Chapter 5 Analytic Techniques By Julia C Drees

Chapter 5: Analytic Techniques By Julia C. Drees, Matthew S. Petrie, and Alan H. B. Wu Copyright © <year> Wolters Kluwer Health | Lippincott Williams & Wilkins

Spectrophotometry and Photometry • Photometric instruments measure light intensity without consideration of wavelength. – Most instruments use filters (photometers), prisms, or gratings (spectrometers) to select (isolate) a narrow range of incident wavelength. • Beer Law: Concentration of a substance is directly proportional to right amount of light absorbed or inversely proportional to logarithm of transmitted light. • Spectrophotometric Instruments: measure light transmitted by a solution to determine concentration of light-absorbing substance in solution. Copyright © 2018 Wolters Kluwer • All Rights Reserved

Spectrophotometry and Photometry (cont. ) • Components of a Spectrophotometer – Light source – Monochromators – Sample cell – Photodetectors • Spectrophotometer Quality Assurance – Wavelength accuracy – Stray light – Linearity Copyright © 2018 Wolters Kluwer • All Rights Reserved

Spectrophotometry and Photometry (cont. ) • Single-beam spectrophotometer—see Figure 5. 5 Copyright © 2018 Wolters Kluwer • All Rights Reserved

Spectrophotometry and Photometry (cont. ) • Atomic Absorption Spectrophotometer – Measures concentration by detecting absorption of electromagnetic radiation by atoms rather than by molecules – Sensitive, precise – Routinely used to measure concentration of trace metals that are not easily excited – See Figure 5. 11 • Flame Photometry – Measures light emitted by excited atoms – Was used to determine concentration of Na, K, or Li – No longer routinely used; replaced by ion-selective electrodes Copyright © 2018 Wolters Kluwer • All Rights Reserved

Spectrophotometry and Photometry (cont. ) • Fluorometry – Basic instrumentation • Measure concentrations of solutions that contain fluorescing molecules. • Emits short-wavelength high-energy excitation light. • Mechanical attenuator controls light intensity. – Advantages and disadvantage of fluorometry • Advantages: greater specificity and sensitivity • Disadvantage: very sensitive to environmental changes Copyright © 2018 Wolters Kluwer • All Rights Reserved
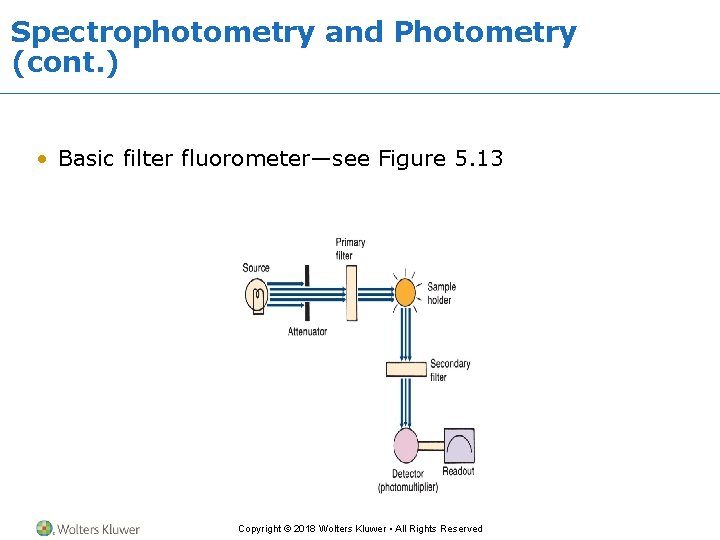
Spectrophotometry and Photometry (cont. ) • Basic filter fluorometer—see Figure 5. 13 Copyright © 2018 Wolters Kluwer • All Rights Reserved

Spectrophotometry and Photometry (cont. ) • Chemiluminescence – Part of chemical energy generated produces excited intermediates that decay to a ground state with emission of photons. – No excitation radiation or monochromators are required. – Involves oxidation reactions of luminol, acridinium esters, and dioxetanes. • Turbidity and Nephelometry – Turbidity measures concentration of particulate matter in sample. – Nephelometry: similar to turbidity, except different angle of measurement. – See Figure 5. 16 Copyright © 2018 Wolters Kluwer • All Rights Reserved

Spectrophotometry and Photometry (cont. ) • Laser Applications – LASER: Light Amplification by Stimulated Emission of Radiation. – Based on interaction of radiant energy and suitably excited atoms or molecules. – Light is polarized and coherent and has narrow spectral width and small cross-section area with low divergence. – Can serve as source of incident energy in spectrometer or nephelometer. – Laser spectrometry can be used for determination of structure, identification of samples, and diagnosis. Copyright © 2018 Wolters Kluwer • All Rights Reserved

Electrochemistry • Galvanic and Electrolytic Cells – Galvanic cells • Spontaneous flow of electrons from electrode with lower electron affinity. • Electrons pass through external meter to cathode, liberating OH− ions; reaction continues until cell is dead. – Electrolytic cells: current is forced to flow through dead cell by applying external electromotive force E. • Half-Cells – Two reactions must be coupled and one reaction compared with other to measure electrochemical activity of half-cell. Copyright © 2018 Wolters Kluwer • All Rights Reserved

Electrochemistry (cont. ) • Ion-Selective Electrodes (ISE): designed to be sensitive toward individual ions • p. H Electrodes—see Figure 5. 18 – Indicator electrode – Reference electrode – Liquid junctions – Readout meter – Nernst equation – Calibration – p. H combination electrode Copyright © 2018 Wolters Kluwer • All Rights Reserved

Electrochemistry (cont. ) • Gas-Sensing Electrodes – Designed to detect specific gases in solutions – Separated from solution by thin, gas-permeable membrane • Enzyme Electrodes – An ISE covered by immobilized enzymes that can catalyze a specific chemical reaction. • Coulometric Chloridometers and Anodic Stripping Voltammetry – Chloride ISEs have largely replaced coulometric titrations. – Anodic stripping voltammetry was widely used for analysis of lead. Copyright © 2018 Wolters Kluwer • All Rights Reserved

Electrophoresis • Migration of charged solutes/particles in an electrical field • Five components: driving force, support medium, buffer, sample, detecting system • See Figure 5. 22 Copyright © 2018 Wolters Kluwer • All Rights Reserved

Electrophoresis (cont. ) • Support materials: cellulose acetate, agarose gel, polyacrylamide gel, starch gel • Treatment and application of sample • Detection and quantitation • Electroendosmosis • Isoelectric focusing • Capillary electrophoresis • Two-dimensional electrophoresis Copyright © 2018 Wolters Kluwer • All Rights Reserved

Osmometry • Measures concentration of solute particles in a solution • Freezing point osmometer—see Figure 5. 27 – Sample in a small tube is lowered into a chamber with cold refrigerant circulating from cooling unit. – Thermistor is immersed in sample. – Wire is used to stir sample until it is cooled to several degrees below its freezing point. Copyright © 2018 Wolters Kluwer • All Rights Reserved
- Slides: 15