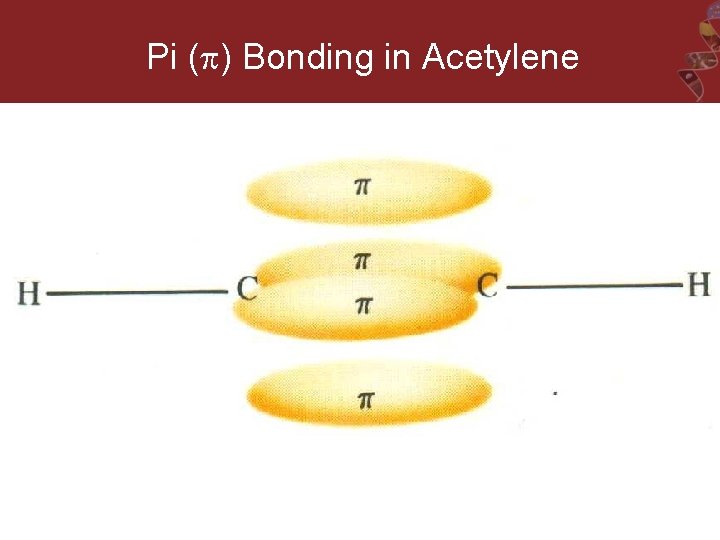
Pi ( ) Bonding in Acetylene

Sample Exercise 5. 5 Use Valence Bond Theory to explain the linear molecular geometry of CO 2. Determine the hybridization of the carbon and oxygen in this molecule, and describe the orbitals that overlap to form the bonds. solution:
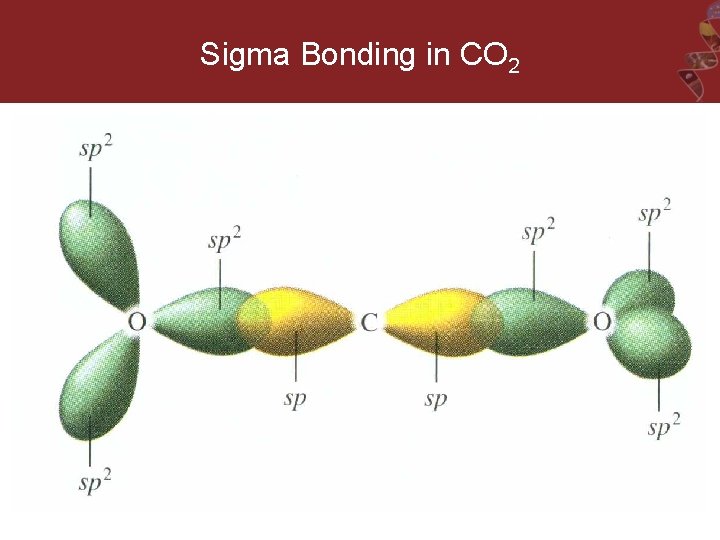
Sigma Bonding in CO 2
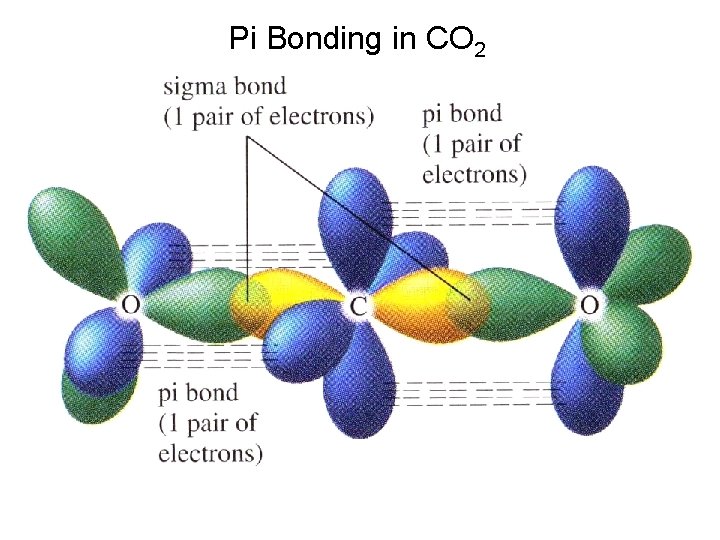
Pi Bonding in CO 2
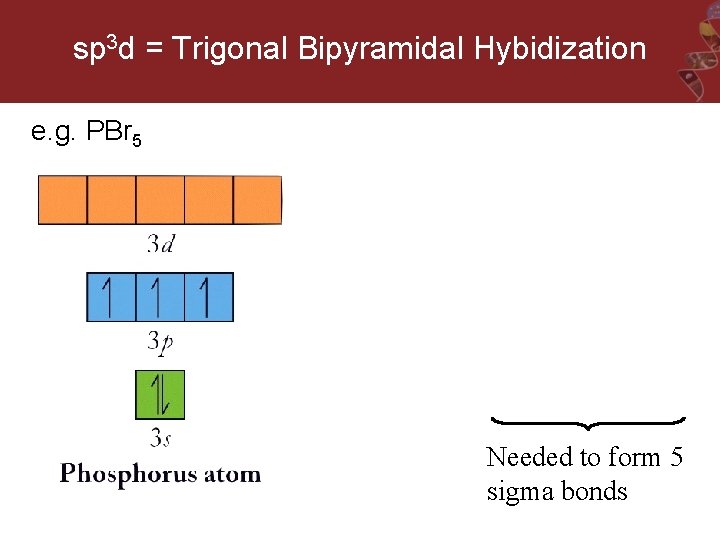
sp 3 d = Trigonal Bipyramidal Hybidization e. g. PBr 5 Needed to form 5 sigma bonds

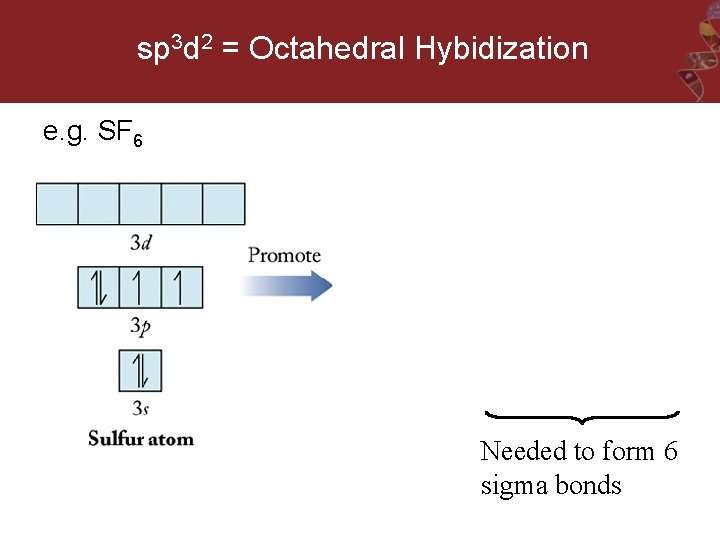
sp 3 d 2 = Octahedral Hybidization e. g. SF 6 Needed to form 6 sigma bonds

Summary of Hybridization


Chapter Outline § 5. 1 Molecular Shape § 5. 2 Valence-Shell Electron-Pair Repulsion Theory (VSEPR) § 5. 3 Polar Bonds and Polar Molecules § 5. 4 Valence Bond Theory § 5. 5 Molecules With Multiple Central Atoms § 5. 6 Chirality and Molecular Recognition § 5. 7 Molecular Orbital Theory
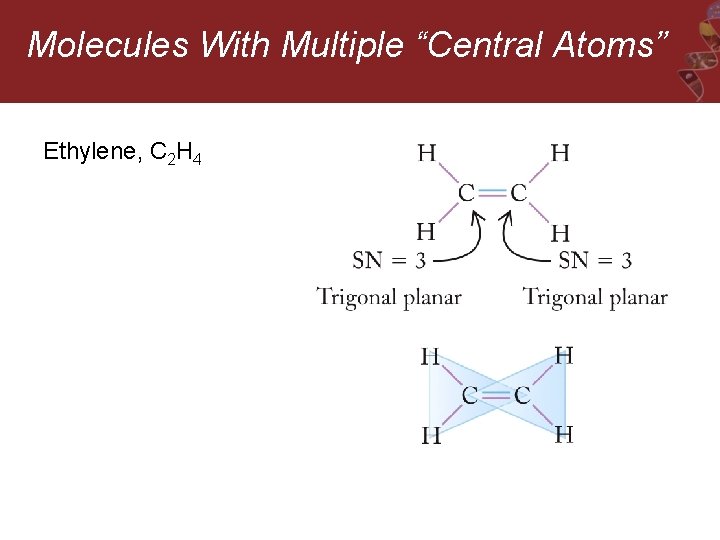
Molecules With Multiple “Central Atoms” Ethylene, C 2 H 4
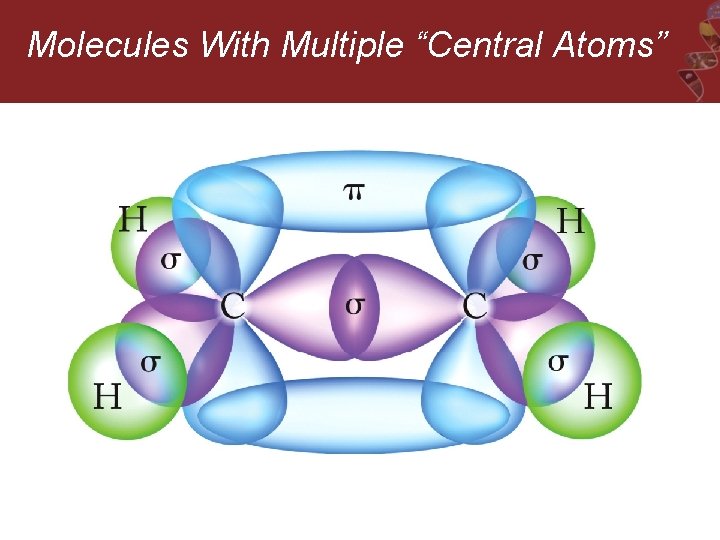
Molecules With Multiple “Central Atoms”
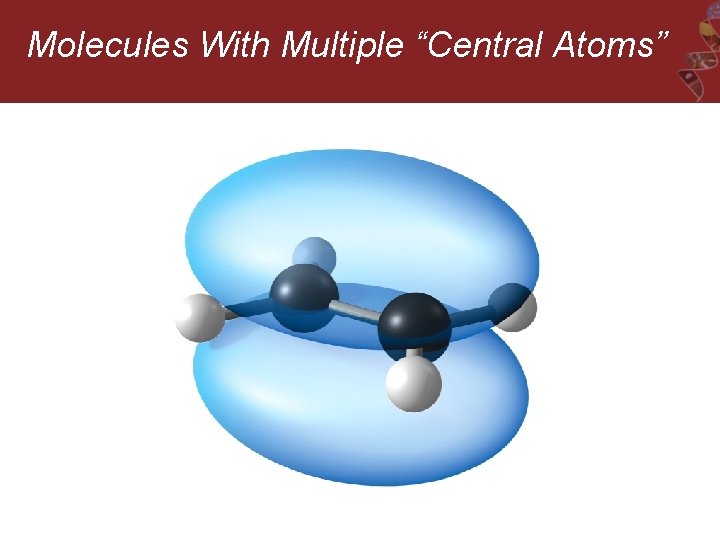
Molecules With Multiple “Central Atoms”
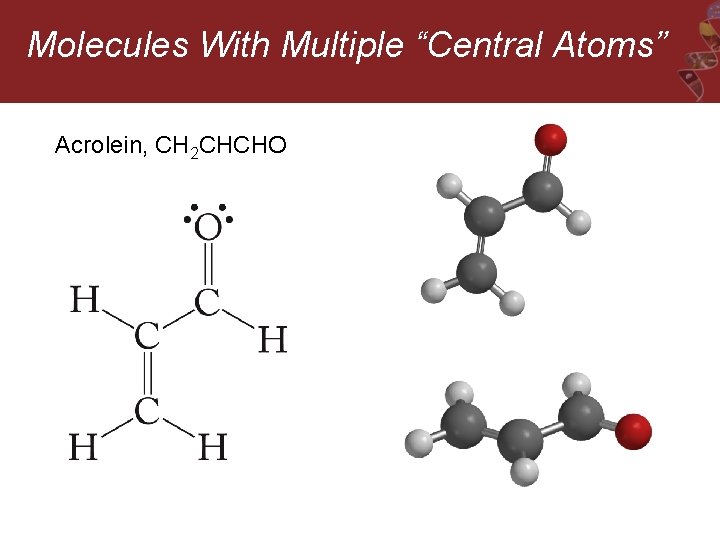
Molecules With Multiple “Central Atoms” Acrolein, CH 2 CHCHO
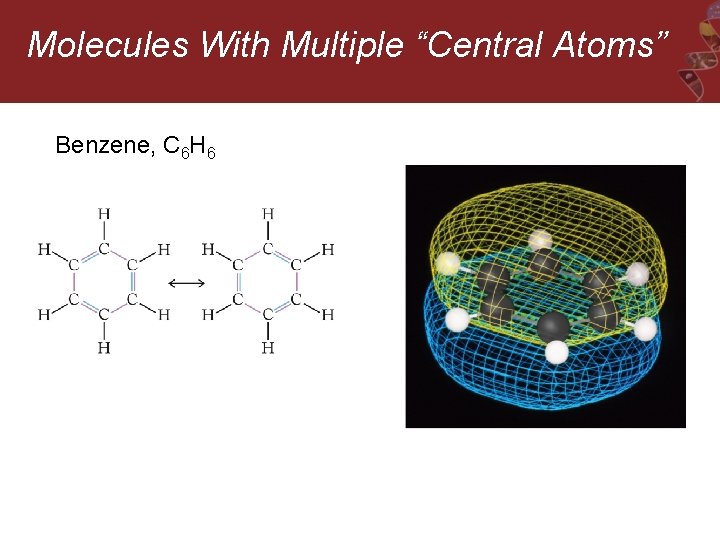
Molecules With Multiple “Central Atoms” Benzene, C 6 H 6


Chapter Outline § 5. 1 Molecular Shape § 5. 2 Valence-Shell Electron-Pair Repulsion Theory (VSEPR) § 5. 3 Polar Bonds and Polar Molecules § 5. 4 Valence Bond Theory § 5. 5 Molecules With Multiple Central Atoms § 5. 6 Chirality and Molecular Recognition § 5. 7 Molecular Orbital Theory
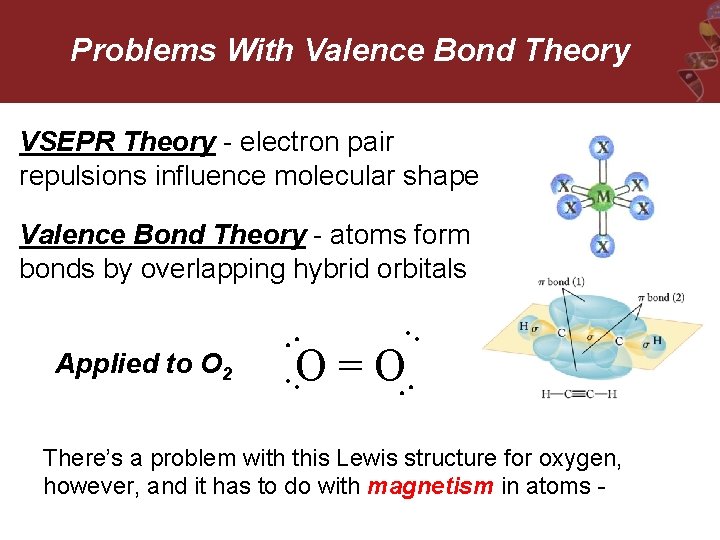
Problems With Valence Bond Theory VSEPR Theory - electron pair repulsions influence molecular shape Valence Bond Theory - atoms form bonds by overlapping hybrid orbitals Applied to O 2 • • O = O • • • • There’s a problem with this Lewis structure for oxygen, however, and it has to do with magnetism in atoms -
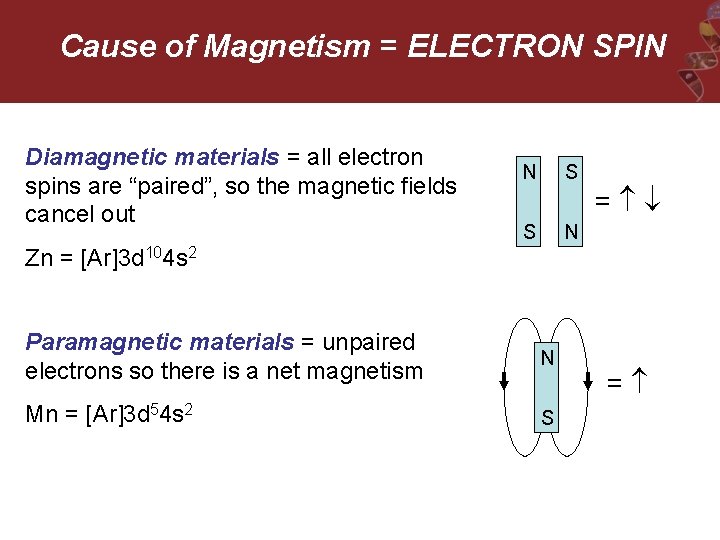
Cause of Magnetism = ELECTRON SPIN Diamagnetic materials = all electron spins are “paired”, so the magnetic fields cancel out N S S N = Zn = [Ar]3 d 104 s 2 Paramagnetic materials = unpaired electrons so there is a net magnetism N Mn = [Ar]3 d 54 s 2 S =

In the Lewis structure for oxygen, all of the electrons are paired up, so the molecule should be diamagnetic, but experiments prove that it is PARAMAGNETIC. An additional refinement in bonding theory is necessary =
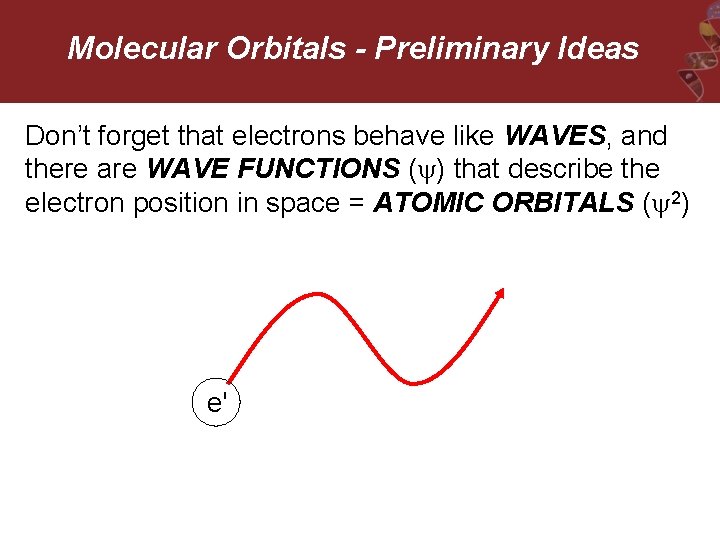
Molecular Orbitals - Preliminary Ideas Don’t forget that electrons behave like WAVES, and there are WAVE FUNCTIONS ( ) that describe the electron position in space = ATOMIC ORBITALS ( 2) e'
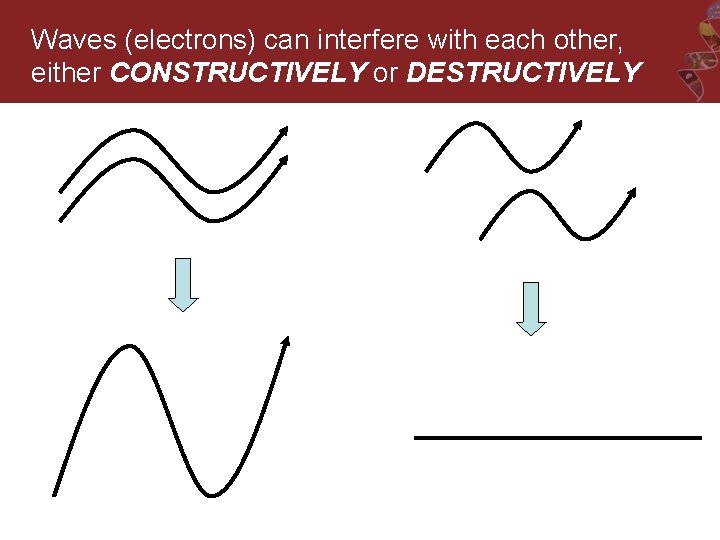
Waves (electrons) can interfere with each other, either CONSTRUCTIVELY or DESTRUCTIVELY
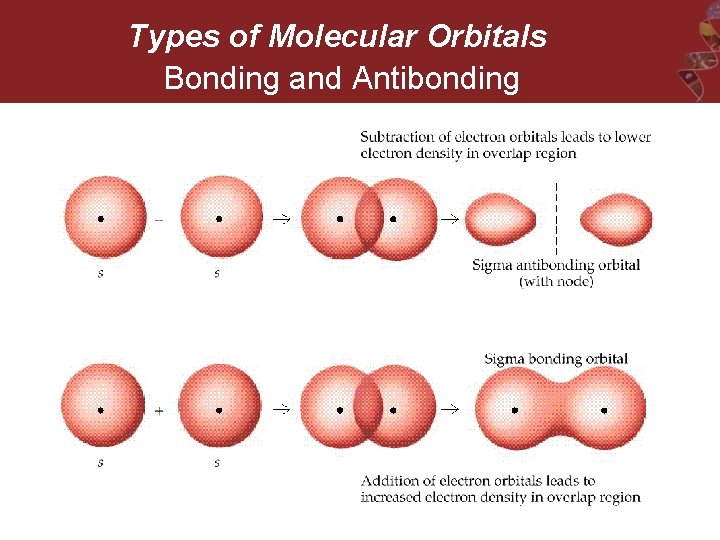
Types of Molecular Orbitals Bonding and Antibonding
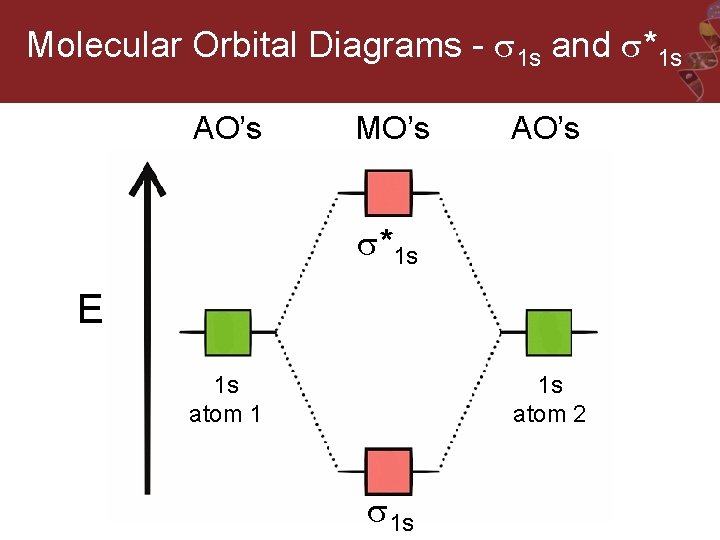
Molecular Orbital Diagrams - 1 s and *1 s AO’s MO’s AO’s *1 s E 1 s atom 1 1 s atom 2 1 s
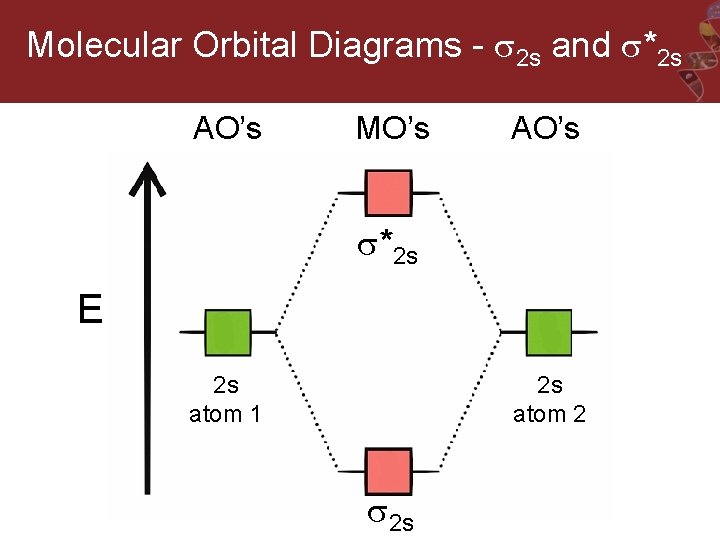
Molecular Orbital Diagrams - 2 s and *2 s AO’s MO’s AO’s *2 s E 2 s atom 1 2 s atom 2 2 s
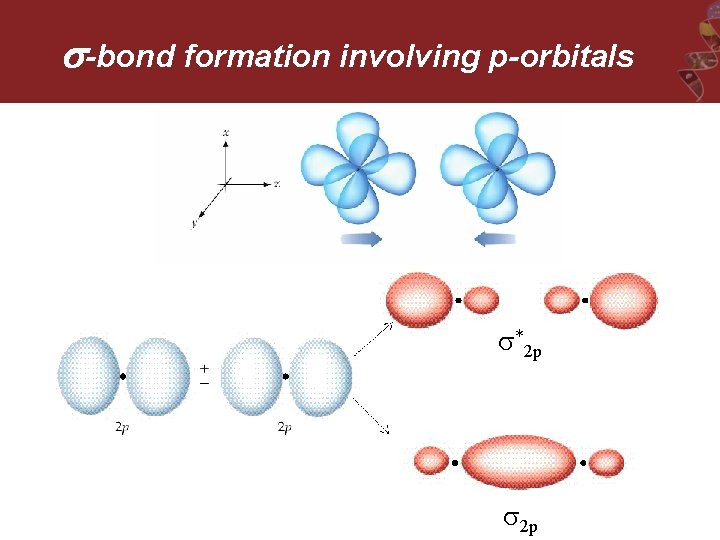
-bond formation involving p-orbitals *2 p 2 p
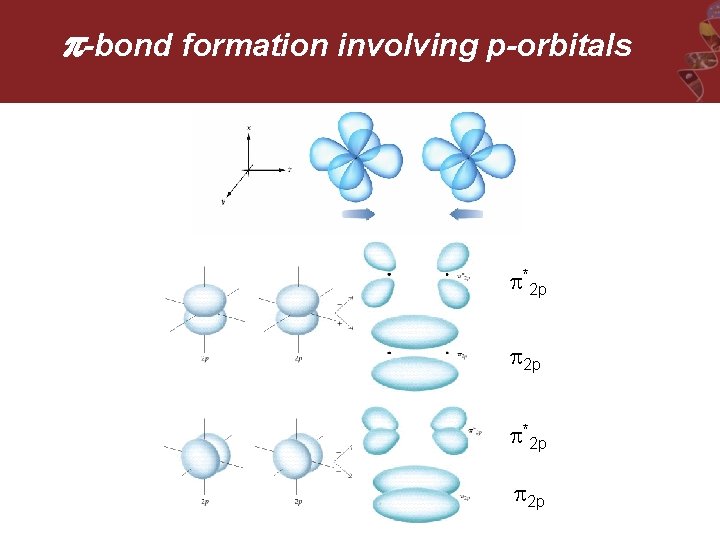
-bond formation involving p-orbitals *2 p 2 p



Applying Molecular Orbital Theory 1. The total number of molecular orbitals = total number of atomic orbitals contributed by the bonding atoms 2. Bonding MO’s are lower in energy (more stable) than anti-bonding MO’s 3. Electrons occupy molecular orbitals following the Pauli Exclusion Principle (spins pair up) and Hund’s Rule (remain unpaired as long as an empty orbital is available of the same energy)
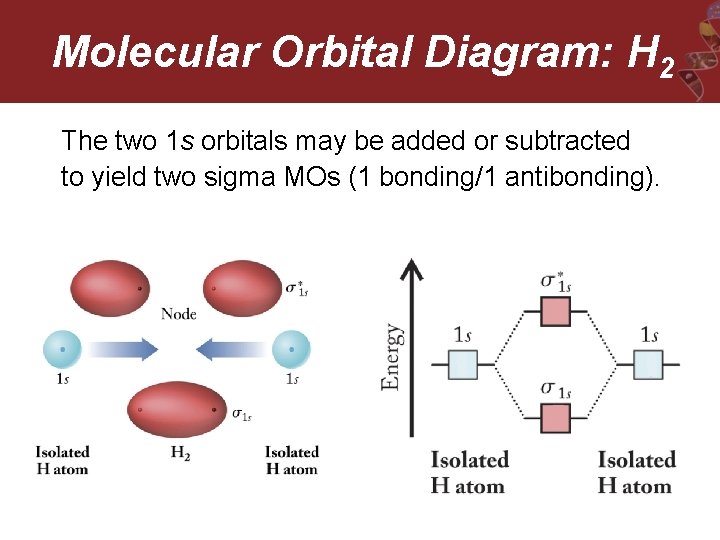
Molecular Orbital Diagram: H 2 The two 1 s orbitals may be added or subtracted to yield two sigma MOs (1 bonding/1 antibonding).
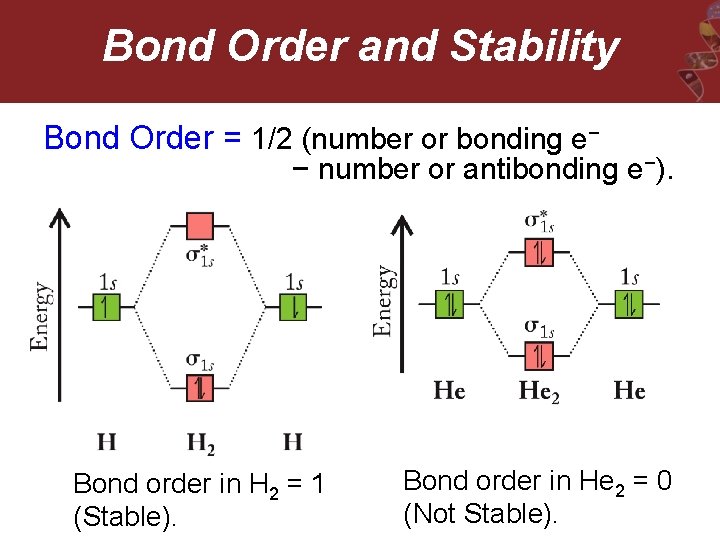
Bond Order and Stability Bond Order = 1/2 (number or bonding e− − number or antibonding e −). Bond order in H 2 = 1 (Stable). Bond order in He 2 = 0 (Not Stable).
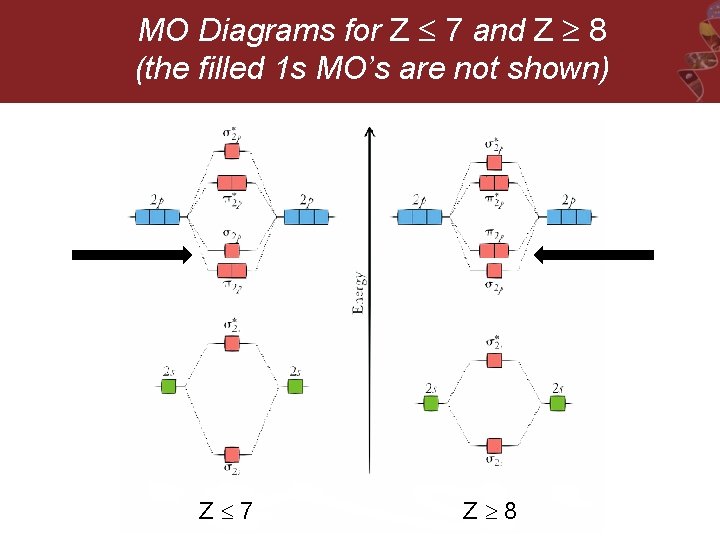
MO Diagrams for Z 7 and Z 8 (the filled 1 s MO’s are not shown) Z 7 Z 8
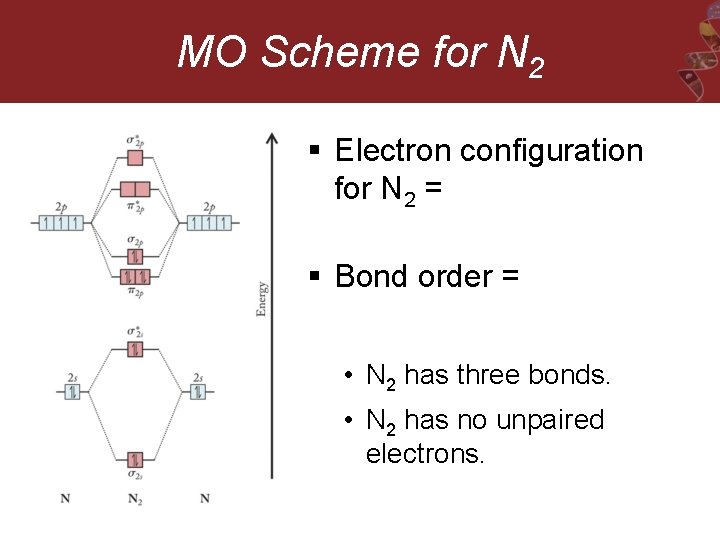
MO Scheme for N 2 § Electron configuration for N 2 = § Bond order = • N 2 has three bonds. • N 2 has no unpaired electrons.
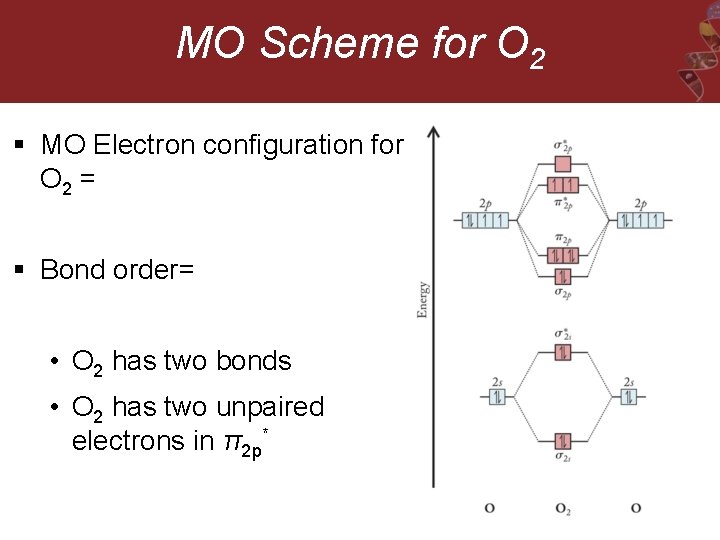
MO Scheme for O 2 § MO Electron configuration for O 2 = § Bond order= • O 2 has two bonds • O 2 has two unpaired electrons in π2 p*
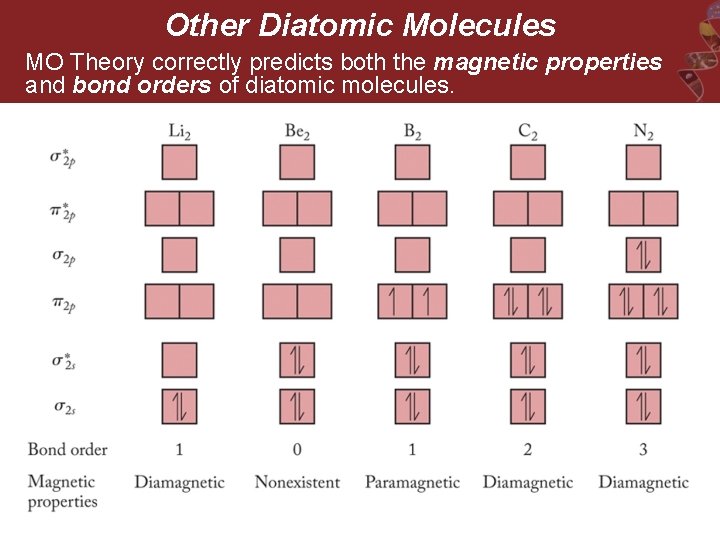
Other Diatomic Molecules MO Theory correctly predicts both the magnetic properties and bond orders of diatomic molecules.

Sample Exercise 5. 9 – Using MO theory for Charged Homonuclear Diatomics In which of the molecules in the proceeding diagram would there be an increase in bond order if they lost one electron each, forming singly charged diatomic cations (X 2+)?
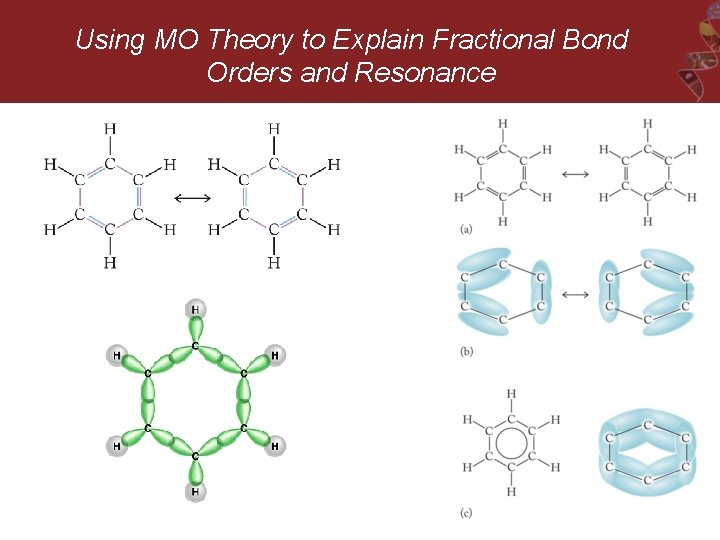
Using MO Theory to Explain Fractional Bond Orders and Resonance
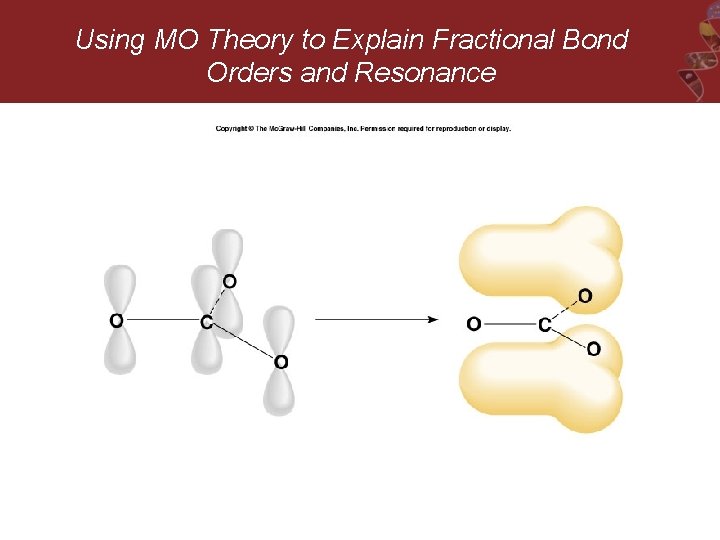
Using MO Theory to Explain Fractional Bond Orders and Resonance

MO Theory: Summary § Advantages: • Provides the most complete picture of covalent bonding, including bond types and bond orders. • Accounts for magnetic properties. § Disadvantages: • The most difficult to apply to large molecules; does not account for molecular shape.
- Slides: 108