Chapter 4 The Modern Model of the Atom


































![• 1 = 1 s 1 • 5 = [He] 2 s 22 • 1 = 1 s 1 • 5 = [He] 2 s 22](https://slidetodoc.com/presentation_image_h2/2c35966d38acb8b7bcbfb85f9c665a7b/image-47.jpg)


![• 1 = 1 s 1 • 5 = [He] 2 s 22 • 1 = 1 s 1 • 5 = [He] 2 s 22](https://slidetodoc.com/presentation_image_h2/2c35966d38acb8b7bcbfb85f9c665a7b/image-51.jpg)









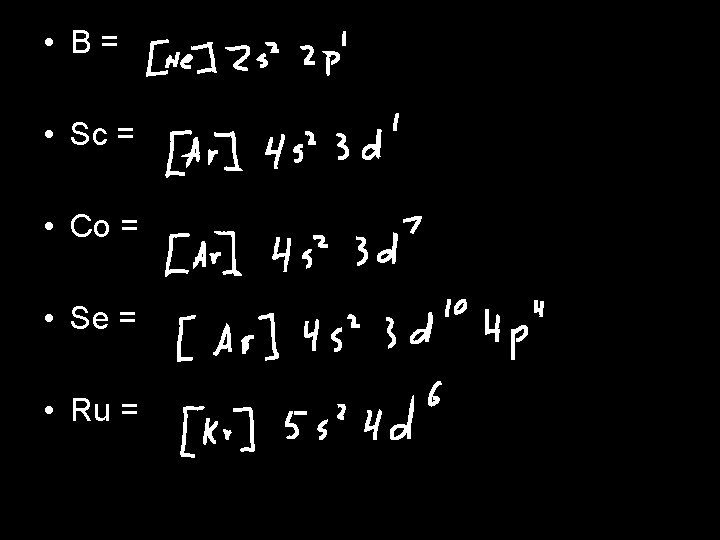
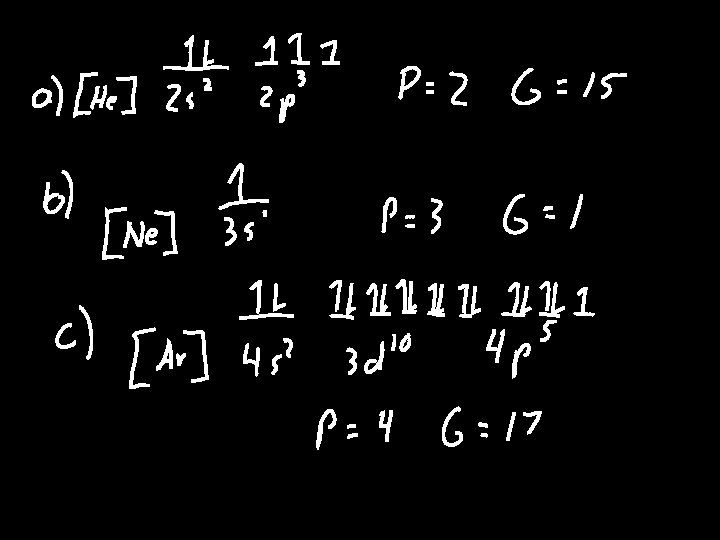


- Slides: 65

Chapter 4 The Modern Model of the Atom

The Puzzle of the Atom § Protons and electrons are attracted to each other because of opposite charges § Electrically charged particles moving in a curved path give off energy § Despite these facts, atoms don’t collapse §HMMMMM §The answer is in the light

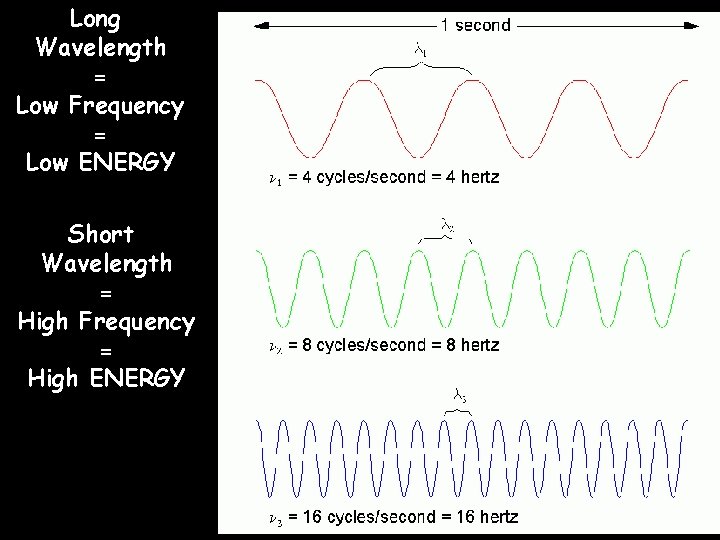
Electromagnetic radiation propagates through space as a wave moving at the speed of light. c c = = speed of light, a constant (3. 00 x 108 m/s) = frequency, in units of hertz (Hz, sec-1) = wavelength, in meters

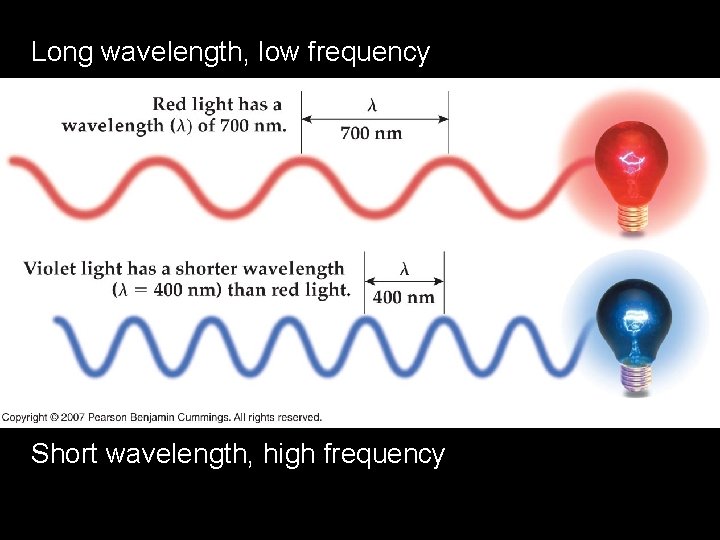
Long wavelength, low frequency Short wavelength, high frequency

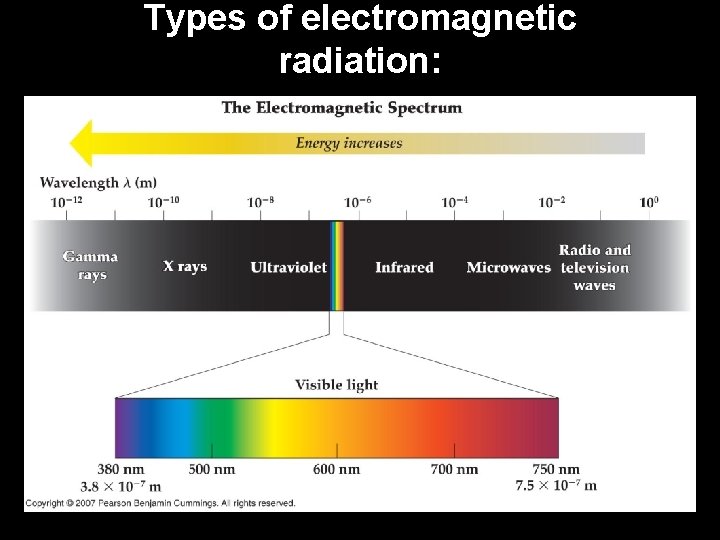
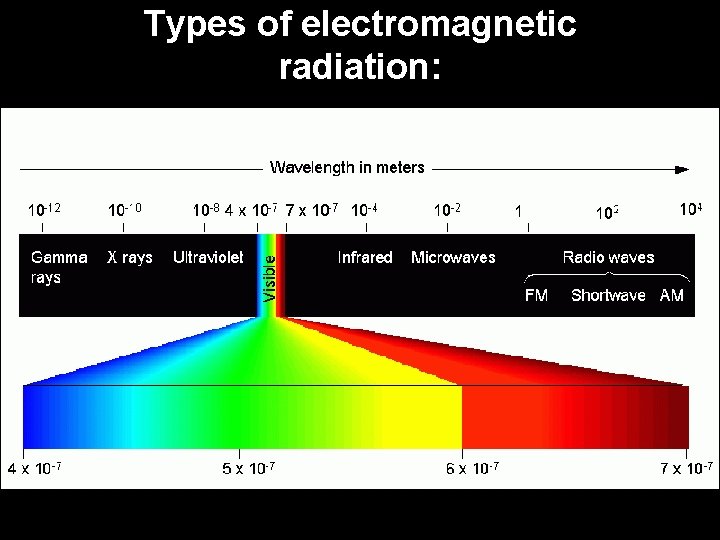
Types of electromagnetic radiation:

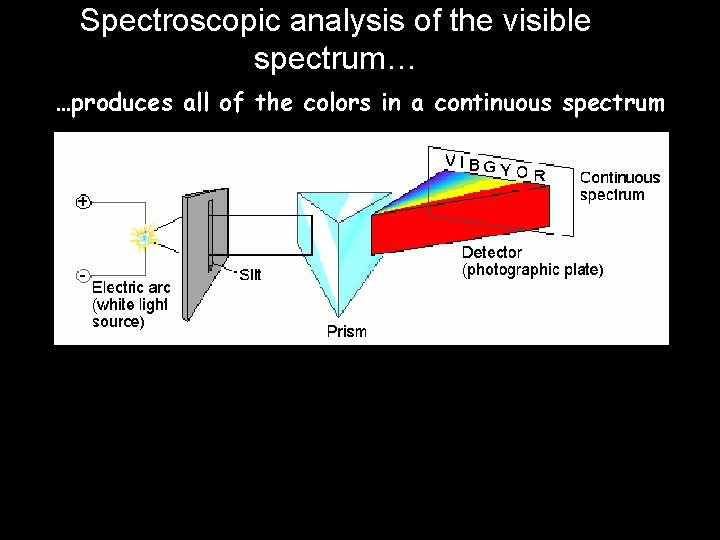
Spectroscopic analysis of the visible spectrum… …produces all of the colors in a continuous spectrum

Example • Calculate the frequency of yellow light that has a wavelength of 559 nm (nanometers)

The energy (E ) of electromagnetic radiation is directly proportional to the frequency ( ) of the radiation. E = h E = Energy, in units of Joules (kg·m 2/s 2) h = Planck’s constant (6. 626 x 10 -34 J·s) = frequency, in units of hertz (hz, sec-1)

Alternate Equation E = hc/ E = Energy, in units of Joules (kg·m 2/s 2) h = Planck’s constant (6. 626 x 10 -34 J·s) c = Speed of Light (3. 00 x 108 m/s) = Wavelength (meters)

Example • What is the energy associated with yellow light at 559 nm? Orange light at 633 nm?

Long Wavelength = Low Frequency = Low ENERGY Short Wavelength = High Frequency = High ENERGY Wavelength Table

Problems o p. 154 #'s 30, 31, 38, 40 include frequency

But What Does It Mean? • Well, it means there are only certain energies available in an atom. Of course! • Energy is “quantized” – Only certain energies are allowed – Ex Cars can only travel at certain speeds

Bohr’s Model of the Atom • Electrons can circle the nucleus only in allowed paths or shells • The energy of the electron is greater when it is in a shell further from the nucleus (excited state) • The atom achieves the ground state when electrons occupy the closest possible positions around the nucleus • Electromagnetic radiation (light) is emitted when electrons move closer to the nucleus

Another Example • Bohr’s Model Applet • http: //www. colorado. edu/physics/2000/qua ntumzone/bohr. html

Calculations • There are only certain energies available to an atom • Bohr’s Model of the Atom • Mathematical Model • E=-2. 178 x 10 -18 J (Z 2/n 2) • Z is nuclear charge • n is energy level • E = 2. 178 x 10 -18 J ((Z/n 2 f) - (Z/n 2 i))

The Bohr Model of the Atom Electrons can jump from a lower shell to a higher shell by gaining a specific amount of energy. Neils Bohr Electrons give off a specific frequency of light when they return to a lower shell

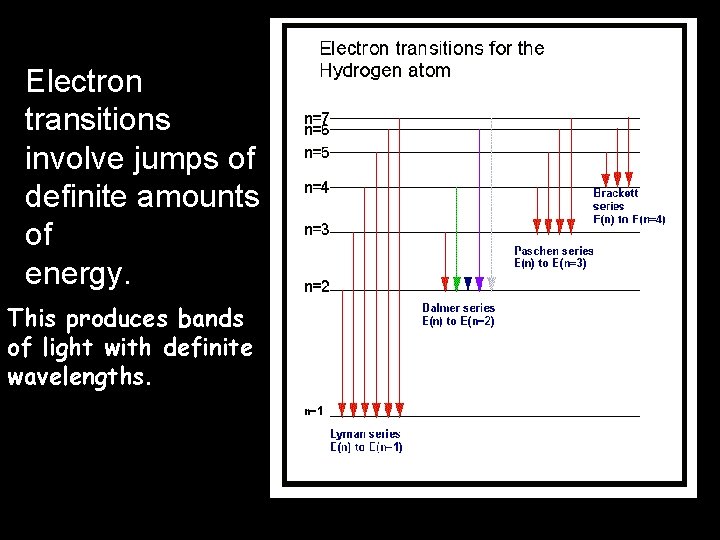
Electron transitions involve jumps of definite amounts of energy. This produces bands of light with definite wavelengths.

Spectroscopic analysis of the visible spectrum… …produces all of the colors in a continuous spectrum

Spectroscopic analysis of the hydrogen spectrum… …produces a “bright line” spectrum

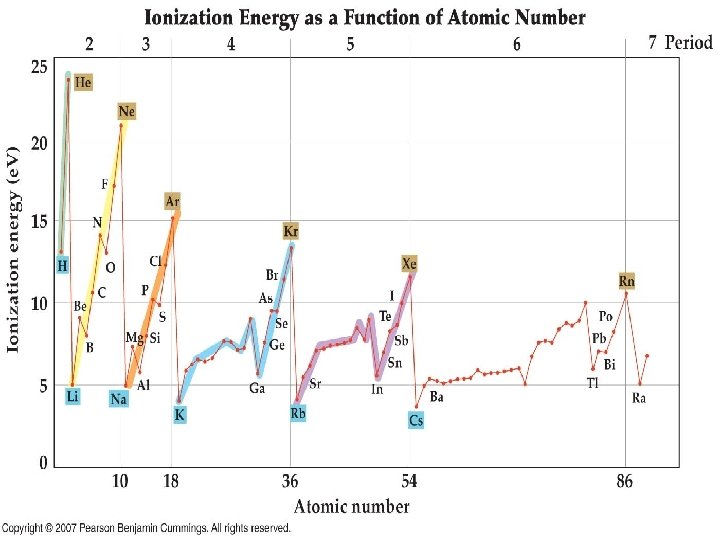
Energy Values for Shells • • • Shell 1 = 1. 0 e. V Shell 2 = 11. 2 e. V Shell 3 = 13. 1 e. V Shell 4 = 13. 8 e. V Shell 5 = 14. 1 e. V Shell 6 = 14. 2 e. V – 1 e. V = 1. 602 x 10 -19 J • Why such a small change in outer shells?

Bohr’s Model cont. • Each shell can hold a specific number of electrons – 2 n 2 where n is the shell number 1, 2, 3. • 1 st shell = 2 electrons • 2 nd shell = 8 electrons • 3 rd shell = 18 electrons • 4 th shell = 32 electrons – Elements in the same group on the periodic table must have the same number of electrons in the outer shell


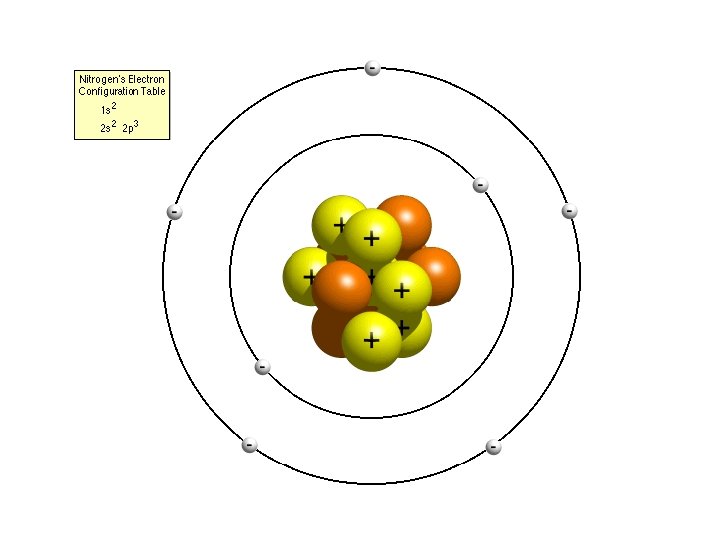
Time to Draw! • Draw a Bohr atom for Lithium-7 and Sodium-23 – Is there anything similar/different about the atoms?

Homework • P. 155 #’s 41, 45, 48, 52, 55, 56

A) Calculate the wavelength of light (nm) associated with an electron transition from the n=5 shell to n=2 shell. B) What color of light is this? C) How would this transition compare to n=5 to n=1?

Types of electromagnetic radiation:

So this is the model of the atom? • No. • There a couple problems. 1. Bohr’s mathematics only predicts hydrogen’s spectrum (and 1 electron systems) 2. Try drawing a Bohr atom for Potassium – Yep, wrong number of electrons in outer shell. Should be 1 not 9

So why waste our time with this? • Good question – Well there a few reasons 1. Easy to predict outer number of electrons 2. Gave evidence for the structure of atoms

Homework o 155 #'s 60, 62, 65, 66 include frequency

So, what’s the fix? • Quantum Mechanics! – Placing electrons in subshells and orbitals – Treating the electron as a standing wave.

Subshells • The spectra (colors) emitted from excited atoms did not match what Bohr predicted. • Scientists proposed the idea of subshells within the shells – Subshell – place of specific energy where electrons have a high probability of being found. – Each subshell has a specific number of orbitals

Subshells cont. • • • Subshells are designated as – s, p, d, f s has 1 orbital and holds 2 electrons. p has 3 orbitals and holds 6 electrons. d has 5 orbitals and holds 10 electrons. f has 7 orbitals and holds 14 electrons. • each subshell is higher in energy for a specific shell only!!!!!!

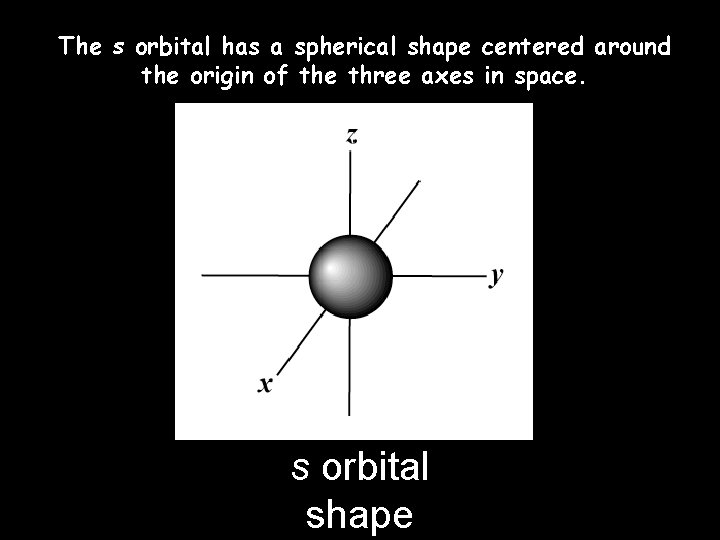
The s orbital has a spherical shape centered around the origin of the three axes in space. s orbital shape

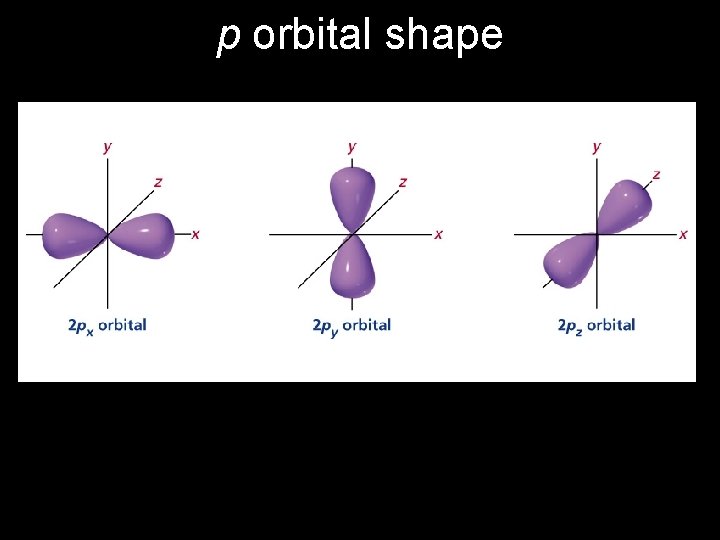
p orbital shape

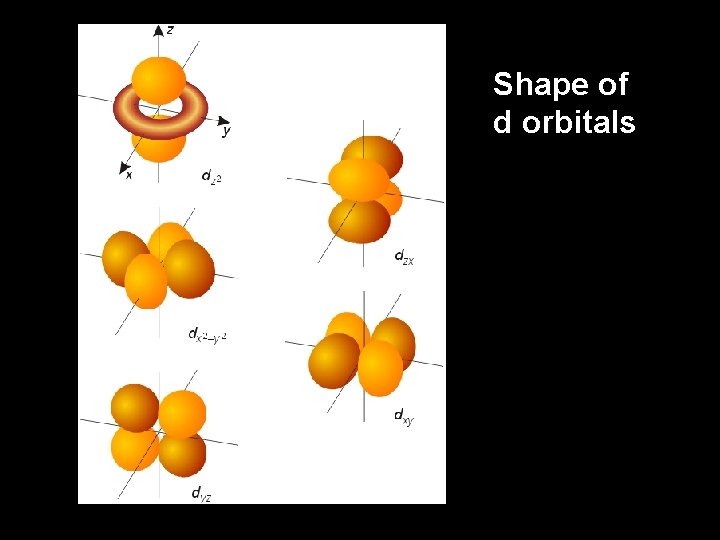
Shape of d orbitals

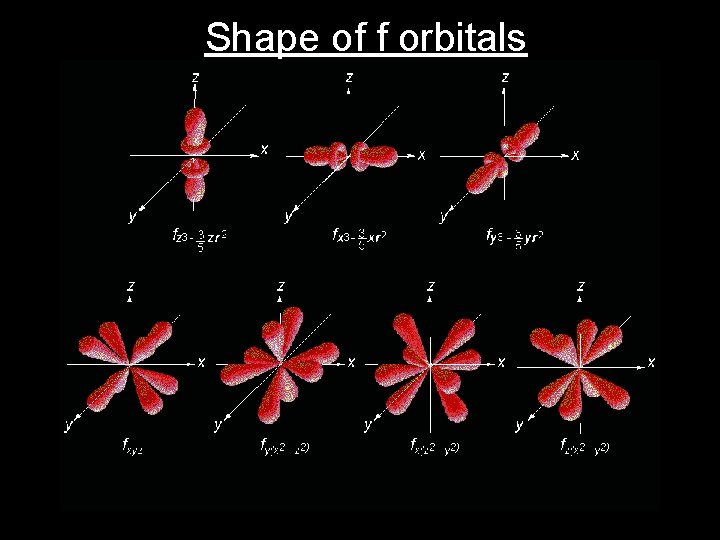
Shape of f orbitals

Relationship to an Atom • Draw the Bohr atom for nitrogen. • What subshells are necessary? – How many orbitals are present?

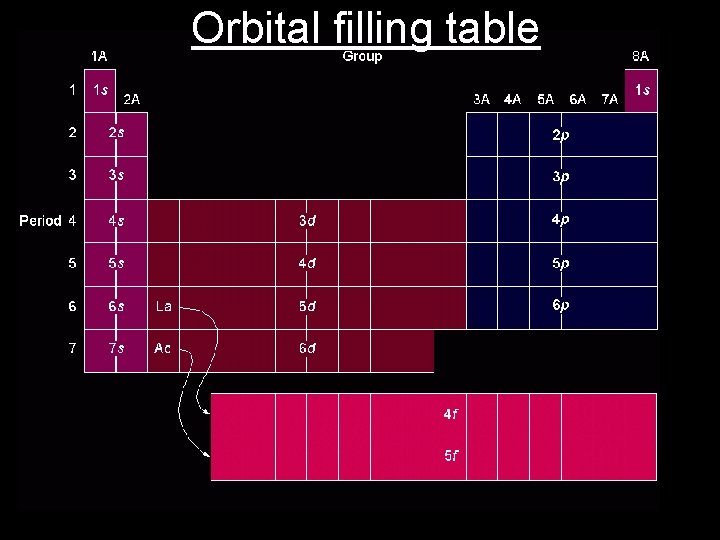
Electron Configurations • Address for electrons • Tells specific shell, subshell type, and # of electrons in that subshell • Electrons always occupy the lowest energy subshell first. – Aufbau principle • 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 6 • What’s up with that 4 s-3 d-4 p thing?

Lets Try a Couple • Write the electron configuration for the following elements • Li, Br, Fe, K • Li = 1 s 22 s 1 • Br = 1 s 22 p 63 s 23 p 64 s 23 d 104 p 5 • Fe = 1 s 22 p 63 s 23 p 64 s 23 d 6 • K = 1 s 22 p 63 s 23 p 64 s 1 • Anything interesting about Li and K?

Valence Electrons • Total number of s and p electrons in last shell • These are the electrons involved in reactions • How many valence electrons are there – Li, Br, Fe, K – Li = 1 – Br = 7 – Fe = 2 –K = 1

Noble Gases • How many valence electrons will noble gases have? • 8 (or 2 if you are helium) • 8 is a stable number of electrons – Octet rule (or duet if you are He) • Use this knowledge to write electron configurations • 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 6

Noble Gas Configuration • Shortened way of writing configurations • Since we always know configurations of noble gases we use them as a starting point. • Electron configuration of P is • P = 1 s 22 p 63 s 23 p 3 • With noble gas configuration choose noble gas that come before which is Ne • P = [Ne] 3 s 23 p 3

You Try It • Write noble gas configuration for • • • Li, Br, Fe, K Li = [He] 2 s 1 Br = [Ar] 4 s 23 d 104 p 5 Fe = [Ar] 4 s 23 d 6 K = [Ar] 4 s 1

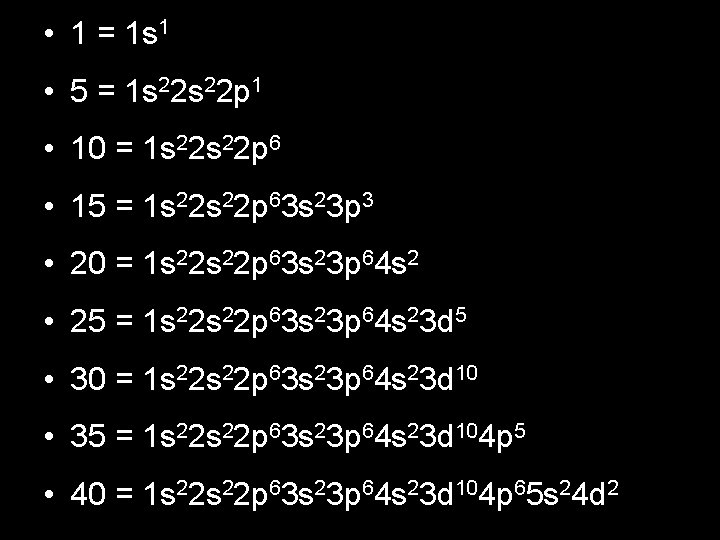
Homework • Write electron configurations for elements 1, 5, 10, 15, 20, 25, 30, 35, 40 • Write noble gas configurations for elements • 1, 5, 10, 15, 20, 25, 30, 35, 40

• 1 = 1 s 1 • 5 = 1 s 22 p 1 • 10 = 1 s 22 p 6 • 15 = 1 s 22 p 63 s 23 p 3 • 20 = 1 s 22 p 63 s 23 p 64 s 2 • 25 = 1 s 22 p 63 s 23 p 64 s 23 d 5 • 30 = 1 s 22 p 63 s 23 p 64 s 23 d 10 • 35 = 1 s 22 p 63 s 23 p 64 s 23 d 104 p 5 • 40 = 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 2
![1 1 s 1 5 He 2 s 22 • 1 = 1 s 1 • 5 = [He] 2 s 22](https://slidetodoc.com/presentation_image_h2/2c35966d38acb8b7bcbfb85f9c665a7b/image-47.jpg)
• 1 = 1 s 1 • 5 = [He] 2 s 22 p 1 • 10 = [He] 2 s 22 p 6 • 15 = [Ne] 3 s 23 p 3 • 20 = [Ar] 4 s 2 • 25 = [Ar]4 s 23 d 5 • 30 = [Ar] 4 s 23 d 10 • 35 = [Ar] 4 s 23 d 104 p 5 • 40 = [Kr] 5 s 24 d 2

Orbital filling table

Electron configuration of the elements of the first three series

Assignment • Use the periodic table to write noble gas configurations for elements • 1, 5, 10, 15, 20, 25, 30, 35, 40
![1 1 s 1 5 He 2 s 22 • 1 = 1 s 1 • 5 = [He] 2 s 22](https://slidetodoc.com/presentation_image_h2/2c35966d38acb8b7bcbfb85f9c665a7b/image-51.jpg)
• 1 = 1 s 1 • 5 = [He] 2 s 22 p 1 • 10 = [He] 2 s 22 p 6 • 15 = [Ne] 3 s 23 p 3 • 20 = [Ar] 4 s 2 • 25 = [Ar]4 s 23 d 5 • 30 = [Ar] 4 s 23 d 10 • 35 = [Ar] 4 s 23 d 104 p 5 • 40 = [Kr] 5 s 24 d 2

Quantum Numbers • Numbers that define an electrons probable position is space • Very similar to electron configurations • 1 extra number • Spin – An electron can only spin in one direction – +1/2 or -1/2

Principle, angular momentum, and magnetic quantum numbers: n, l, and ml

Pauli Exclusion Principle • No two electrons can have the same 4 quantum number • This means when there are two electrons in an orbital they have to be spinning in different directions

Hunds Rule • Every orbital in a subshell is singly occupied with one electron before any one orbital is doubly occupied, and all electrons in singly occupied orbitals spin in the same direction

Orbital Diagrams • A representation of the location and spin of electrons within an atom • Process – Write the electron configuration – Place bars above configuration to represent the number of orbitals • s = 1 p=3 d=5 – Place arrows on the bars to represent the spin

• Example Draw the orbital diagram for Be Nitrogen Cobalt

Ionization Energy Revisited • The trend for ionization energy has some exceptions • Use orbital diagrams to explain the trend


Unpaired Electrons • Electrons that are by themselves in an orbital • How many unpaired electrons are there in a oxygen atom? • Unpaired electrons make an atom magnetic – Paramagnetic

Problems • P. 156 #’s 81, 84 and orbital diagram
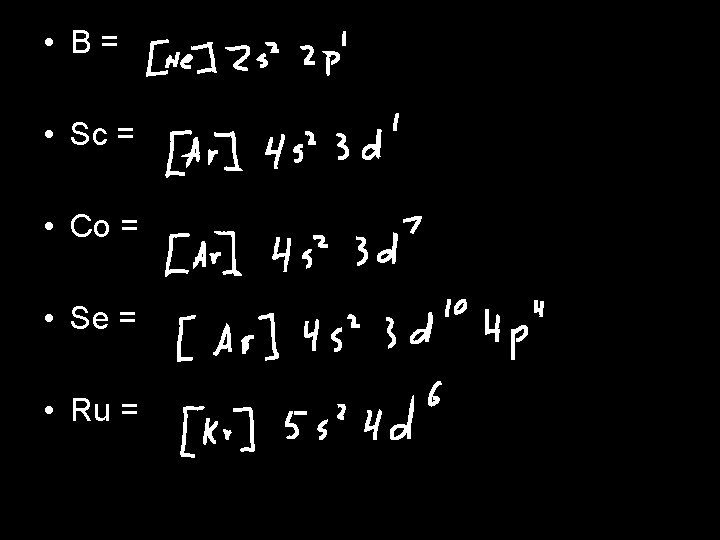
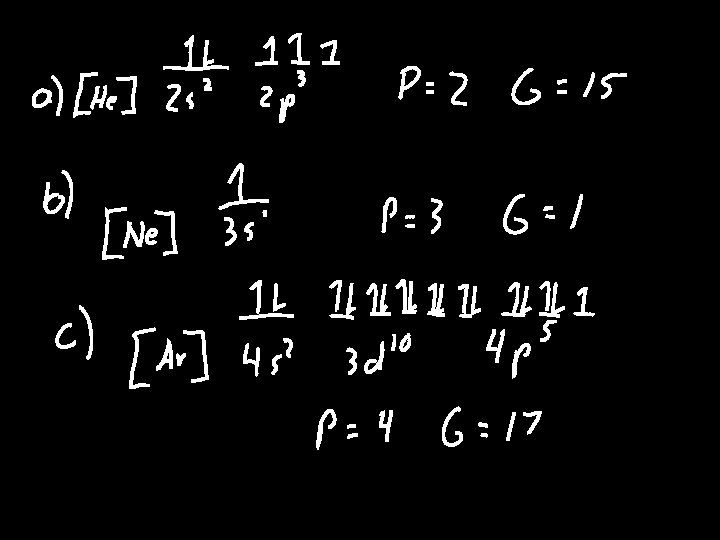

Problems • P. 114 #’s 95, 96, 99, 100, 103, 110 • P. 157 #’s 122, 124, 126, 129, 131, 134, 139

Problems • P. 114 #’s 95, 96, 99, 100, 103, 110