Chapter 4 Types of Chemical Reactions Goals To








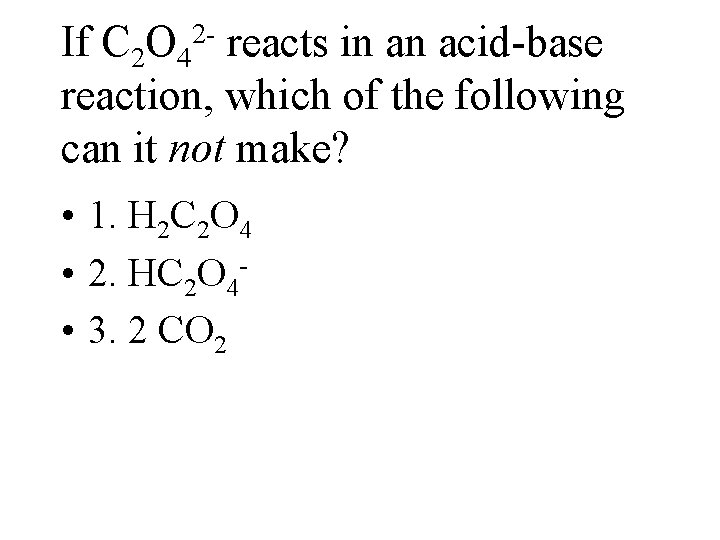






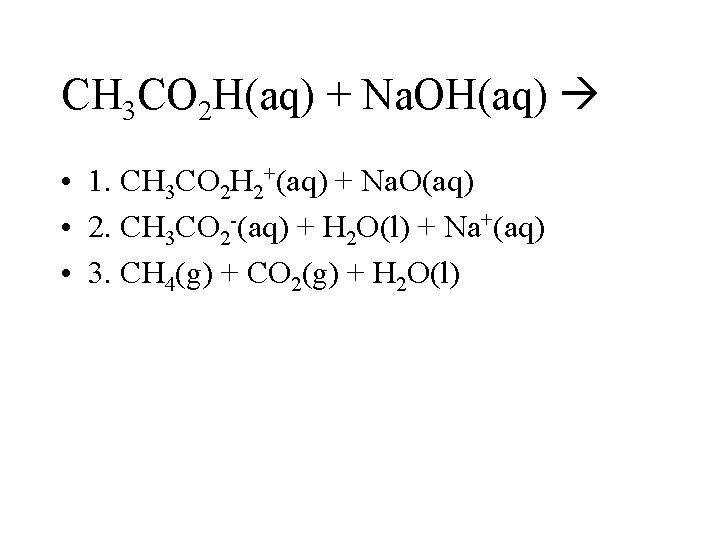


![Finding + [H 3 O ] [H 3 O+] = 10 -p. H What Finding + [H 3 O ] [H 3 O+] = 10 -p. H What](https://slidetodoc.com/presentation_image_h2/88c16297992134352d3a2f20bf45b016/image-26.jpg)

- Slides: 27

Chapter 4: Types of Chemical Reactions Goals: • To be able to predict chemical reactivity. • To know how to synthesize specific compounds.

Types of Reactions • • • Acid-Base: proton-transfer Oxidation-Reduction: electron-transfer Precipitation: formation of insoluble salts Gas Forming Organic: – Substitution – Addition – Elimination

Reactions in Aqueous Solution Unless mentioned, all reactions studied this and next week occur in aqueous solution.

Electrolytes Strong Electrolytes: solute breaks apart to give ions in solution. Na. Cl Na+ + Cl. Weak Electrolytes: solute partially breaks apart to give ions. CH 3 CO 2 H CH 3 CO 2 - + H+ Nonelectrolytes: no ions formed. CH 3 CH 2 OH happens less than 5%

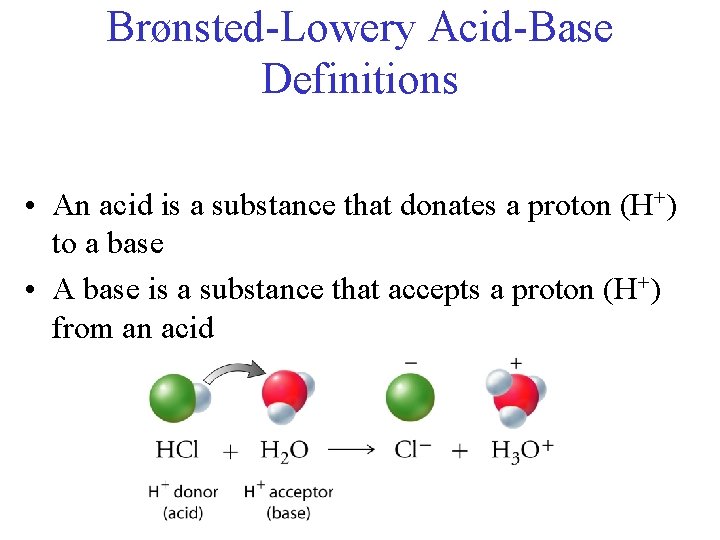
Brønsted-Lowery Acid-Base Definitions • An acid is a substance that donates a proton (H+) to a base • A base is a substance that accepts a proton (H+) from an acid

Brønsted-Lowery Definitions • acid: donates a proton (H+) to a base • base: accepts a proton (H+) from an acid • Acid-base reactions can be reversible: reactants products or products reactants

Brønsted-Lowery Definitions • An acid is a substance that donates a proton (H+) to a base • A base is a substance that accepts a proton (H+) from an acid • Acid-base reactions can be reversible: reactants products or products reactants

Important Acids and Bases Strong Acids: Strong Bases: HCl hydrochloric HBr hydrobromic HI hydroiodic HNO 3 nitric H 2 SO 4 sulfuric HCl. O 4 perchloric Weak Acid: CH 3 CO 2 H acetic Any other acids are WEAK Li. OH lithium hydroxide Na. OH sodium hydroxide KOH potassium hydroxide Ca(OH)2 calcium hydroxide Ba(OH)2 barium hydroxide Weak Base: NH 3 ammonia

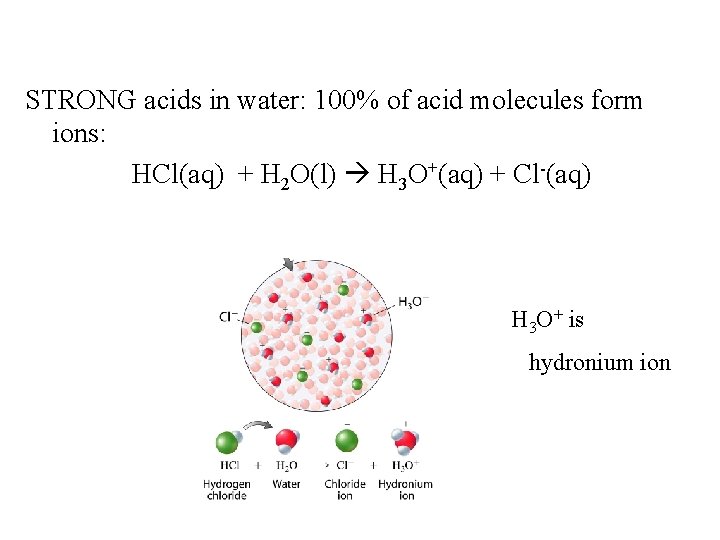
STRONG acids in water: 100% of acid molecules form ions: HCl(aq) + H 2 O(l) H 3 O+(aq) + Cl-(aq) H 3 O+ is hydronium ion

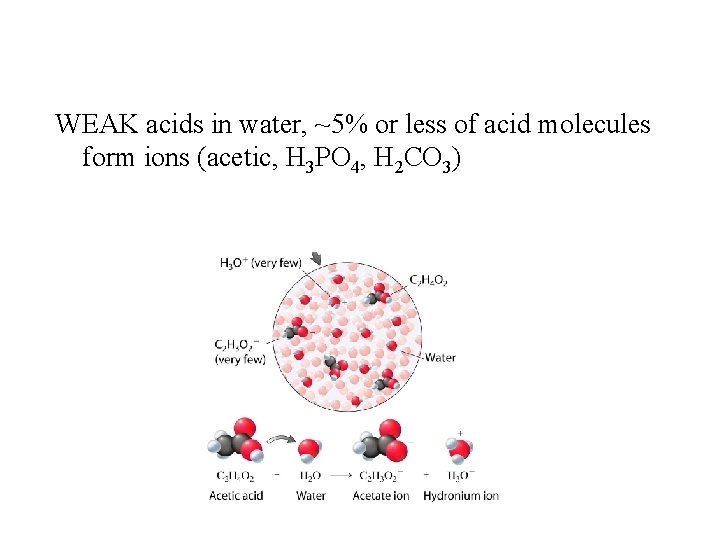
WEAK acids in water, ~5% or less of acid molecules form ions (acetic, H 3 PO 4, H 2 CO 3)

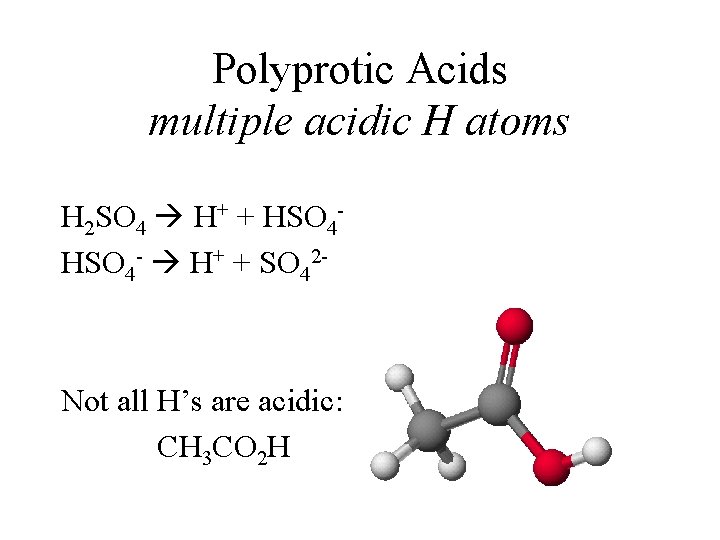
Polyprotic Acids multiple acidic H atoms H 2 SO 4 H+ + HSO 4 - H+ + SO 42 - Not all H’s are acidic: CH 3 CO 2 H

If H 3 PO 4 reacts as an acid, which of the following can it not make? • • 1. H 4 PO 4+ 2. H 2 PO 43. HPO 424. PO 43 -
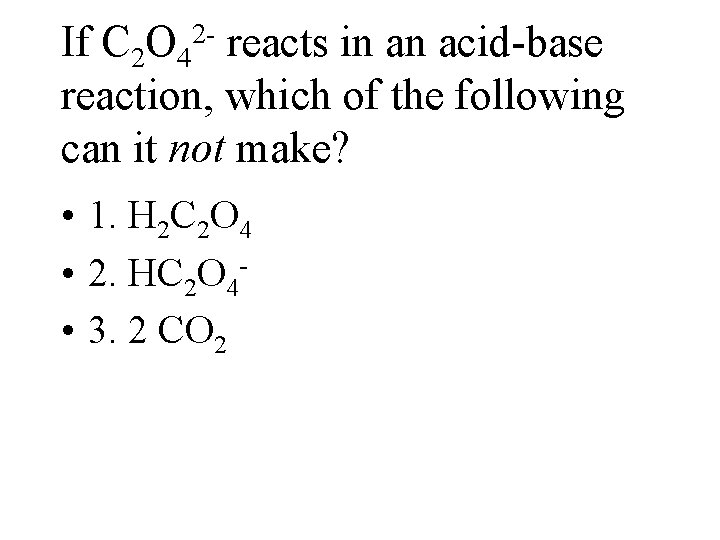
If C 2 O 4 reacts in an acid-base reaction, which of the following can it not make? 2 - • 1. H 2 C 2 O 4 • 2. HC 2 O 4 • 3. 2 CO 2

Acid-Base Reactions Strong Acid + Strong Base HCl(aq) + Na. OH(aq) Na. Cl(aq) + H 2 O(l) acid base “salt” water

Acid-Base Reactions Diprotic Acids or Bases H 2 SO 4(aq) + Na. OH(aq) H 2 SO 4(aq) + Ba(OH)2(aq) HCl(aq) + Ba(OH)2(aq)

Acid-Base Reactions Strong Acid + Weak Base HCl(aq) + NH 3(aq) NH 4 Cl(aq)

Acid-Base Reactions Weak Acid + Strong Base HCN(aq) + Na. OH(aq) Na. CN(aq) + H 2 O(l) acid base “salt” water

Net Ionic Equations HCl(aq) + Na. OH(aq) Na. Cl(aq) + H 2 O(l) What really happens: H+(aq) + OH-(aq) H 2 O(l) Sodium ion and chloride ion are “spectator ions”

Reactions involving weak bases HCl(aq) + NH 3(aq) NH 4+(aq) + Cl-(aq) Net-Ionic Equation: NH 3(aq) + H+(aq) NH 4+(aq)
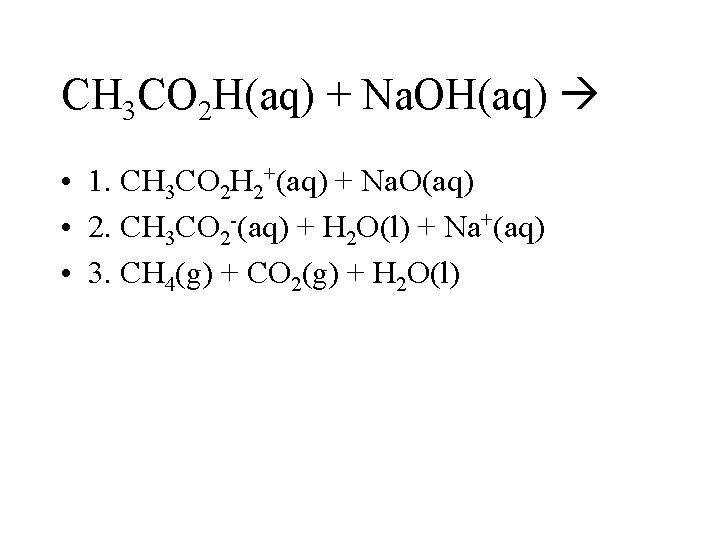
CH 3 CO 2 H(aq) + Na. OH(aq) • 1. CH 3 CO 2 H 2+(aq) + Na. O(aq) • 2. CH 3 CO 2 -(aq) + H 2 O(l) + Na+(aq) • 3. CH 4(g) + CO 2(g) + H 2 O(l)

HCN(aq) + NH 3(aq) • 1. NH 4+(aq) + CN-(aq) • 2. H 2 CN+(aq) + NH 2 -(aq) • 3. C 2 N 2(s) + 3 H 2(g)

Solution Concentration: Molarity • Molarity = moles solute per liter of solution • 0. 30 mol NH 3 dissolved in 0. 500 L Concentration = • Written like: [NH 3] = 0. 60 M

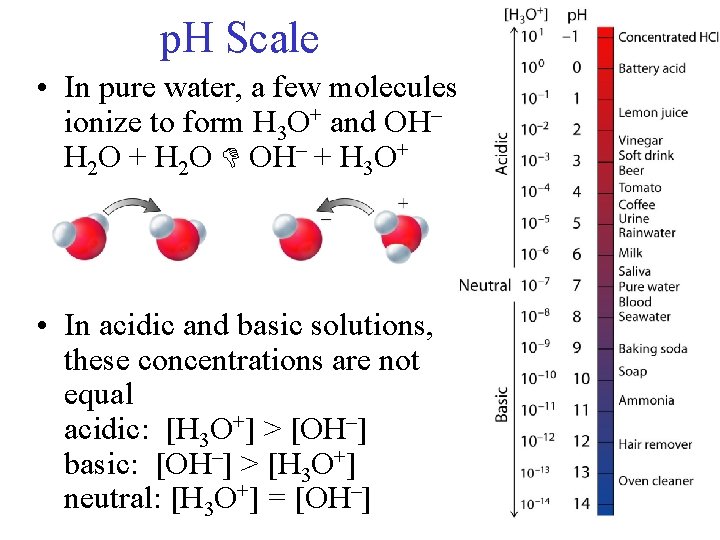
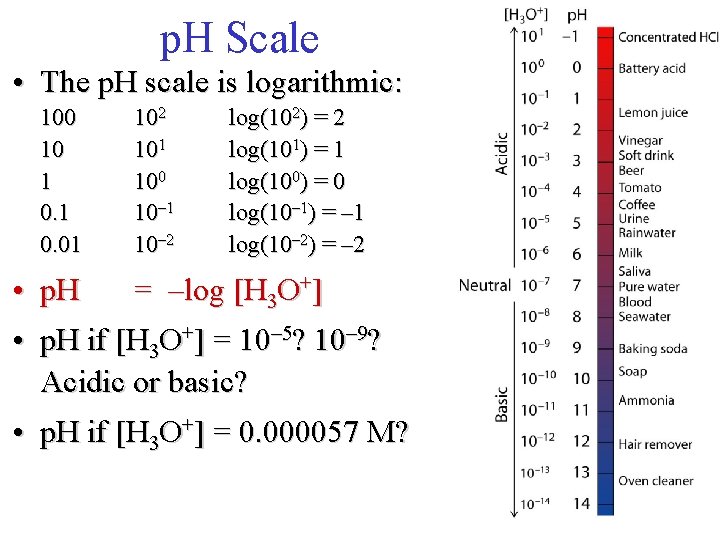
p. H Scale • In pure water, a few molecules ionize to form H 3 O+ and OH– H 2 O + H 2 O OH– + H 3 O+ • In acidic and basic solutions, these concentrations are not equal acidic: [H 3 O+] > [OH–] basic: [OH–] > [H 3 O+] neutral: [H 3 O+] = [OH–]

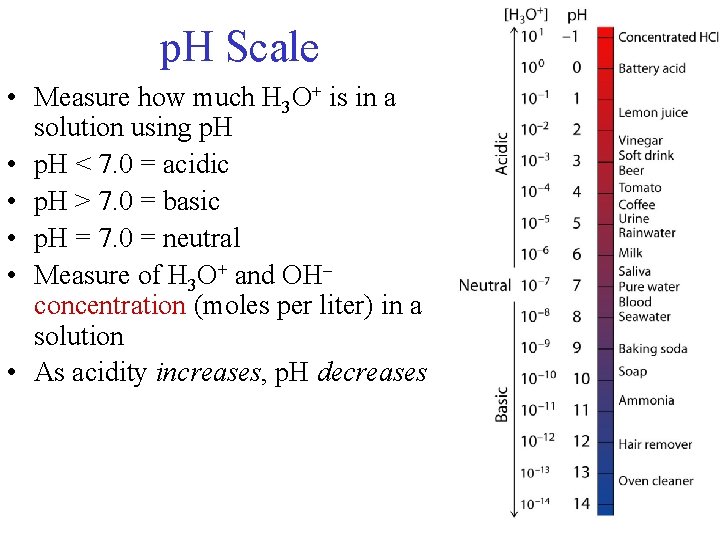
p. H Scale • Measure how much H 3 O+ is in a solution using p. H • p. H < 7. 0 = acidic • p. H > 7. 0 = basic • p. H = 7. 0 = neutral • Measure of H 3 O+ and OH– concentration (moles per liter) in a solution • As acidity increases, p. H decreases

p. H Scale • The p. H scale is logarithmic: 100 10 1 0. 01 102 101 100 10– 1 10– 2 log(102) = 2 log(101) = 1 log(100) = 0 log(10– 1) = – 1 log(10– 2) = – 2 • p. H = –log [H 3 O+] • p. H if [H 3 O+] = 10– 5? 10– 9? Acidic or basic? • p. H if [H 3 O+] = 0. 000057 M?
![Finding H 3 O H 3 O 10 p H What Finding + [H 3 O ] [H 3 O+] = 10 -p. H What](https://slidetodoc.com/presentation_image_h2/88c16297992134352d3a2f20bf45b016/image-26.jpg)
Finding + [H 3 O ] [H 3 O+] = 10 -p. H What is [H 3 O+] if p. H = 8. 9? from p. H

p. H: Quantitative Measure of Acidity • Acidity is related to concentration of H+ (or H 3 O+) • p. H = -log[H 3 O+]