Chapter 4 The First Law of Thermodynamics for












- Slides: 20

Chapter 4 The First Law of Thermodynamics: for Control Volume Assoc. Prof. Dr. Sommai Priprem Department of Mechanical Engineering Khon Kaen University ��. �������

Topics n Analysis of control volume n Conservation of mass principle n Flow Work n Total Energy of flowing fluid n 1 st Law for a control volume (general) n 1 st Law for SSSF Processes & Applications n 1 st Law for USUF Processes & Applications ��. �������

4. 1 Thermodynamics Analysis of Control Volume n A large number of engineering problem involves mass flows in and out of system n Therefore, they have to analyzed using Control Volume (Open System) approach. n Boundary of the control volume is called “Control Surface” n Control Surface can be FIX or MOVABLE ��. �������

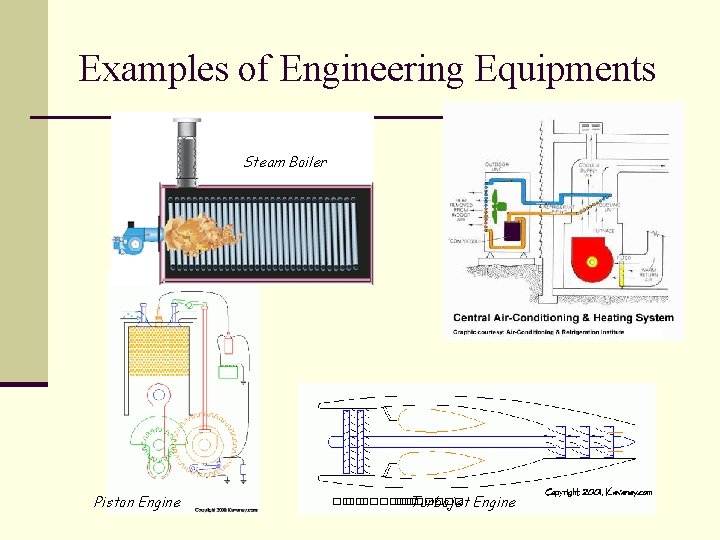
Examples of Engineering Equipments Steam Boiler Piston Engine ��. ������� Turbojet Engine

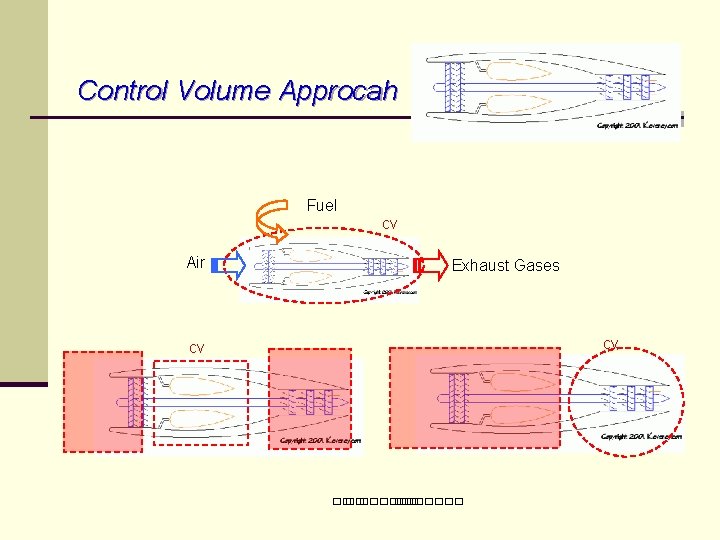
Control Volume Approcah Fuel cv Air Exhaust Gases cv cv ��. �������

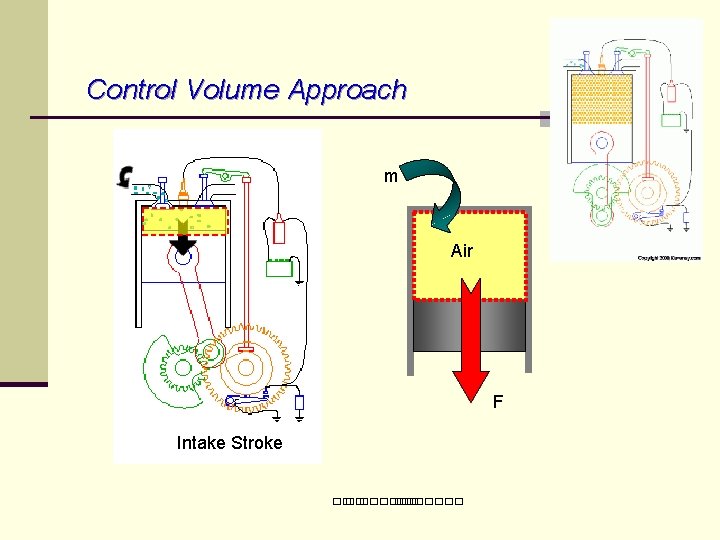
Control Volume Approach m Air F Intake Stroke ��. �������

Some Type of FLOW n Steady = no change with TIME (opposite to : Unsteady or Transient) n Uniform = no change with LOCATION over a specified region n Unsteady = Transient = Change over time and location ��. �������

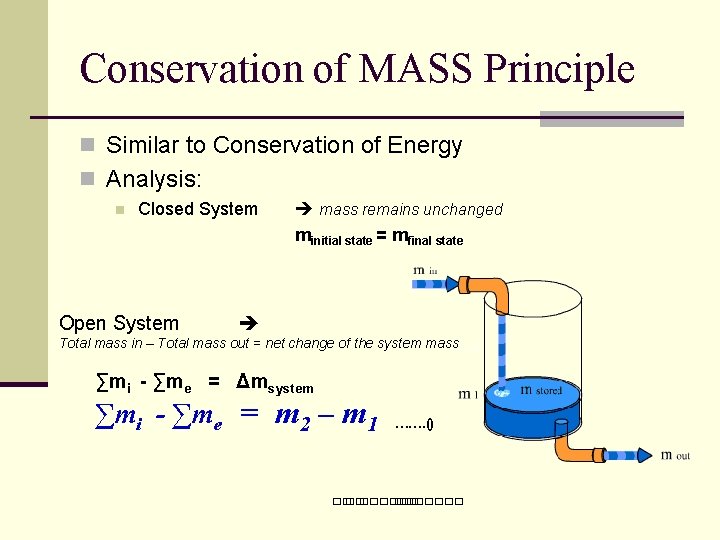
Conservation of MASS Principle n Similar to Conservation of Energy n Analysis: n Closed System Open System mass remains unchanged minitial state = mfinal state Total mass in – Total mass out = net change of the system mass ∑mi - ∑me = Δmsystem ∑mi - ∑me = m 2 – m 1 ……. () ��. �������

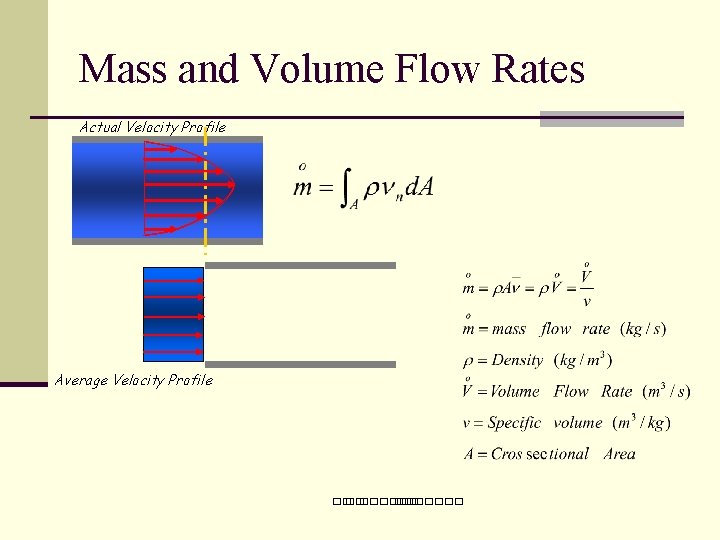
Mass and Volume Flow Rates Actual Velocity Profile Average Velocity Profile ��. �������

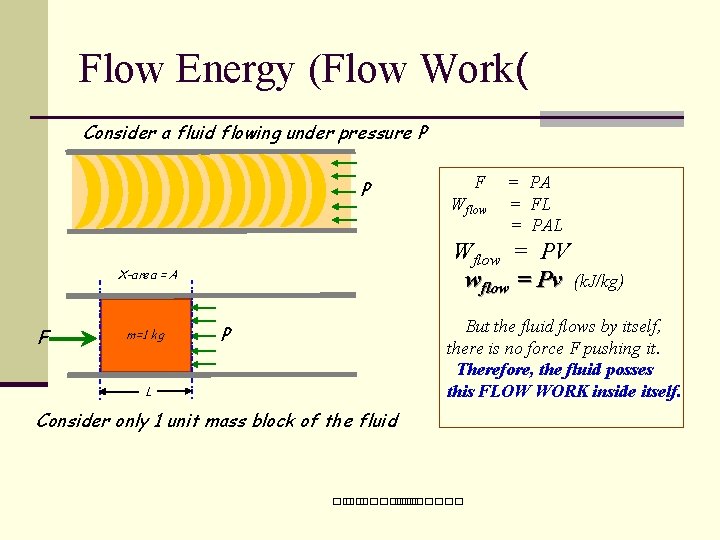
Flow Energy (Flow Work( Consider a fluid flowing under pressure P P m=1 kg Wflow = PA = FL = PAL Wflow = PV wflow = Pv (k. J/kg) X-area = A F F But the fluid flows by itself, there is no force F pushing it. Therefore, the fluid posses this FLOW WORK inside itself. P L Consider only 1 unit mass block of the fluid ��. �������

Total Energy of Fluid n for non-flow mass inside control volume (cv) n enonflow = u + ke + pe k. J/kg J Enonflow = m(u + ½ V 2 + zg) n for flowing fluid mass flowing in/out of cv. n n eflow = enonflow+ flow work eflow = u + Pv + ke + pe defined: h = u + Pv n eflow = h + ke + pe n Eflow = m(h + ½ V 2 + zg) ��. ������� k. J/kg J

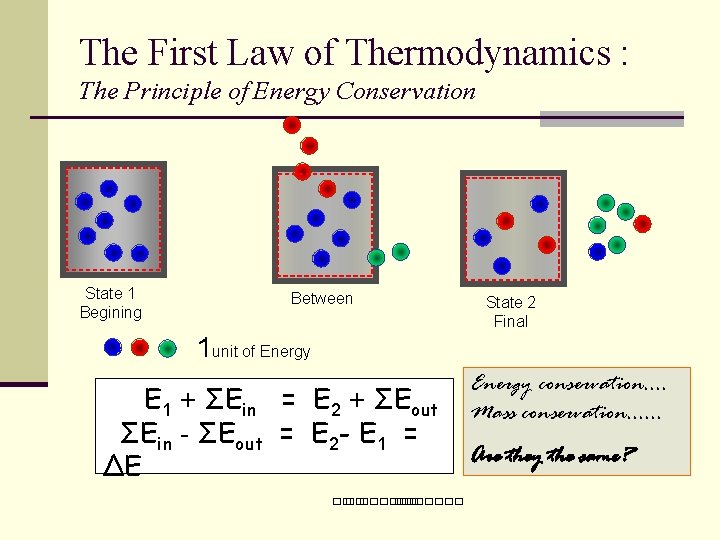
The First Law of Thermodynamics : The Principle of Energy Conservation State 1 Begining Between State 2 Final 1 unit of Energy E 1 + ΣEin = E 2 + ΣEout ΣEin - ΣEout = E 2 - E 1 = ΔE ��. ������� Energy conservation. . Mass conservation. . . Are they the same?

The First Law of Thermodynamics : Open system Heat Engine System Model: Add heat to system, Qin System gives WORK, Wout +Q +W From ) ΣEin = ΣEout + (E 2 - E 1 ΣEin = ΣQin + ΣWin + ΣEflow-in ΣEout = ΣQout + ΣWout + ΣEflow-out Let Q = Σ(Qin - Qout) W = Σ(Wout-Win) Q + ∑ Eflow-in = W + ∑ Eflow-out + (E 2 -E 1) What is the difference from 1 st law for Closed Systems ��. �������

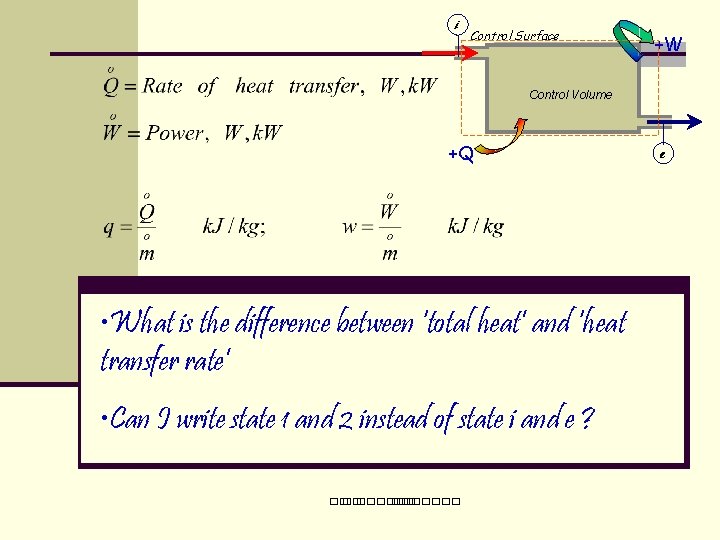
1 st Law for a Control Volume (General) i Control Volume +W m 2 m 1 +Q e Q + ∑Eflow-in = W + ∑Eflow-out+ (E 2 -E 1) Subscripts: i = at inlet, e = at exit 1 = at time start, 2 = at time end ��. �������

The Steady-Flow Process n Many devices operate under steady-flow process. n for a steady-flow process Properties within cv. not change with time, no boundary work involves n Properties at boundary not change with time, but can be different at different Location in cv. n Q and W also unchange with time their Rates are constant. n Mass inside cv. constant Δmsys=0 n ��. �������

1 st Law for Steady-Flow Process 0 Qcv + ∑mi(h + ½ V 2 +zg)i = Wcv + ∑me(h + ½ V 2 +zg)e + (E 2 – E)1 ∑mi - ∑me ∑mi Subscripts: = m 2 – m 1 0 = ∑me i = at inlet, e = at exit 1 = at time start, 2 = at time end Qcv + ∑mi(h + ½ V 2 +zg)i = Wcv + ∑me(h + ½ V 2 +zg)e ��. �������

SSSF 1 -inlet and 1 -exit i Control Surface +W Control Volume +Q ��. ������� e

i Control Surface +W Control Volume +Q • What is the difference between ‘total heat’ and ‘heat transfer rate’ • Can I write state 1 and 2 instead of state i and e ? ��. ������� e

Exercise – Group work n. Write 1 st law equation for SSSF Process starts from the concept of ‘Energy Conservation Principle’ ��. �������

End of Part 1 Read text and do some exercises please. Sommai ��. �������