Chapter 41 Fluid Electrolyte and AcidBase Balance Bonnie

Chapter 41: Fluid, Electrolyte, and Acid-Base Balance Bonnie M. Wivell, MS, RN, CNS
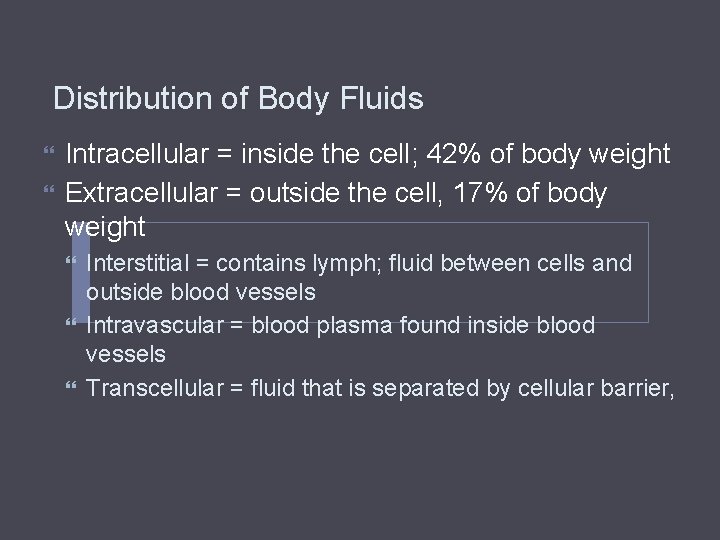
Distribution of Body Fluids Intracellular = inside the cell; 42% of body weight Extracellular = outside the cell, 17% of body weight Interstitial = contains lymph; fluid between cells and outside blood vessels Intravascular = blood plasma found inside blood vessels Transcellular = fluid that is separated by cellular barrier,
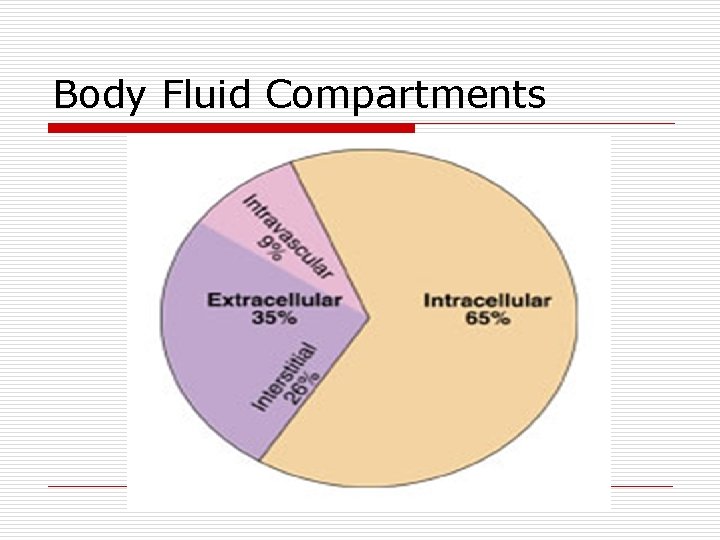
Body Fluid Compartments

Functions of Body Fluid o o o o Major component of blood plasma Solvent for nutrients and waste products Necessary for hydrolysis of nutrients Essential for metabolism Lubricant in joints and GI tract Cools the body through perspiration Provides some mineral elements

Composition of Body Fluids Body fluids contain Electrolytes Anions – negative charge Cations – positive charge Cl, HCO 3, SO 4 Na, K, Ca Electrolytes are measured in m. Eq Minerals are ingested as compounds and are constituents of all body tissues and fluids Minerals act as catalysts

Electrolytes in Body Fluids o Normal Values n n n n Sodium (Na+) 35 – 145 m. Eq/L Potassium (K+) 3. 5 – 5. 0 m. Eq/L Ionized Calcium (Ca++) 4. 5 – 5. 5 mg/d. L Calcium (Ca++) 8. 5 – 10. 5 mg/d. L Bicarbonate (HCO 3) 24 – 30 m. Eq/L Chloride (Cl--) 95 – 105 m. Eq/L Magnesium (Mg++) 1. 5 – 2. 5 m. Eq/L Phosphate (PO 4 ---) 2. 8 – 4. 5 mg/d. L

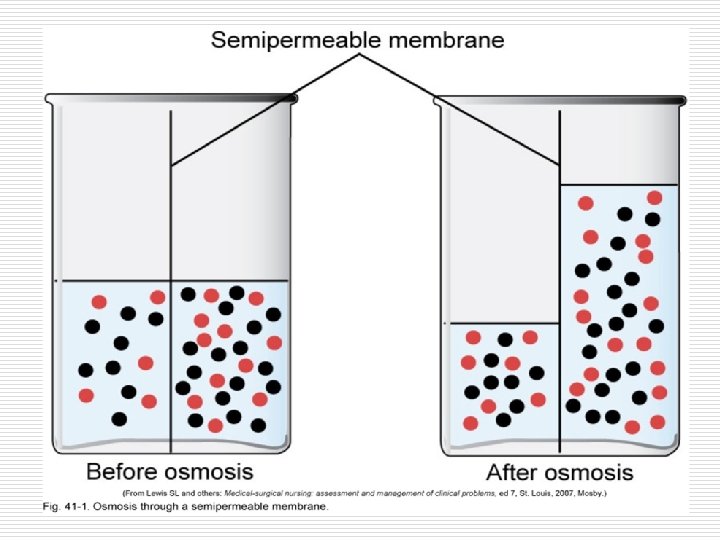
Movement of Body Fluids Osmosis = movement across a semi-permeable membrane from area of lesser concentration to are of higher concentration; high solute concentration has a high osmotic pressure and draws water toward itself Osmotic pressure = drawing power of water (Osmolality) Osmolarity = concentration of solution

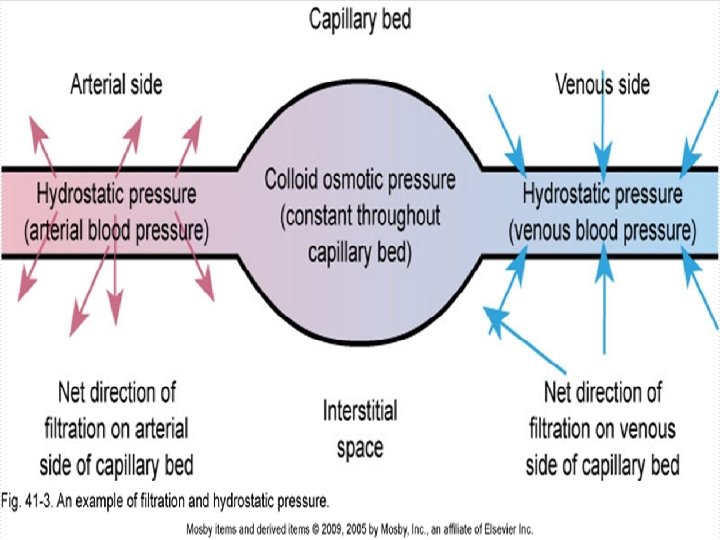
Movement of Body Fluids o Colloid or Oncotic pressure = keeps fluid in the intravascular compartment by pulling water from the interstitial space back into the capillaries

Solutions o Isotonic Solution n o Hypotonic Solution n o The same concentration as blood plasma; expand fluid volume without causing fluid shift Lower concentration than blood plasma; moves fluid into the cells causing them to enlarge Hypertonic solution n Higher concentration than blood plasma; pulls fluid from cells causing them to shrink

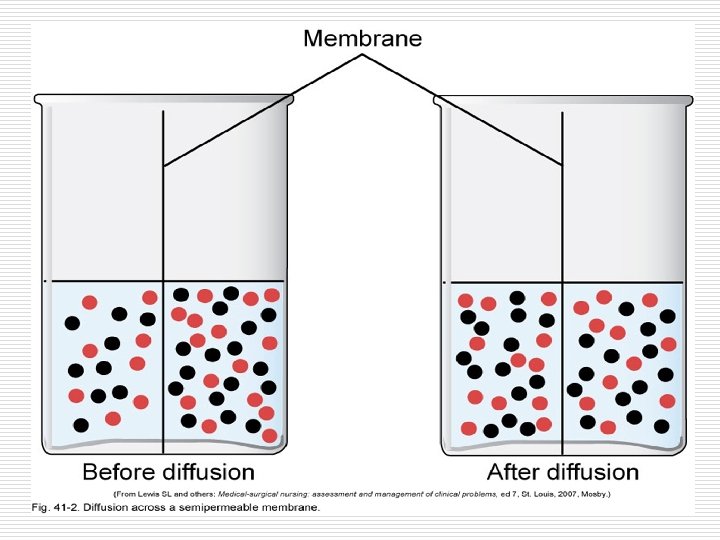
Movement of Body Fluids Cont’d. o o Diffusion = Molecules move from higher concentration to lower o Concentration gradient Filtration = water and diffusible substances move together across a membrane; moving from higher pressure to lower pressure Edema results from accumulation of excess fluid in the interstitial space Hydrostatic pressure causes the movement of fluids from an area of higher pressure to area of lower pressure

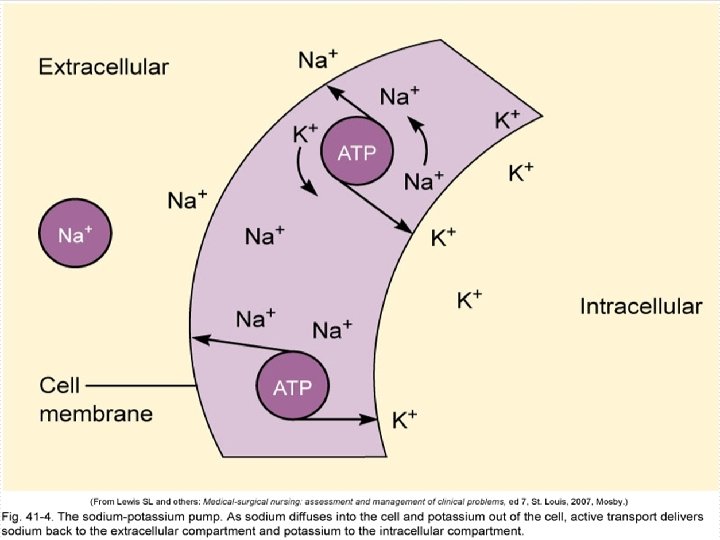
Active Transport o Requires metabolic activity and uses energy to move substances across cell membranes n n n Enables larger substances to move into cells Molecules can also move to an area of higher concentration (Uphill) Sodium-Potassium Pump o o Potassium pumped in – higher concentration in ICF Sodium pumped out – higher concentration in ECF

Regulation of Body Fluids o Homeostasis is maintained through n n n Fluid intake Hormonal regulation Fluid output regulation

Fluid Intake o o Thirst control center located in the hypothalamus n Osmoreceptors monitor the serum osmotic pressure n When osmolarity increases (blood becomes more concentrated), the hypothalamus is stimulated resulting in thirst sensation o Salt increases serum osmolarity Hypovolemia occurs when excess fluid is lost

Fluid Intake o Average adult intake n o 2200 – 2700 m. L per day o Oral intake accounts for 1100 – 1400 m. L per day o Solid foods about 800 – 1000 m. L per day o Oxidative metabolism – 300 m. L per day Those unable to respond to the thirst mechanism are at risk for dehydration n Infants, patients with neuro or psych problems, and older adults

Hormonal Regulation o ADH (Antidiuretic hormone) n n n Stored in the posterior pituitary and released in response to serum osmolarity Pain, stress, circulating blood volume effect the release of ADH o Increase in ADH = Decrease in urine output = Body saves water Makes renal tubules and ducts more permeable to water

Hormonal Regulation Cont’d. o Renin-angiotensin-aldosterone mechanism n n Changes in renal perfusion initiates this mechanism Renin responds to decrease in renal perfusion secondary to decrease in extracellular volume Renin acts to produce angiotensin I which converts to angiotensin II which causes vasoconstriction, increasing renal perfusion Angiotensin II stimulates the release of aldosterone when sodium concentration is low

Hormonal Regulation Cont’d. o Aldosterone n n Released in response to increased plasma potassium levels or as part of the reninangiotensin-aldosterone mechanism to counteract hypovolemia Acts on the distal portion of the renal tubules to increase the reabsorption of sodium and the secretion and excretion of potassium and hydrogen Water is retained because sodium is retained Volume regulator resulting in restoration of blood volume

Hormonal Regulation Cont’d. o Atrial Natriuretic Peptide (ANP) n n n ANP is a hormone secreted from atrial cells of the heart in response to atrial stretching and an increase in circulating blood volume ANP acts like a diuretic that causes sodium loss and inhibits the thirst mechanism Monitored in CHF

Fluid Output Regulation o Organs of water loss n n Kidneys Lungs Skin GI tract

Fluid Output Regulation Cont’d. o Kidneys are major regulatory organ of fluid balance n n n o Receive about 180 liters of plasma to filter daily 1200 – 1500 m. L of urine produced daily Urine volume changes related to variation in the amount and type of fluid ingested Skin n Insensible Water Loss o o n Sensible Water Loss occurs through excess perspiration o n Continuous and occurs through the skin and lungs Can significantly increase with fever or burns Can be sensible or insensible via diffusion or perspiration 500 – 600 m. L of insensible and sensible fluid lost through skin each day

Fluid Output Regulation Cont’d. o Lungs n n o Expire approx 500 m. L of water daily Insensible water loss increases in response to changes in resp rate and depth and oxygen administration GI Tract n n 3 – 6 liters of isotonic fluid moves into the GI tract and then returns to the ECF 200 m. L of fluid is lost in the feces each day o Diarrhea can increase this loss significantly

Regulation of Electrolytes o Major Cations in body fluids n n Sodium (Na+) Potassium (K+) Calcium (Ca++) Magnesium (Mg++)

Sodium Regulation o Most abundant cation in the extracellular fluid n o Major contributor to maintaining water balance o Nerve transmission o Regulation of acid-base balance o Contributes to cellular chemical reactions Sodium is taken in via food and balance is maintained through aldosterone

Potassium Regulation o Major electrolyte and principle cation in the extracellular fluid n n Regulates metabolic activities Required for glycogen deposits in the liver and skeletal muscle Required for transmission of nerve impulses, normal cardiac conduction and normal smooth and skeletal muscle contraction Regulated by dietary intake and renal excretion

Calcium Regulation o Stored in the bone, plasma and body cells n n n 99% of calcium is in the bones and teeth 1% is in ECF 50% of calcium in the ECF is bound to protein (albumin) 40% is free ionized calcium Is necessary for o Bone and teeth formation o Blood clotting o Hormone secretion o Cell membrane integrity o Cardiac conduction o Transmission of nerve impulses

Magnesium Regulation o o Essential for enzyme activities Neurochemical activities Cardiac and skeletal muscle excitability Regulation n o Dietary Renal mechanisms Parathyroid hormone action 50 – 60% of magnesium contained in bones n n 1% in ECF Minimal amount in cell

Anions o Chloride (Cl-) n n o Major anion in ECF Follows sodium Bicarbonate (HCO 3 -) n n n Is the major chemical base buffer Is found in ECF and ICF Regulated by kidneys

Anions Cont’d. o Phosphate (PO 4 ---) n n n n Buffer ion found in ICF Assists in acid-base regulation Helps to develop and maintain bones and teeth Calcium and phosphate are inversely proportional Promotes normal neuromuscular action and participates in carbohydrate metabolism Absorbed through GI tract Regulated by diet, renal excretion, intestinal absorption and PTH

Regulation of Acid-Base Balance o o Lungs and kidneys are our buffering systems A buffer is a substance that can absorb or release H+ to correct an acid-base imbalance Arterial p. H is an indirect measure of hydrogen ion concentration Greater concentration of H+, more acidic, lower p. H

Regulation of Acid-Base Balance o o o Lower concentration of H+, more alkaline, higher p. H The p. H is also a reflection of the balance between CO 2 (regulated by lungs) and bicarb (regulated by kidneys) Normal H+ level is necessary to n n Maintain cell membrane integrity Maintain speed of cellular enzymatic actions

Chemical Regulation o o o Carbonic acid-bicarbonate buffer system is the first to react to change in the p. H of ECF H+ and CO 2 concentrations are directly related ECF becomes more acidic, the p. H decreases, producing acidosis ECF receives more base substances, the p. H rises, producing alkalosis Lungs primarily control excretion of CO 2 resulting from metabolism Kidneys control excretion of hydrogen and bicarb

Biological Regulation 1. Buffer actions that occur 1. 2. 3. Exchange of K+ and H+ Carbon dioxide goes into RBC carbonic acid (HCO 3 -) 1. HCO 3 ready to exchange with Cl. Chloride shift within RBC H+ H+ K+ K+ K+ H+ H+ H+

Acidosis vs Alkalosis o o o Acidosis n Acids have high H+ ions in solution n Bases have low H+ ion concentration Alkalosis Acidity or Alkalinity of a solution measured by p. H
Physiological Regulators o Lungs n n n o Regulate by altering H+ ions Metabolic acidosis Metabolic alkalosis Kidneys n Regulate by altering HCO 3 and H+ ions H+ H+ HCO 3

Causes of Electrolyte Imbalances o o o o Excessive sweating Fluid loss leading to dehydration Excessive vomiting Diuretics like Lasix (K+ depletion) Massive blood loss Dehydration may go unnoticed in hot, dry climates Renal failure
Sodium o o o Most abundant in extracellular space Moves among three fluid compartments Found in most body secretions Na Na Na

Hyponatremia – Low Sodium o o o Seizures Personality changes Nausea/vomiting Tachycardia Convulsion Normal Na (135145)
Hypernatremia o o Excessive Na in ECF Loss of water n n n o Diarrhea Insensible water loss Water deprivation Gain of Sodium n n Diabetes insipidus Heat stroke
Hypokalemia – Low Potassium o o o Severe leg cramps Flaccid muscles Fatigue Irregular pulse Chest discomfort EKG changes n o T wave flattens Normal Potassium 3. 5 -5
Hyperkalemia o CNS n o Peripheral Nervous System n o Nausea and vomiting Tremors, twitching Heart n Bradycardia, peaked T wave
Hypocalcemia – Low Calcium o o Tingling of fingers Tetany Muscle cramps Positive Trousseau’s n o Carpal spasm Positive Chvostek’s n Contraction of facial muscle when facial nerve tapped
Hypercalcemia o Causes n n o Prolonged immobility Osteoporosis Thiazide diuretics Acidosis Signs/symptoms n n n N/V, weakness Hypoactive reflexes Cardiac arrest

Hypomagnesemia o Causes n n o Malnutrition Alcoholism Polyuria Pre-ecclampsia Signs/symptoms n n Muscle tremor Hyperactive deep reflexes Chvostek’s/Trousseau’s Difficulty breathing

Hypermagnesemia o Causes n n o Renal failure Excessive intake Signs/symptoms n n Low BP Muscle weakness Absent reflexes Bradycardia

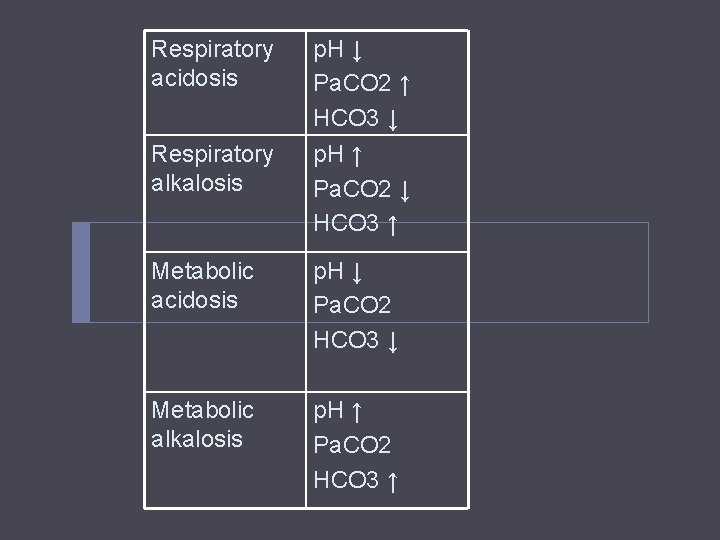
Respiratory acidosis Respiratory alkalosis p. H ↓ Pa. CO 2 ↑ HCO 3 ↓ p. H ↑ Pa. CO 2 ↓ HCO 3 ↑ Metabolic acidosis p. H ↓ Pa. CO 2 HCO 3 ↓ Metabolic alkalosis p. H ↑ Pa. CO 2 HCO 3 ↑

Cheat Sheet o o o Increase p. H – alkalosis Decrease p. H – acidosis Respiratory – CO 2 Metabolic (kidneys)– HCO 3 CO 2 has an inverse relationship with p. H When p. H goes down, CO 2 goes up HCO 3 follows p. H. If p. H goes up so does HCO 3 CO 2 increases, p. H decreases – resp. acidosis CO 2 decreases, p. H increases – resp. alkalosis HCO 3 increases, p. H increases – metabolic alkalosis HCO 3 decreases, p. H decreases – metabolic acidosis

Question An older client comes to the emergency department experiencing chest pain and shortness of breath. An arterial blood gas is ordered. Which of the following ABG results indicates respiratory acidosis? 1. p. H - 7. 54, Pa. CO 2 – 28, HCO 3 – 22 2. p. H – 7. 32, Pa. CO 2 – 46, HCO 3 – 24 3. p. H – 7. 31, Pa. CO 2 – 35, HCO 3 – 20 4. p. H – 7. 5, Pa. CO 2 – 37, HCO 3 - 28

Review Acid/Base Imbalance Tutorial How do we assess for acid-base balance?

Assessment o Nursing history n o Age Prior Medical History n n n Acute illness Surgery Burns increase fluid loss Resp. disorder predisposes to resp. acidosis Head Injury can alter ADH secretion Chronic illness o o Cancer CVD Renal disorders GI disturbances

Assessment Cont’d. o o o Environmental factors affecting fluid/electrolyte alterations Diet Lifestyle – smoking, ETOH Medications Physical Assessment n n n o Daily weights I&O Vital signs Laboratory Studies

Nursing Diagnosis o o o o o Decreased cardiac output Acute confusion Deficient fluid volume Excess fluid volume Impaired gas exchange Risk for injury Deficient knowledge regarding disease management Impaired oral mucous membrane Impaired skin integrity Ineffective tissue perfusion

Planning o o o Determine goals and outcomes Set priorities Collaborative care n n n MD Dietician Pharmacy

Implementation o Health promotion n o Education Acute care n n n Enteral replacement of fluids Restriction of fluids Parenteral replacement of fluids and electrolytes o o o n TPN IV fluids and electrolyte therapy (crystalloids) Blood and blood components (colloids) n Blood groups and types n Autologous transfusion n Transfusion reactions ABGs

Restorative Care o o o Home IV therapy Nutritional support Medication safety n Pt. education

Evaluation o o o Have goals been met? Have changes in assessment occurred? Progress determines need to continue or revise plan of care
- Slides: 60