Chapter 4 The Chemical Basis of Life 4


Chapter 4 The Chemical Basis of Life

4. 1 Life requires about 25 chemical elements… n. A. Matter n. Anything that occupies space and has mass n 3 states: solid, liquid, gas

4. 1 Elements & Compounds… n a. Elements n. Pure substance, simplest form n. Made up of 1 type of atom n. Ex: Hydrogen (H), oxygen (O), nitrogen (N) (~110 elements on Periodic Table)

Open your Agenda…. n. Page 124
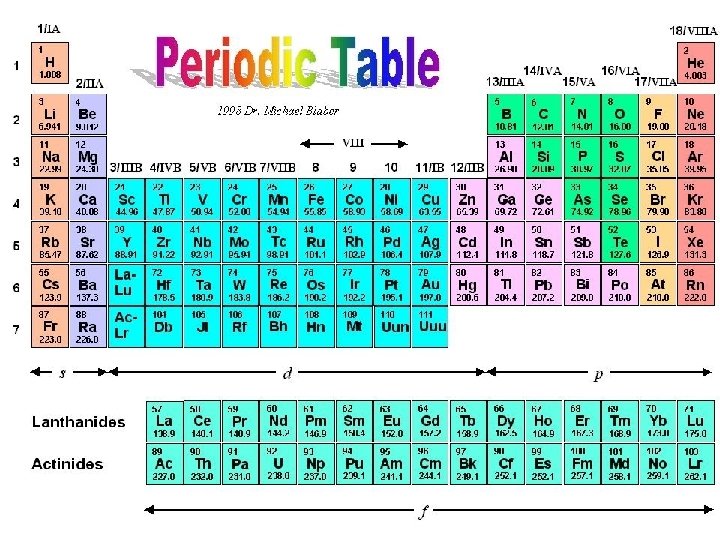

4. 1 Elements & Compounds… n. Compound n 2 or more elements n. Chemically combined in a fixed ratio n. Written as formulas n. Ex: water – H 2 O carbon dioxide – CO 2

4. 1 Elemental Composition of the Human Body Chemical Symbol O C H N Ca P K Mass (70 Kg person) 43 Kg 16 Kg 7 Kg 1. 8 Kg 1 Kg 780 g 140 g Trace elements make up <0. 01% of your body

Important Elements in Life…. C HOPKINS Ca. Fe Mg Na. Cl (What elements are represented here? )

The Most Important Elements in Life…. HONC (What elements are represented here? )

4. 2 What are atoms? n. Basic building blocks of matter (A desk, the air, you are made up of atoms!)
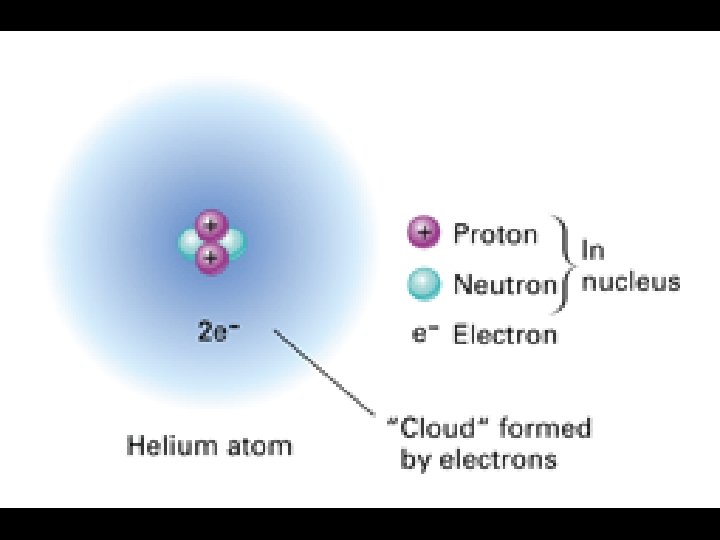

4. 2 Atoms are made out of 3 basic particles… n. Electrons nnegatively charged particles that move around the nucleus in orbitals e

4. 2 The other 2 particles are found within the nucleus… n. Nucleus – center of the atom n. Proton – positively charged particle n. Neutron – neutral particle
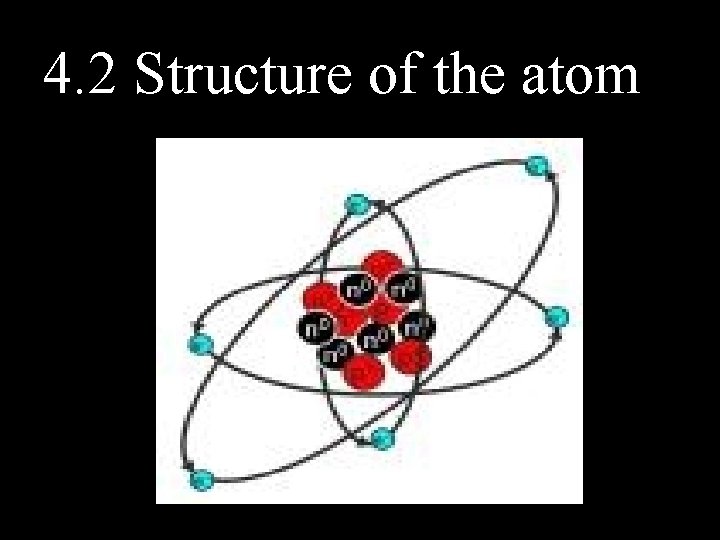
4. 2 Structure of the atom

4. 3 Chemical bonds join atoms n. Bonds hold atoms together n 2 types of bonds: n. Covalent n. Ionic

4. 3 Covalent Bond n. A bond formed when atoms SHARE electrons n 2 or more atoms held together by covalent bonds form a molecule
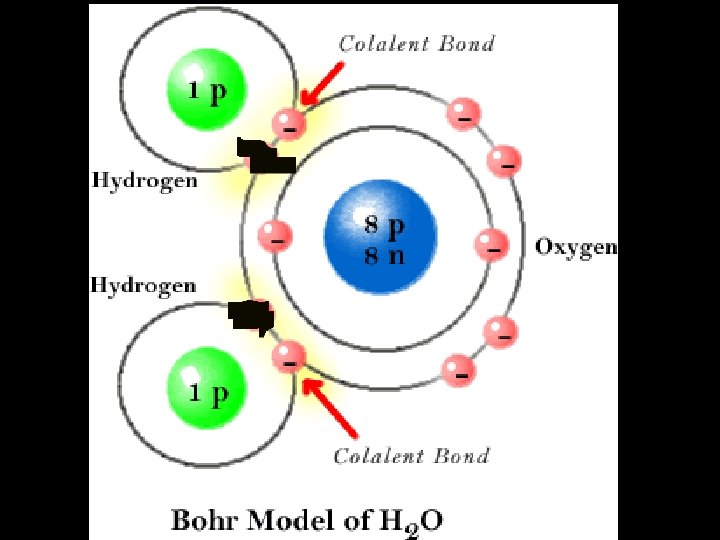

4. 3 Ionic Bond n. A bond formed when atoms TRANSFER electrons. n. One atom LOSES electrons; another GAINS electrons. n. An ION is formed
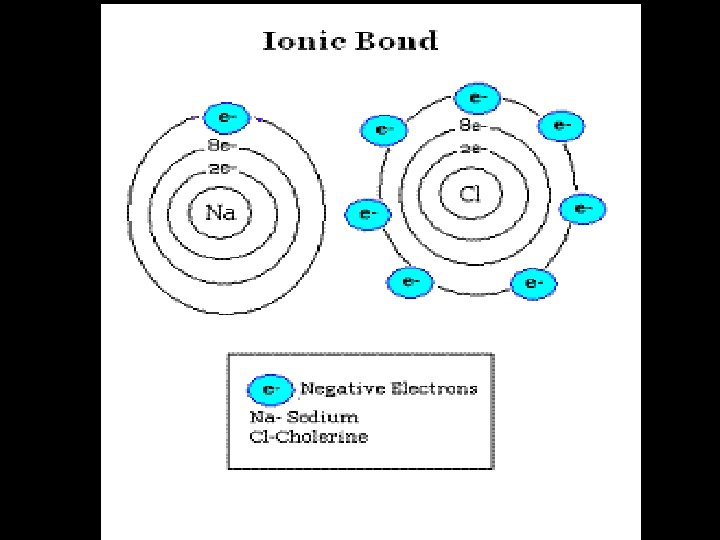

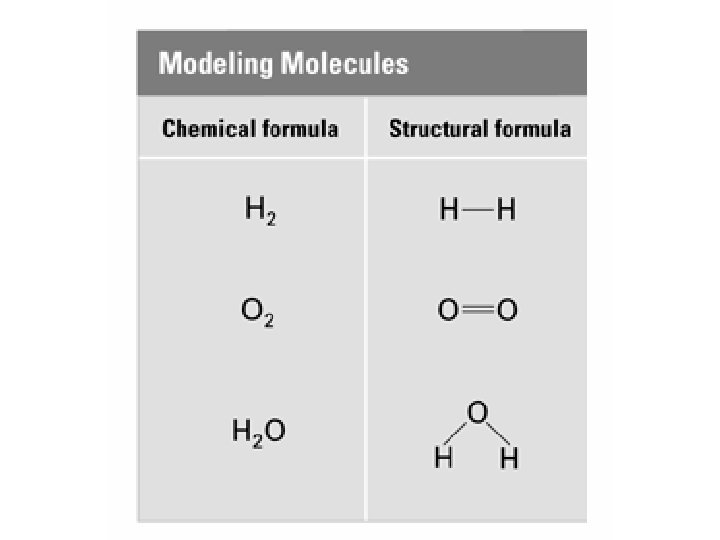
4. 3 Chemical Formulas… Formulas n. Molecular (Chemical): shows the kind of atoms and the number of each n. Structural: Structural shows the arrangement of atoms in a compound (the bonds)
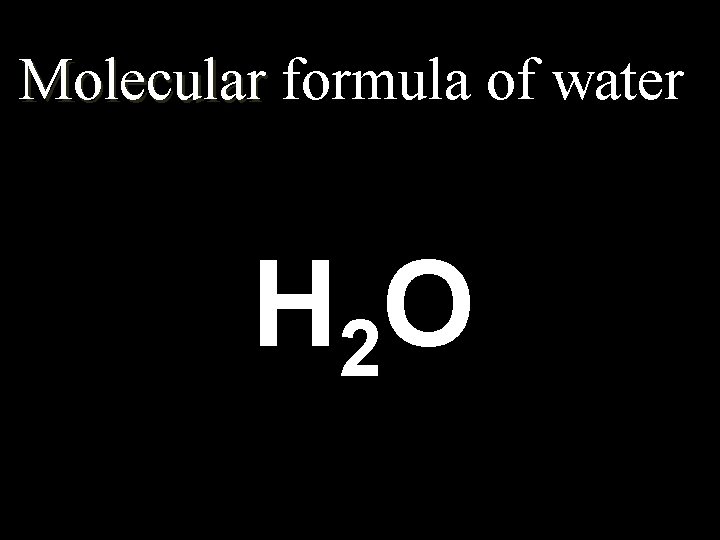
Molecular formula of water H 2 O
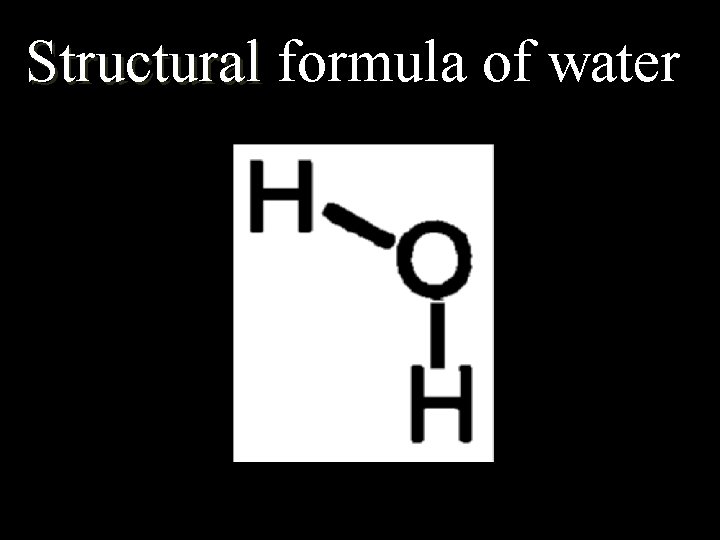
Structural formula of water
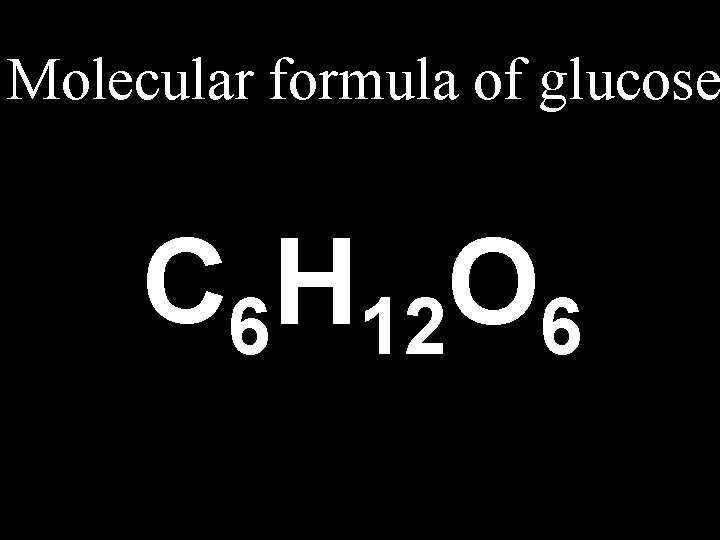
Molecular formula of glucose C 6 H 12 O 6

Structural formula of glucose
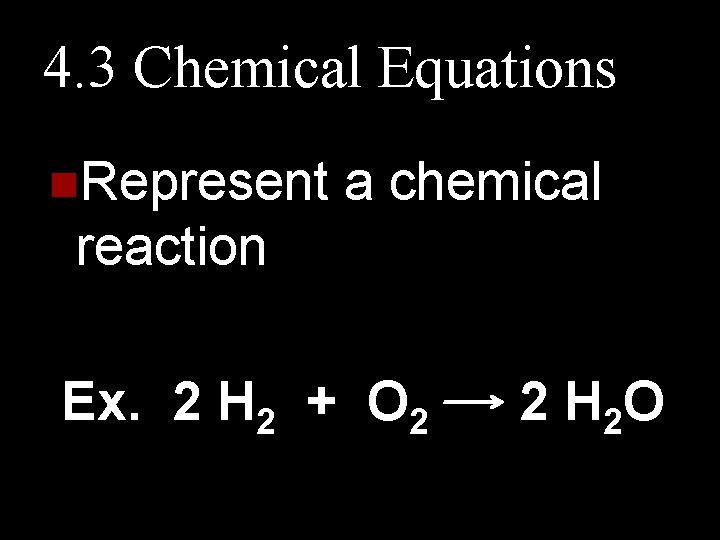
4. 3 Chemical Equations n. Represent a chemical reaction Ex. 2 H 2 + O 2 2 H 2 O

4. 3 Reactants & Products n. Reactants = the chemicals on the LEFT side of the equation n. Products = the chemicals on the RIGHT side of the equation (are made or produced in the reaction)

4. 3 Balanced Equations n. Number of each atom on the right equals the number on the left.
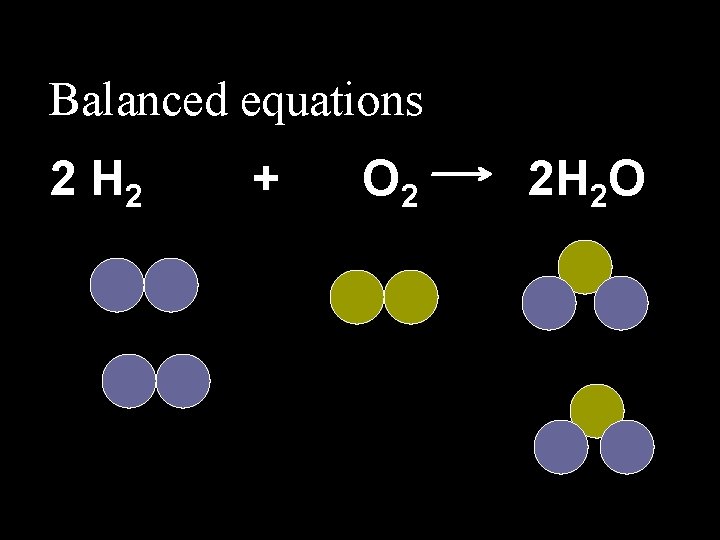
Balanced equations 2 H 2 + O 2 2 H 2 O
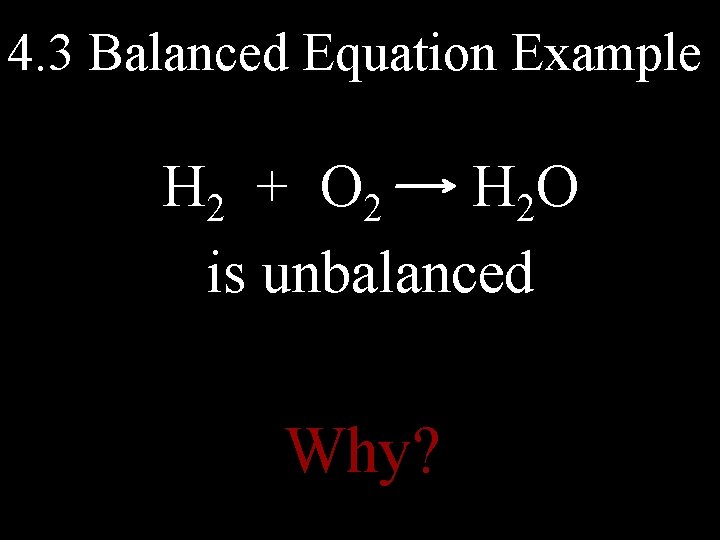
4. 3 Balanced Equation Example H 2 + O 2 H 2 O is unbalanced Why?

4. 3 Balanced Equations n. To BALANCE equations, numbers called COEFFICIENTS are added in front of the compounds.

Chemistry of Life Movie Link Start at 2: 30 End at 8: 48
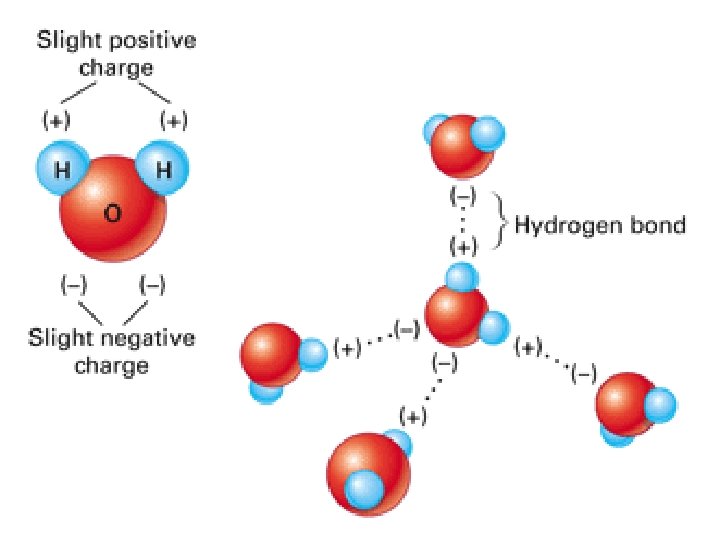

4. 4 Unique properties of water n. Cohesion – molecules of the same kind “stick” to one another n. Adhesion – attraction between unlike molecules n. Ex: meniscus

4. 4 Unique properties of water n. Cohesion n Trees & Adhesion: depend on these forces!! Why? Water evaporates, pulls another water up (cohesion) from leaves down stems into roots!

H 2 O Adhesion & Cohesion

4. 4 Water dissolves other substances n. Solutions are formed from a uniform mixture of 2 or more substances n. Ex. Dissolving salt into water

4. 4 What makes up a solution? n. Solvent – substance that dissolves the other and is in greater quantity n. Water = universal solvent

4. 4 What makes up a solution? ? n. Solute – substance that is dissolved and is present in lesser amounts n. Ex. salt

4. 4 What makes up a solution? n. When water is the solvent it forms an aqueous solution

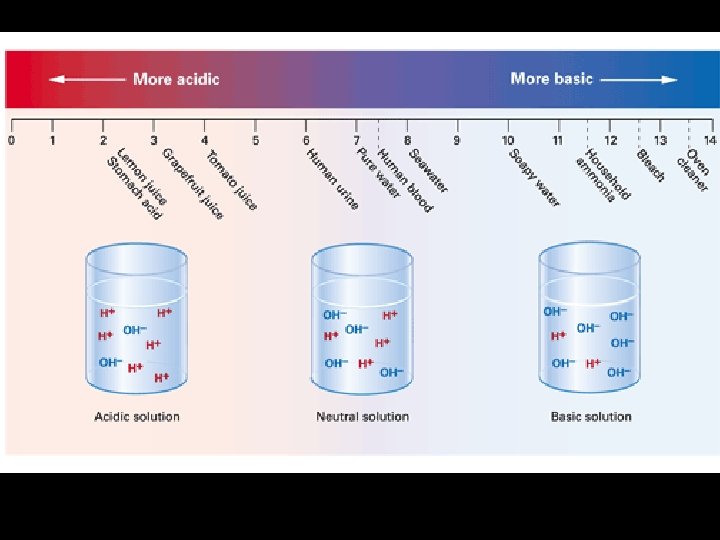
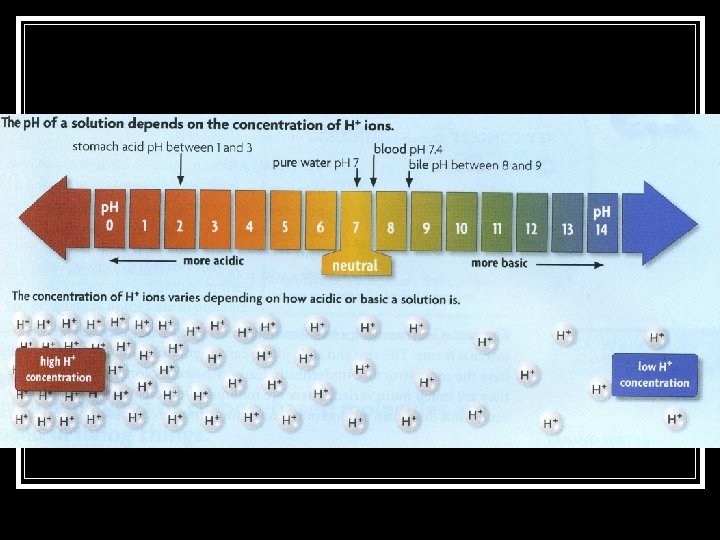
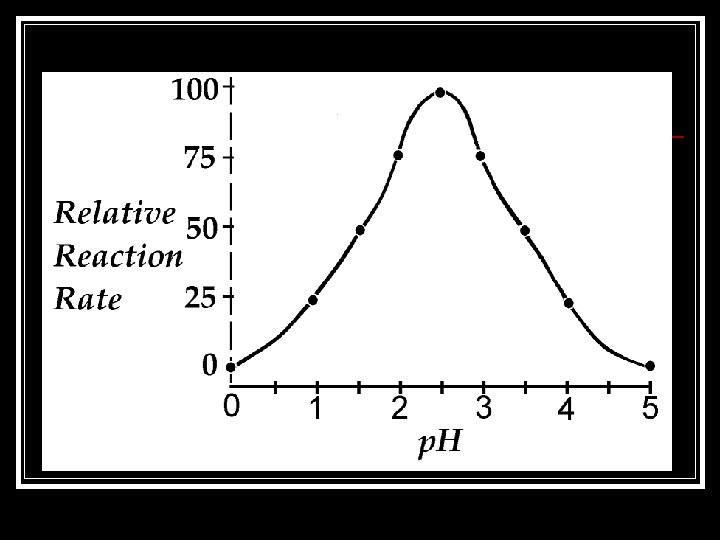
Note Packet Page 4: 4. 4 The p. H Scale n. It is a measure of the concentration of hydrogen ions in a solution. n. It ranges from 0 to 14. Neutral _________________________________ 0 Acids 7 Bases 14

4. 4 The p. H Scale n. Hydrogen ions + (lost an electron) H n. Hydroxide ions (gained an electron) OH

4. 4 Acids… np. H range – less than 7 n. Examples: n. HCl (hydrochloric acid) n. Vinegar (acetic acid) n. Lemon juice (citric

4. 4 Acids… Have more H+ ions than water does. n. Have fewer OH- ions n

4. 4 Bases… np. H range–greater than 7 n. Examples: n. Na. OH (sodium hydroxide) n. Ca(OH)2 (calcium hydroxide) n. NH 3 (ammonia)
4. 4 Bases… n. Have fewer H+ ions than water does n. Have many OH ions

4. 4 Neutral Substances… np. H – equals 7 n. Example: n. Pure water (H 2 O)

4. 4 Buffers… n. Helps maintain constant p. H + n. Accepts or donates H ions n. Many biological fluids contain buffers!! Why? ?

4. 4 Indicators… n. Chemicals that turn different colors in different p. H values n. Bromothymol Blue, Phenolphthalein, p. H paper n. DEMO: take notes on page 8 of your observations!

Chemistry of Life Movie Link Start at 2: 30 End at 8: 48
- Slides: 54